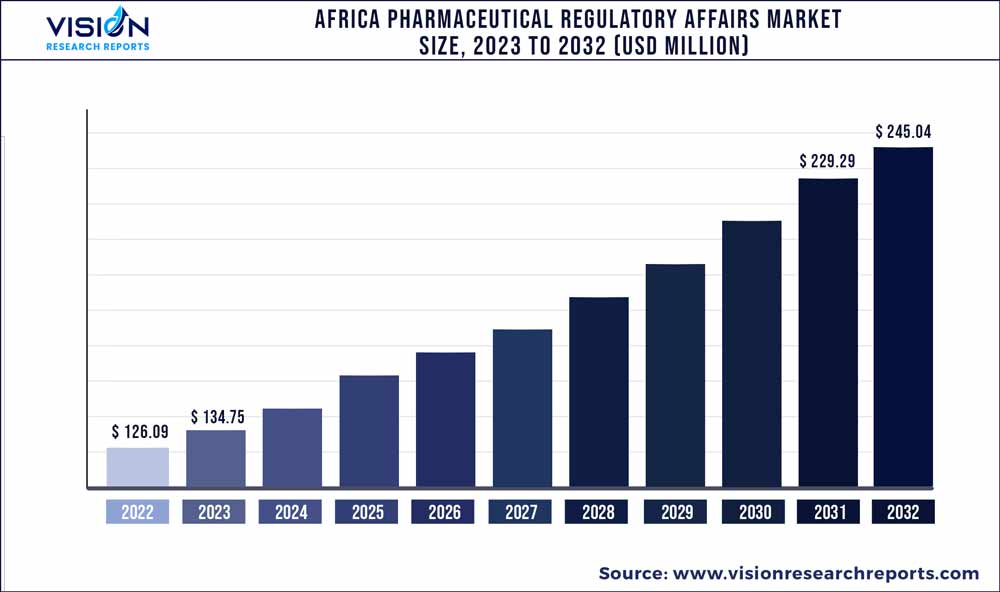
The Africa pharmaceutical regulatory affairs market was estimated at USD 126.09 million in 2022 and it is expected to surpass around USD 245.04 million by 2032, poised to grow at a CAGR of 6.87% from 2023 to 2032.
Key Pointers
Report Scope of the Africa Pharmaceutical Regulatory Affairs Market
| Report Coverage | Details |
| Market Size in 2022 | USD 126.09 million |
| Revenue Forecast by 2032 | USD 245.04 million |
| Growth rate from 2023 to 2032 | CAGR of 6.87% |
| Base Year | 2022 |
| Forecast Period | 2023 to 2032 |
| Market Analysis (Terms Used) | Value (US$ Million/Billion) or (Volume/Units) |
| Companies Covered | Freyr; IQVIA Inc; ICON plc; WuXi AppTec; Charles River Laboratories; Labcorp Drug Development; Parexel International Corporation; Pharmalex GmbH; Pharmexon; Genpact |
The market is driven by several factors such as the increasing prevalence of chronic diseases, a growing elderly population, rising healthcare expenditure, and the demand for innovative and cost-effective medicines across the continent. Additionally, there has been an increasing focus on promoting local pharmaceutical manufacturing in Africa. The government and international organizations have been supporting domestic production to reduce dependence on imports and improve access to affordable medicines. This would further create opportunities for local pharmaceutical companies, leading to a positive impact on the market.
The COVID-19 pandemic of 2020-21 and the macroeconomic headwinds of 2022-23 had a notable impact on the market. The pandemic highlighted the need for robust regulatory systems to expedite the approval and distribution of vaccines, therapeutics, and medical devices. Regulatory agencies in various African countries had to adapt quickly to expedite emergency approvals while ensuring safety and efficacy. These factors have led to potential advancements in the regulatory framework and have brought about collaborative efforts between regulatory authorities and pharmaceutical companies.
Furthermore, there have been notable advancements in the regulatory landscape in Africa. Several countries have been strengthening their regulatory capabilities and implementing reforms to harmonize their processes with international standards. The African Medicines Agency (AMA), launched in 2019, is a notable development aimed at promoting access to safe, effective, and quality medicines across the continent. Such initiatives are expected to streamline the regulatory pathway, encourage innovation, and enhance healthcare outcomes.
The increasing focus on clinical research and development in Africa is also contributing to boosting the demand for a robust regulatory framework. Many countries in the region are participating in clinical trials for various diseases, including infectious diseases, oncology, and tropical illnesses. This shift towards clinical research is attributed to the availability of diverse patient populations, relatively lower costs, and increasing partnerships between global research organizations and African institutions.
Services Insights
By services, the regulatory writing & publishing segment dominated the market with a share of over 37% in 2022. The growth is attributed to factors such as increased outsourcing of these services by large- and mid-size biopharmaceutical and medical device companies. Regulatory writing and publishing services are offered from the early stages of development to the post-marketing authorization phase. Various functions performed under this segment include submission management services such as writing and publishing applications for various regulatory agencies across the globe. Some organizations also provide nonclinical writing and publishing services such as product labeling, aggregate reporting, patient narratives, & investigator brochures.
The legal representation segment is projected to witness the fastest growth rate in the coming years. These services are provided by CROs to biopharmaceutical companies that require regulatory support services for their medicinal products. Different countries require in-house representatives as regulations are very complex and ever-changing. CROs provide legal representatives who can handle regulatory requirements pertaining to the commercialization of their medicinal products. The demand for legal representatives has considerably increased over the years owing to complex and changing healthcare regulations.
Category Insights
By category, the drugs segment dominated the market in 2022 with a market share of 59%. Regulatory affairs play a very important role in the entire drug development, manufacturing, and commercialization continuum. This can be attributed to various regulations and related regulatory submissions/documentation at each of the steps involved in the process. A clear understanding of these regulations enables timely and cost-effective product launches in the market and can also help pharmaceutical companies gain the first-mover advantage.
The biologics segment is projected to witness considerable growth during the forecasted period. Increasing healthcare needs, coupled with increasing investments in healthcare infrastructure and improving regulatory frameworks, are driving the demand for biologics in the region. The region’s increasing middle-class population and expanding access to healthcare services contribute to the growing demand for advanced and innovative therapies.
Indication Insights
By indication, the oncology segment held the highest share of the market in 2022. This can be attributed to the high prevalence of cancer, which is boosting the need for safe and effective treatment options. Africa is expected to experience the most significant increase in cancer cases and deaths compared to other regions. According to data published by GLOBOCAN, the number of cancer cases in Africa is estimated to increase from 1.1 million new cases in 2020 to 2.1 million cases in 2040. Furthermore, oncology is one of the most profitable markets for pharmaceutical & biotechnology companies, thereby increasing the R&D projects undertaken by these players.
The immunology segment is projected to witness considerable growth during the forecasted period. The robust immunology pipeline of pharmaceutical and biopharmaceutical companies is anticipated to further boost segment growth. For instance, as of 2022, Novartis had nearly 33 drugs in the pipeline for treating immunological conditions. The high number of drugs in the pipeline for immunological drugs is expected to support the demand for regulatory services during the forecast period.
Product Stage Insights
By product stage, the clinical studies segment held the highest share of the market in 2022. This can be attributed to the increasing number of clinical trial registrations over the past few years. The COVID-19 pandemic has increased the number of clinical trials in the region, due to the urgency to identify and commercialize an effective cure and/or vaccine for the disease. Furthermore, supportive regulatory actions, such as shortening trial approval time, waiving the waiting period, release of industry guidance documents, and funding clinical trials undertaken by regulatory authorities, are anticipated to boost this segment in the pharmaceutical regulatory affairs market.
The preclinical segment is projected to witness considerable growth during the forecasted period. This can be attributed to the increasing demand for novel disease treatments, such as COVID-19, the Zika virus, & Ebola, and the increasing prevalence of existing diseases such as CVDs, cancer, & neurological diseases. The main goals of preclinical studies are to determine a molecule’s safety profile, bioanalysis, pharmacodynamics, and ADME & DMPK analysis.
Service Provider Insights
By service provider, the outsourcing segment dominated the market in 2022. This can be attributed to the increasing popularity of these services as outsourcing enables healthcare companies to reduce costs, prioritize strategic projects, reduce staff training time, and improve overall efficiency as well as provide greater flexibility. The availability of various outsourcing models suitable for different company sizes is also anticipated to boost the outsourcing market.
The in-house segment is projected to witness slow growth during the forecasted period. The slow growth is due to an increasing number of small- and medium-scale pharma/biotech/medical device companies, which lack funds and infrastructure to support an in-house regulatory affairs department. Furthermore, these firms find it difficult to hire experienced and technically sound full-time employees as they are emerging companies & have budget constraints. Hence, in-house regulatory affairs department is not preferred by small-scale companies, and they outsource nearly 90% of their regulatory solutions.
Company Size Insights
By company size, the medium segment dominated the market in 2022. These companies have a broader product portfolio and a wider geographic presence, allowing them to serve a larger customer base across different African countries. They can invest in R&D, clinical trials, and regulatory compliance, ensuring that their products meet the necessary standards and regulations. Moreover, medium-sized companies can establish strategic partnerships with local stakeholders, including regulatory authorities and healthcare providers, to streamline the regulatory process & enhance market access.
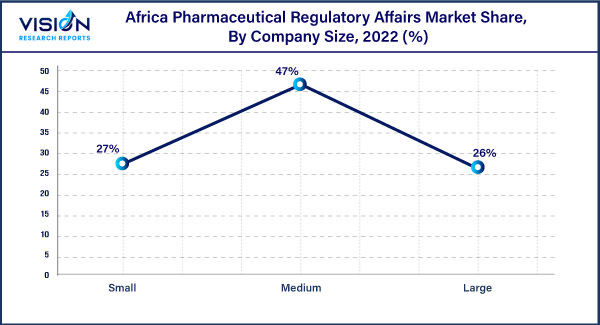
The large-size segment is projected to witness considerable growth during the forecasted period. Large companies have extensive resources, global presence, and expertise in navigating complex regulatory frameworks. These companies often have established relationships with regulatory authorities, enabling them to influence policy and shape regulatory standards. Large companies have the financial strength to invest in extensive clinical trials, advanced manufacturing facilities, and robust regulatory compliance systems. This allows them to bring innovative and high-quality pharmaceutical products to the African market while meeting stringent regulatory requirements.
Africa Pharmaceutical Regulatory Affairs Market Segmentations:
By Services
By Category
By Indication
By Product Stage
By Service Provider
By Company Size
Chapter 1. Introduction
1.1. Research Objective
1.2. Scope of the Study
1.3. Definition
Chapter 2. Research Methodology
2.1. Research Approach
2.2. Data Sources
2.3. Assumptions & Limitations
Chapter 3. Executive Summary
3.1. Market Snapshot
Chapter 4. Market Variables and Scope
4.1. Introduction
4.2. Market Classification and Scope
4.3. Industry Value Chain Analysis
4.3.1. Company Size Procurement Analysis
4.3.2. Sales and Distribution Analysis
4.3.3. Downstream Buyer Analysis
Chapter 5. COVID 19 Impact on Africa Pharmaceutical Regulatory Affairs Market
5.1. COVID-19 Landscape: Africa Pharmaceutical Regulatory Affairs Industry Impact
5.2. COVID 19 - Impact Assessment for the Industry
5.3. COVID 19 Impact: Major Government Policy
5.4. Market Trends and Opportunities in the COVID-19 Landscape
Chapter 6. Market Dynamics Analysis and Trends
6.1. Market Dynamics
6.1.1. Market Drivers
6.1.2. Market Restraints
6.1.3. Market Opportunities
6.2. Porter’s Five Forces Analysis
6.2.1. Bargaining power of suppliers
6.2.2. Bargaining power of buyers
6.2.3. Threat of substitute
6.2.4. Threat of new entrants
6.2.5. Degree of competition
Chapter 7. Competitive Landscape
7.1.1. Company Market Share/Positioning Analysis
7.1.2. Key Strategies Adopted by Players
7.1.3. Vendor Landscape
7.1.3.1. List of Suppliers
7.1.3.2. List of Buyers
Chapter 8. Africa Pharmaceutical Regulatory Affairs Market, By Services
8.1. Africa Pharmaceutical Regulatory Affairs Market, by Services, 2023-2032
8.1.1. Regulatory Consulting
8.1.1.1. Market Revenue and Forecast (2020-2032)
8.1.2. Legal Representation
8.1.2.1. Market Revenue and Forecast (2020-2032)
8.1.3. Regulatory Writing & Publishing
8.1.3.1. Market Revenue and Forecast (2020-2032)
8.1.4. Product Registration & Clinical Trial Applications
8.1.4.1. Market Revenue and Forecast (2020-2032)
8.1.5. Other Services
8.1.5.1. Market Revenue and Forecast (2020-2032)
Chapter 9. Africa Pharmaceutical Regulatory Affairs Market, By Category
9.1. Africa Pharmaceutical Regulatory Affairs Market, by Category, 2023-2032
9.1.1. Drugs
9.1.1.1. Market Revenue and Forecast (2020-2032)
9.1.2. Biologics
9.1.2.1. Market Revenue and Forecast (2020-2032)
9.1.3. ATMP
9.1.3.1. Market Revenue and Forecast (2020-2032)
9.1.4. Biosimilars
9.1.4.1. Market Revenue and Forecast (2020-2032)
Chapter 10. Africa Pharmaceutical Regulatory Affairs Market, By Indication
10.1. Africa Pharmaceutical Regulatory Affairs Market, by Indication, 2023-2032
10.1.1. Oncology
10.1.1.1. Market Revenue and Forecast (2020-2032)
10.1.2. Neurology
10.1.2.1. Market Revenue and Forecast (2020-2032)
10.1.3. Cardiology
10.1.3.1. Market Revenue and Forecast (2020-2032)
10.1.4. Immunology
10.1.4.1. Market Revenue and Forecast (2020-2032)
10.1.5. Others
10.1.5.1. Market Revenue and Forecast (2020-2032)
Chapter 11. Africa Pharmaceutical Regulatory Affairs Market, By Product Stage
11.1. Africa Pharmaceutical Regulatory Affairs Market, by Product Stage, 2023-2032
11.1.1. Preclinical
11.1.1.1. Market Revenue and Forecast (2020-2032)
11.1.2. Clinical studies
11.1.2.1. Market Revenue and Forecast (2020-2032)
11.1.3. PMA
11.1.3.1. Market Revenue and Forecast (2020-2032)
Chapter 12. Africa Pharmaceutical Regulatory Affairs Market, By Service Provider
12.1. Africa Pharmaceutical Regulatory Affairs Market, by Service Provider, 2023-2032
12.1.1. In-house
12.1.1.1. Market Revenue and Forecast (2020-2032)
12.1.2. Outsourcing
12.1.2.1. Market Revenue and Forecast (2020-2032)
Chapter 13. Africa Pharmaceutical Regulatory Affairs Market, By Company Size
13.1. Africa Pharmaceutical Regulatory Affairs Market, by Company Size, 2023-2032
13.1.1. Small
13.1.1.1. Market Revenue and Forecast (2020-2032)
13.1.2. Medium
13.1.2.1. Market Revenue and Forecast (2020-2032)
13.1.3. Large
13.1.3.1. Market Revenue and Forecast (2020-2032)
Chapter 14. Africa Pharmaceutical Regulatory Affairs Market, Regional Estimates and Trend Forecast
14.1. Africa
14.1.1. Market Revenue and Forecast, by Services (2020-2032)
14.1.2. Market Revenue and Forecast, by Category (2020-2032)
14.1.3. Market Revenue and Forecast, by Indication (2020-2032)
14.1.4. Market Revenue and Forecast, by Product Stage (2020-2032)
14.1.5. Market Revenue and Forecast, by Service Provider (2020-2032)
14.1.6. Market Revenue and Forecast, by Company Size (2020-2032)
Chapter 15. Company Profiles
15.1. Freyr
15.1.1. Company Overview
15.1.2. Product Offerings
15.1.3. Financial Performance
15.1.4. Recent Initiatives
15.2. IQVIA Inc
15.2.1. Company Overview
15.2.2. Product Offerings
15.2.3. Financial Performance
15.2.4. Recent Initiatives
15.3. ICON plc
15.3.1. Company Overview
15.3.2. Product Offerings
15.3.3. Financial Performance
15.3.4. Recent Initiatives
15.4. WuXi AppTec
15.4.1. Company Overview
15.4.2. Product Offerings
15.4.3. Financial Performance
15.4.4. Recent Initiatives
15.5. Charles River Laboratories
15.5.1. Company Overview
15.5.2. Product Offerings
15.5.3. Financial Performance
15.5.4. Recent Initiatives
15.6. Labcorp Drug Development
15.6.1. Company Overview
15.6.2. Product Offerings
15.6.3. Financial Performance
15.6.4. Recent Initiatives
15.7. Parexel International Corporation
15.7.1. Company Overview
15.7.2. Product Offerings
15.7.3. Financial Performance
15.7.4. Recent Initiatives
15.8. Pharmalex GmbH
15.8.1. Company Overview
15.8.2. Product Offerings
15.8.3. Financial Performance
15.8.4. Recent Initiatives
15.9. Pharmexon
15.9.1. Company Overview
15.9.2. Product Offerings
15.9.3. Financial Performance
15.9.4. Recent Initiatives
15.10. Genpact
15.10.1. Company Overview
15.10.2. Product Offerings
15.10.3. Financial Performance
15.10.4. Recent Initiatives
Chapter 16. Research Methodology
16.1. Primary Research
16.2. Secondary Research
16.3. Assumptions
Chapter 17. Appendix
17.1. About Us
17.2. Glossary of Terms
 Cross-segment Market Size and Analysis for
Mentioned Segments
Cross-segment Market Size and Analysis for
Mentioned Segments
 Additional Company Profiles (Upto 5 With No Cost)
Additional Company Profiles (Upto 5 With No Cost)
 Additional Countries (Apart From Mentioned Countries)
Additional Countries (Apart From Mentioned Countries)
 Country/Region-specific Report
Country/Region-specific Report
 Go To Market Strategy
Go To Market Strategy
 Region Specific Market Dynamics
Region Specific Market Dynamics Region Level Market Share
Region Level Market Share Import Export Analysis
Import Export Analysis Production Analysis
Production Analysis Others
Others