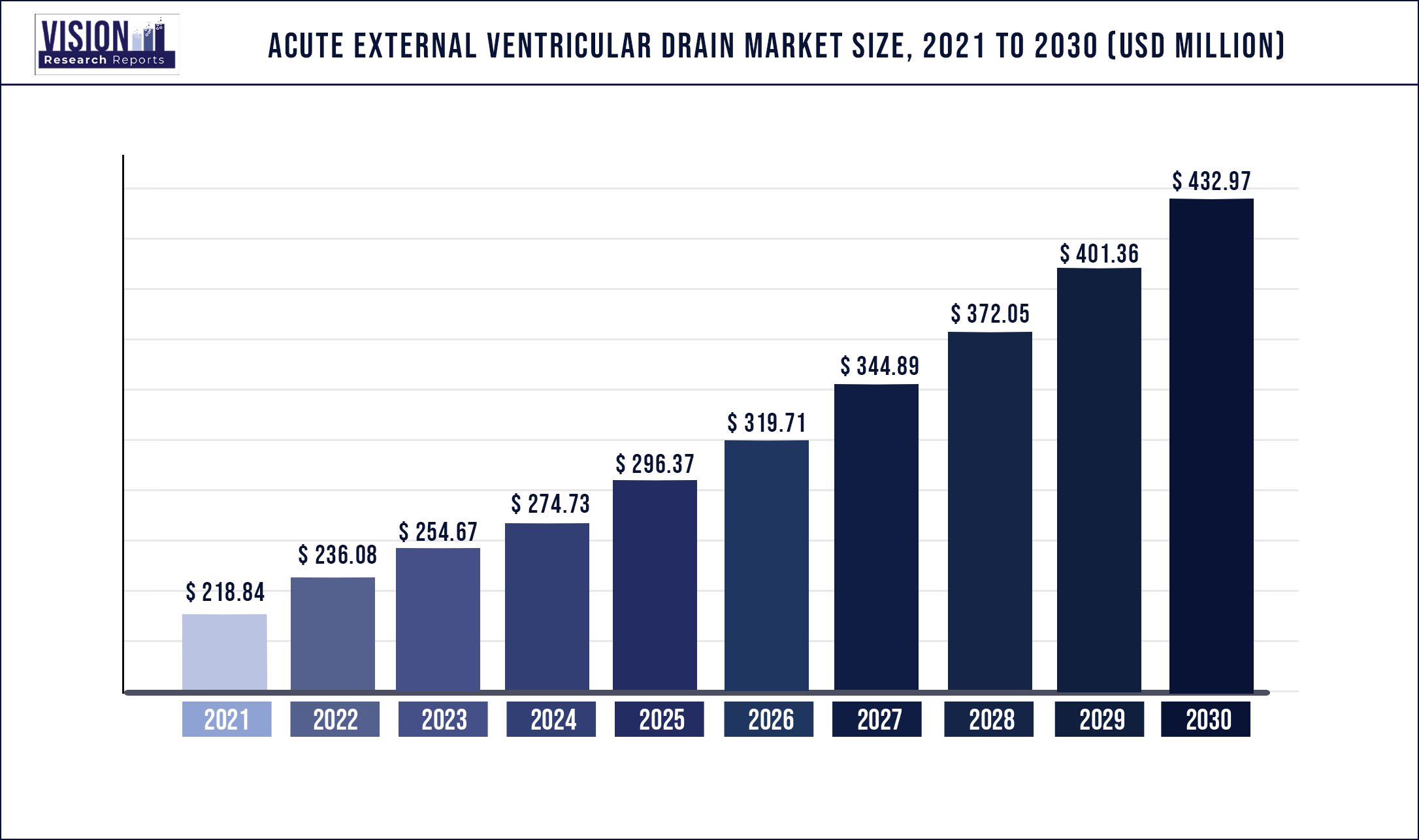
The global acute external ventricular drain market size was estimated at around USD 218.84 million in 2021 and it is projected to hit around USD 432.97 million by 2030, growing at a CAGR of 7.88% from 2022 to 2030
Report Highlights
Rising adoption of conventional over the traditional method, increasing prevalence of acute hydrocephalus, and increasing adoption of technologically advanced devices for acute external ventricular drain are driving the industry’s growth over the forecast period.
EVDs provide benefits for the treatment of acute hydrocephalus. Hydrocephalus is a medical emergency that requires the placement of an EVD into one of the brain's lateral ventricles to drain excess CSF. Each year, 740,000 instances of hydrocephalus in LMICs and about 30,000 in HICs require neurosurgery. Similarly, according to Hydrocephalus Association, hydrocephalus affects about 1 million Americans.
As per the same source, one in every 770 babies develops hydrocephalus in the U.S. Intraventricular hemorrhage is the most frequent cause of acquired hydrocephalus in babies, and studies have indicated that early intervention with an EVD (at the age of 25 days or younger) leads to improved neurological results. Since EVDs are most used in the abovementioned conditions, these factors have directly impacted the industry.
Furthermore, government initiatives such as grant allocation and approval, and investments to identify the most effective care and treatments to aid patient recovery after a brain injury are being implemented in various countries. According to the Hydrocephalus Association, approximately USD 12 million in research was invested in 2021, with an additional USD 35 million in federal funding provided for the study and development of hydrocephalus disease care in Canada.
Additionally, in April 2022, researchers were given a US 2.85 Mn NIH grant to explore a novel method for evaluating cerebrospinal fluid in infants with hydrocephalus. As a result of the aforementioned factors, the market is predicted to increase significantly in the near future.
Scope of The Report
| Report Coverage | Details |
| Market Size in 2021 | USD 218.84 million |
| Revenue Forecast by 2030 | USD 432.97 million |
| Growth rate from 2022 to 2030 | CAGR of 7.88% |
| Base Year | 2021 |
| Forecast Period | 2022 to 2030 |
| Segmentation | Application, patient type, region |
| Companies Covered | Medtronic; Integra LifeSciences Corporation; Natus Medical Incorporated; Fuji Systems; Spiegelberg GmbH; SOPHYSA; Neuromedex |
Application Insights
Based on application, the traumatic brain injury segment held the largest market share of 37.81% in 2021 and is expected to grow at the fastest CAGR from 2022 to 2030. Every year, more than 60 million new cases of traumatic brain injury are reported worldwide. The most common causes are car accidents and falls, both of which are on the rise.
TBI is associated with intracranial hemorrhage, which increases the risk of mortality and disability. The severity of such an injury might range from “mild,” which is defined as a transient change in mental status or consciousness, to “severe,” which is defined as an extended period of amnesia or unconsciousness following the damage. A TBI can cause short- or long-term issues with independent functions.
According to CDC, the number of TBIs has increased considerably over recent years. Based on the recent data, there were 16,070 TBI-related hospitalizations among children (birth to 17 years old) in 2019 and 2,774 TBI-related deaths in 2020. Moreover, the number and rate of TBI-related hospitalizations and deaths were higher among people aged 75 and over. This age group is responsible for around 28% of TBI-related deaths and 32% of TBI-related hospitalizations.
Thus, a high incidence of traumatic brain injuries leads to intraventricular bleeding (hemorrhage), which in turn demands the use of acute EVD products to remove excess cerebrospinal fluid from the brain. This in turn is expected to drive the market over the forecast period.
Patient Type Insights
The adult patient type segment held the largest revenue share of 84.03% in 2021. The prevalence of hydrocephalus has risen dramatically over time, raising the demand for EVD devices as a treatment option in adults. The increased frequency of neurological illnesses due to road accidents, falls, and violence all contribute to the external ventricular drain market's rapid expansion. For instance, as per the European Injury Database (IDB), approximately 6.1 million people are treated in hospitals for injuries every year. These injuries include facial and traumatic brain injuries.
Similarly, according to the CDC report, an estimated 56,000 older are hospitalized every year as a result of head injuries suffered due to falls and 775,000 older adults are living with TBI-related disabilities. This is expected to fuel segment growth during the forecast period. Moreover, growing research activities on diagnosis and surgical management along with a number of new pipelines of products are further boosting the industry growth.
The pediatric patient type segment is expected to grow at the fastest CAGR from 2022 to 2030. The increased frequency of neurological illnesses and other medical conditions, such as subarachnoid hemorrhages, hydrocephalus, intracranial hypertension, head injury, and inflammatory diseases of the cerebrospinal space, are expected to drive the segment.
Hydrocephalus affects one to two out of every 1,000 babies, according to the National Institute of Neurological Disorders and Stroke (NINDS). When the flow of cerebrospinal fluid inside the brain is impeded, an external ventricular drain device is used in neurosurgery to treat and alleviate elevated intracranial pressure and hydrocephalus. During the forecast period, this is expected to fuel market expansion.
Furthermore, various technological advancements and innovations, extensive research and development activities, and a large volume of clinical trials being conducted for the development of traumatic brain injuries and other diseases, as well as an increase in the number of falls and accident cases worldwide are expected to propel the segment growth.
Regional Insights
North America dominated the overall Acute EVD market in 2021 with a revenue share of around 35.6%. This can be attributed to the high prevalence of TBI and the high incidence of neurovascular illnesses in this region. Moreover, an increase in the number of FDA approvals for acute external ventricular drain products in clinical applications such as head injury, hydrocephalus, subarachnoid hemorrhages, and other diseases of cerebrospinal space are responsible for fueling the regional growth. In addition, increasing government funding and growing initiatives for raising awareness about neurological disorders are among the factors expected to drive the demand for external ventricular drain in this region.
In addition, favorable reimbursement structure and rising healthcare expenditure are factors contributing to the growth of the industry in the country. For instance, according to statistics published on CMS.gov, U.S. healthcare expenditure increased by 9.7% in 2020, reaching USD 4.1 trillion per person and accounting for a 19.7% share of the nation’s GDP. Thus, these factors are likely to boost the growth in the U.S. Furthermore, the favorable reimbursement structure is a major factor contributing to the growth.
Asia Pacific is expected to register the highest CAGR during the forecast period. Asia Pacific acute external ventricular drain market is primarily driven by the increasing incidence of sports injuries & trauma. Emerging economies, such as China, Japan, South Korea, and India, are expected to witness considerable market growth over the forecast period. The presence of a large patient pool and the growing need for technologically advanced & cost-efficient healthcare solutions are among the factors expected to present significant regional growth opportunities.
Moreover, the market is driven by high-impact R&D investments made by international businesses to reach underserved markets in Asia Pacific as a result of low-cost services and an increase in clinical trials. Other factors supporting market expansion in the region include an increase in hospital admissions and improvements in clinical development framework in developing nations. However, the region's expansion could be hampered by a lack of advanced healthcare facilities, a shortage of experienced experts, and a bad reimbursement environment.
Key Players
Market Segmentation
Chapter 1. Introduction
1.1. Research Objective
1.2. Scope of the Study
1.3. Definition
Chapter 2. Research Methodology
2.1. Research Approach
2.2. Data Sources
2.3. Assumptions & Limitations
Chapter 3. Executive Summary
3.1. Market Snapshot
Chapter 4. Market Variables and Scope
4.1. Introduction
4.2. Market Classification and Scope
4.3. Industry Value Chain Analysis
4.3.1. Raw Material Procurement Analysis
4.3.2. Sales and Distribution Channel Analysis
4.3.3. Downstream Buyer Analysis
Chapter 5. COVID 19 Impact on Acute External Ventricular Drain Market
5.1. COVID-19 Landscape: Acute External Ventricular Drain Industry Impact
5.2. COVID 19 - Impact Assessment for the Industry
5.3. COVID 19 Impact: Global Major Government Policy
5.4. Market Trends and Opportunities in the COVID-19 Landscape
Chapter 6. Market Dynamics Analysis and Trends
6.1. Market Dynamics
6.1.1. Market Drivers
6.1.2. Market Restraints
6.1.3. Market Opportunities
6.2. Porter’s Five Forces Analysis
6.2.1. Bargaining power of suppliers
6.2.2. Bargaining power of buyers
6.2.3. Threat of substitute
6.2.4. Threat of new entrants
6.2.5. Degree of competition
Chapter 7. Competitive Landscape
7.1.1. Company Market Share/Positioning Analysis
7.1.2. Key Strategies Adopted by Players
7.1.3. Vendor Landscape
7.1.3.1. List of Suppliers
7.1.3.2. List of Buyers
Chapter 8. Global Acute External Ventricular Drain Market, By Application
8.1. Acute External Ventricular Drain Market, by Application, 2022-2030
8.1.1. Traumatic Brain Injury
8.1.1.1. Market Revenue and Forecast (2017-2030)
8.1.2. Subarachnoid Hemorrhage
8.1.2.1. Market Revenue and Forecast (2017-2030)
8.1.3. Intracerebral Hemorrhage
8.1.3.1. Market Revenue and Forecast (2017-2030)
8.1.4. Other Non-traumatic Hydrocephalus Conditions
8.1.4.1. Market Revenue and Forecast (2017-2030)
Chapter 9. Global Acute External Ventricular Drain Market, By Patient Type
9.1. Acute External Ventricular Drain Market, by Patient Type, 2022-2030
9.1.1. Pediatric
9.1.1.1. Market Revenue and Forecast (2017-2030)
9.1.2. Adult
9.1.2.1. Market Revenue and Forecast (2017-2030)
Chapter 10. Global Acute External Ventricular Drain Market, Regional Estimates and Trend Forecast
10.1. North America
10.1.1. Market Revenue and Forecast, by Application (2017-2030)
10.1.2. Market Revenue and Forecast, by Patient Type (2017-2030)
10.1.3. U.S.
10.1.3.1. Market Revenue and Forecast, by Application (2017-2030)
10.1.3.2. Market Revenue and Forecast, by Patient Type (2017-2030)
10.1.4. Rest of North America
10.1.4.1. Market Revenue and Forecast, by Application (2017-2030)
10.1.4.2. Market Revenue and Forecast, by Patient Type (2017-2030)
10.2. Europe
10.2.1. Market Revenue and Forecast, by Application (2017-2030)
10.2.2. Market Revenue and Forecast, by Patient Type (2017-2030)
10.2.3. UK
10.2.3.1. Market Revenue and Forecast, by Application (2017-2030)
10.2.3.2. Market Revenue and Forecast, by Patient Type (2017-2030)
10.2.4. Germany
10.2.4.1. Market Revenue and Forecast, by Application (2017-2030)
10.2.4.2. Market Revenue and Forecast, by Patient Type (2017-2030)
10.2.5. France
10.2.5.1. Market Revenue and Forecast, by Application (2017-2030)
10.2.5.2. Market Revenue and Forecast, by Patient Type (2017-2030)
10.2.6. Rest of Europe
10.2.6.1. Market Revenue and Forecast, by Application (2017-2030)
10.2.6.2. Market Revenue and Forecast, by Patient Type (2017-2030)
10.3. APAC
10.3.1. Market Revenue and Forecast, by Application (2017-2030)
10.3.2. Market Revenue and Forecast, by Patient Type (2017-2030)
10.3.3. India
10.3.3.1. Market Revenue and Forecast, by Application (2017-2030)
10.3.3.2. Market Revenue and Forecast, by Patient Type (2017-2030)
10.3.4. China
10.3.4.1. Market Revenue and Forecast, by Application (2017-2030)
10.3.4.2. Market Revenue and Forecast, by Patient Type (2017-2030)
10.3.5. Japan
10.3.5.1. Market Revenue and Forecast, by Application (2017-2030)
10.3.5.2. Market Revenue and Forecast, by Patient Type (2017-2030)
10.3.6. Rest of APAC
10.3.6.1. Market Revenue and Forecast, by Application (2017-2030)
10.3.6.2. Market Revenue and Forecast, by Patient Type (2017-2030)
10.4. MEA
10.4.1. Market Revenue and Forecast, by Application (2017-2030)
10.4.2. Market Revenue and Forecast, by Patient Type (2017-2030)
10.4.3. GCC
10.4.3.1. Market Revenue and Forecast, by Application (2017-2030)
10.4.3.2. Market Revenue and Forecast, by Patient Type (2017-2030)
10.4.4. North Africa
10.4.4.1. Market Revenue and Forecast, by Application (2017-2030)
10.4.4.2. Market Revenue and Forecast, by Patient Type (2017-2030)
10.4.5. South Africa
10.4.5.1. Market Revenue and Forecast, by Application (2017-2030)
10.4.5.2. Market Revenue and Forecast, by Patient Type (2017-2030)
10.4.6. Rest of MEA
10.4.6.1. Market Revenue and Forecast, by Application (2017-2030)
10.4.6.2. Market Revenue and Forecast, by Patient Type (2017-2030)
10.5. Latin America
10.5.1. Market Revenue and Forecast, by Application (2017-2030)
10.5.2. Market Revenue and Forecast, by Patient Type (2017-2030)
10.5.3. Brazil
10.5.3.1. Market Revenue and Forecast, by Application (2017-2030)
10.5.3.2. Market Revenue and Forecast, by Patient Type (2017-2030)
10.5.4. Rest of LATAM
10.5.4.1. Market Revenue and Forecast, by Application (2017-2030)
10.5.4.2. Market Revenue and Forecast, by Patient Type (2017-2030)
Chapter 11. Company Profiles
11.1. Medtronic
11.1.1. Company Overview
11.1.2. Product Offerings
11.1.3. Financial Performance
11.1.4. Recent Initiatives
11.2. Integra LifeSciences Corporation
11.2.1. Company Overview
11.2.2. Product Offerings
11.2.3. Financial Performance
11.2.4. Recent Initiatives
11.3. Natus Medical Incorporated
11.3.1. Company Overview
11.3.2. Product Offerings
11.3.3. Financial Performance
11.3.4. Recent Initiatives
11.4. Fuji Systems
11.4.1. Company Overview
11.4.2. Product Offerings
11.4.3. Financial Performance
11.4.4. LTE Scientific
11.5. Spiegelberg GmbH
11.5.1. Company Overview
11.5.2. Product Offerings
11.5.3. Financial Performance
11.5.4. Recent Initiatives
11.6. SOPHYSA
11.6.1. Company Overview
11.6.2. Product Offerings
11.6.3. Financial Performance
11.6.4. Recent Initiatives
11.7. Neuromedex
11.7.1. Company Overview
11.7.2. Product Offerings
11.7.3. Financial Performance
11.7.4. Recent Initiatives
Chapter 12. Research Methodology
12.1. Primary Research
12.2. Secondary Research
12.3. Assumptions
Chapter 13. Appendix
13.1. About Us
13.2. Glossary of Terms
 Cross-segment Market Size and Analysis for
Mentioned Segments
Cross-segment Market Size and Analysis for
Mentioned Segments
 Additional Company Profiles (Upto 5 With No Cost)
Additional Company Profiles (Upto 5 With No Cost)
 Additional Countries (Apart From Mentioned Countries)
Additional Countries (Apart From Mentioned Countries)
 Country/Region-specific Report
Country/Region-specific Report
 Go To Market Strategy
Go To Market Strategy
 Region Specific Market Dynamics
Region Specific Market Dynamics Region Level Market Share
Region Level Market Share Import Export Analysis
Import Export Analysis Production Analysis
Production Analysis Others
Others