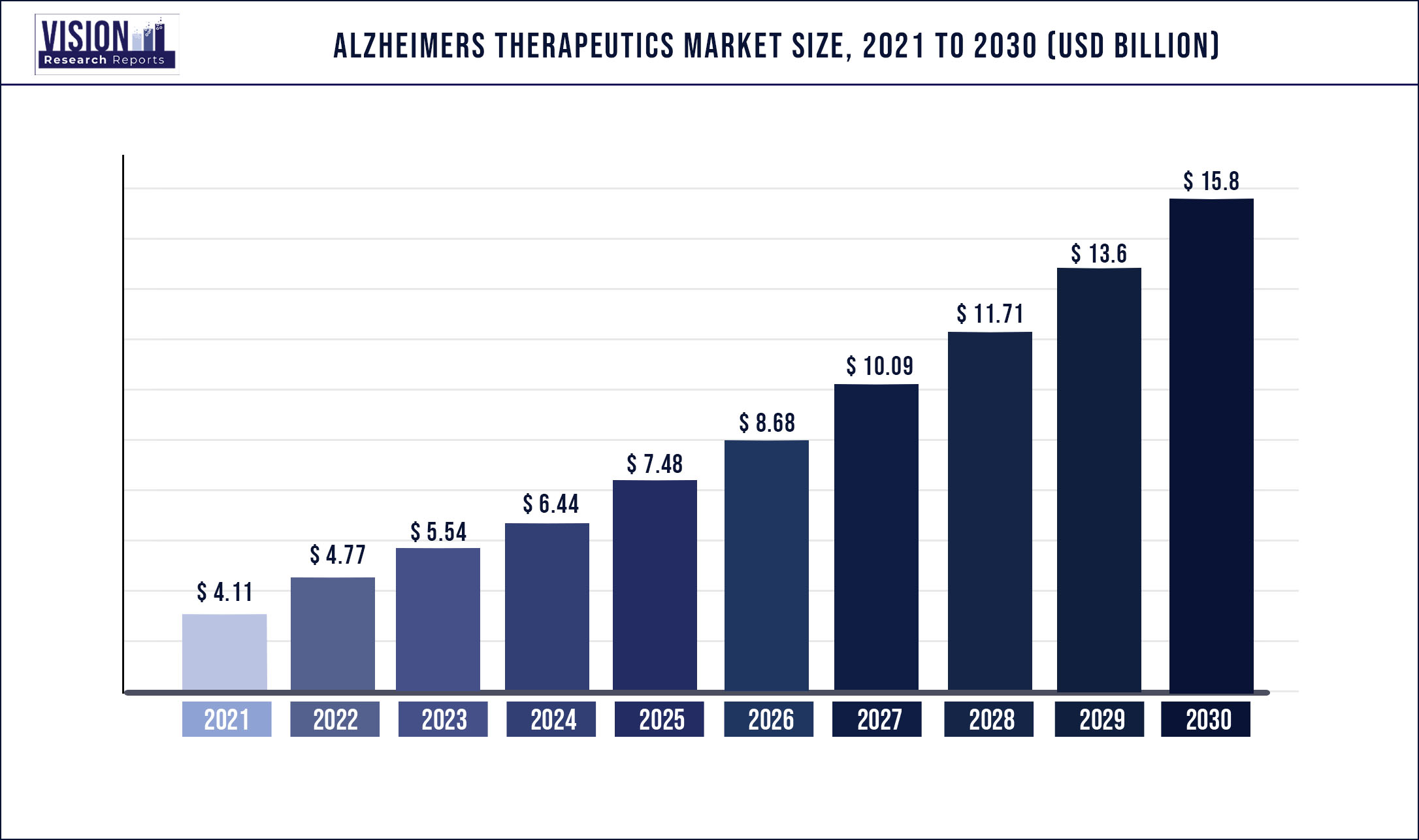
The global Alzheimer's therapeutics market size was estimated at around USD 4.11 billion in 2021 and it is projected to hit around USD 15.8 billion by 2030, growing at a CAGR of 16.14% from 2022 to 2030
Report Highlights
Government organizations are engaged in providing funds for Alzheimer's support, care, and research activities, which is projected to fuel the market growth. For instance, in March 2022, the Alzheimer's Society announced to spend around USD 3.5 billion in the coming years on dementia research to improve the care of patients.
Additionally, the Alzheimer's Association with the Alzheimer's Impact Movement (AIM) announced to utilize USD 25 million to implement the BOLD Infrastructure for Alzheimer's Act to create an effective public health substructure for patients. In addition, in September 2021, the South Texas Alzheimer's Disease Center and the Duke/University of North Carolina Alzheimer's Disease Research Center were granted USD 14.8 million over five years by the National Institute of Aging to encourage research on risk factors related to AD.
Furthermore, an increase in the incidence and cost burden of dementia associated with AD is expected to boost the market growth. According to the Alzheimer's Society, around 900,000 people are living with dementia in the United Kingdom, and it is projected to rise to 1.6 million by 2040 and the incidence of dementia will be 209,600 in the same year.
The current standard of care is limited to symptom-relieving drugs. Therefore, the approval of disease-modifying anti-amyloid therapeutics is expected to change the treatment landscape. For instance, in June 2021, the U.S. FDA granted accelerated approval to Aduhelm (aducanumab) for the treatment of patients with Alzheimer’s disease. It is a monoclonal antibody that targets cluster forms of amyloid beta present in the brains of patients and stops its formation. Other key anti-amyloid monoclonal antibodies in late-stage clinical trials include Gantenerumab (F. Hoffmann-La Roche Ltd.), Solanezumab (Eli Lilly and Company), and Lecanemab (Eisai Inc./Biogen), among others.
Scope of The Report
| Report Coverage | Details |
| Market Size in 2021 | USD 4.11 billion |
| Revenue Forecast by 2030 | USD 15.8 billion |
| Growth rate from 2022 to 2030 | CAGR of 16.14% |
| Base Year | 2021 |
| Forecast Period | 2022 to 2030 |
| Segmentation | Product, end-user, region |
| Companies Covered | Eisai Co., Ltd.; Novartis AG; AbbVie Inc. (Allergan Plc.); Adamas Pharmaceuticals, Inc.; H. Lundbeck A/S; Biogen; AC Immune; F. Hoffmann La Roche Ltd.; Daiichi Sankyo Company, Limited; Johnson & Johnson Services, Inc.; TauRx Pharmaceuticals Ltd. |
Product Insights
The cholinesterase inhibitors segment held the largest revenue share of over 50.2% in 2021 as it is the standard of care for Alzheimer’s treatment. In March 2022, the U.S. FDA approved the cholinesterase inhibitor Adlarity (donepezil hydrochloride) developed by Corium Inc., for the treatment of patients with severe dementia of Alzheimer’s disease (AD). It is a transdermal patch administered once a week and delivers a stable dose of donepezil and was the fourth cholinesterase drug approved and launched in the U.S. Based on product, the market is divided into cholinesterase inhibitors, NMDA receptor antagonists, combination drugs, and pipeline drugs.
The pipeline drugs segment is expected to witness the fastest growth over the forecast period owing to the impending launch of multiple disease-modifying therapies. Companies such as F. Hoffmann La Roche Ltd.; Biogen/Eisai Inc.; and Eli Lilly and Company have drug candidates under development that will modify the treatment landscape of AD. According to Alzheimer’s Association, there were 143 drug candidates in 172 clinical trials for AD. Of these, 31 drug candidates in phase 3 clinical trial, 82 drug candidates in phase 2, and 30 drug candidates in phase 1. Successful completion of trials and subsequent approval of these products are anticipated to boost the market growth.
End-user Insights
The hospital pharmacy segment dominated the market in 2021 with a revenue share of over 55.04% owing to the increasing hospitalization due to a rise in the geriatric population and Alzheimer’s patients. According to Alzheimer’s Association report 2022, there are 518 hospitalizations per 1,000 Medicare beneficiaries in the geriatric population having Alzheimer’s or other dementias as compared to only 234 hospitalizations per 1,000 Medicare beneficiaries without these conditions. However, the total number of hospitalizations of AD patients is expected to remain low compared to the prevalence of Alzheimer’s disease.
The e-commerce segment is expected to expand at a CAGR of 15.33% over the forecast period. An increase in the usage of the internet and smartphone, ease of ordering medications through an e-commerce platform, and increasing e-commerce services globally are expected to fuel the segment growth in the coming years.
Regional Insights
North America dominated the market with a share of over 40.23% in 2021 and is anticipated to grow at a significant rate over the forecast period. The growing research for the treatment of Alzheimer’s is propelling the market growth. In June 2021, Eisai Co., Ltd. and Biogen's product lecanemab (BAN2401), an anti-amyloid beta protofibril antibody for treating Alzheimer's disease, was granted the Breakthrough Therapy designation by the U.S. FDA. Moreover, the increasing government initiatives and funding are expected to boost the growth of the market.
The Asia Pacific region is expected to emerge as the fastest-growing region over the forecast period owing to increasing awareness among people and the increasing research activities to develop novel drugs to treat AD. In 2022, there are around 487,500 Australians living with dementia and this number is expected to increase to 1.1 million by 2058. Dementia is the second leading cause of death in Australia.
Key Players
Market Segmentation
Chapter 1. Introduction
1.1. Research Objective
1.2. Scope of the Study
1.3. Definition
Chapter 2. Research Methodology
2.1. Research Approach
2.2. Data Sources
2.3. Assumptions & Limitations
Chapter 3. Executive Summary
3.1. Market Snapshot
Chapter 4. Market Variables and Scope
4.1. Introduction
4.2. Market Classification and Scope
4.3. Industry Value Chain Analysis
4.3.1. Raw Material Procurement Analysis
4.3.2. Sales and Distribution Channel Analysis
4.3.3. Downstream Buyer Analysis
Chapter 5. COVID 19 Impact on Alzheimer’s Therapeutics Market
5.1. COVID-19 Landscape: Alzheimer’s Therapeutics Industry Impact
5.2. COVID 19 - Impact Assessment for the Industry
5.3. COVID 19 Impact: Global Major Government Policy
5.4. Market Trends and Opportunities in the COVID-19 Landscape
Chapter 6. Market Dynamics Analysis and Trends
6.1. Market Dynamics
6.1.1. Market Drivers
6.1.2. Market Restraints
6.1.3. Market Opportunities
6.2. Porter’s Five Forces Analysis
6.2.1. Bargaining power of suppliers
6.2.2. Bargaining power of buyers
6.2.3. Threat of substitute
6.2.4. Threat of new entrants
6.2.5. Degree of competition
Chapter 7. Competitive Landscape
7.1.1. Company Market Share/Positioning Analysis
7.1.2. Key Strategies Adopted by Players
7.1.3. Vendor Landscape
7.1.3.1. List of Suppliers
7.1.3.2. List of Buyers
Chapter 8. Global Alzheimer’s Therapeutics Market, By Product
8.1. Alzheimer’s Therapeutics Market, by Product, 2022-2030
8.1.1. Cholinesterase Inhibitors
8.1.1.1. Market Revenue and Forecast (2017-2030)
8.1.2. NMDA Receptor Antagonist
8.1.2.1. Market Revenue and Forecast (2017-2030)
8.1.3. Combination Drug
8.1.3.1. Market Revenue and Forecast (2017-2030)
8.1.4. Pipeline Drugs
8.1.4.1. Market Revenue and Forecast (2017-2030)
Chapter 9. Global Alzheimer’s Therapeutics Market, By End-user
9.1. Alzheimer’s Therapeutics Market, by End-user, 2022-2030
9.1.1. Hospital Pharmacy
9.1.1.1. Market Revenue and Forecast (2017-2030)
9.1.2. Retail Pharmacy
9.1.2.1. Market Revenue and Forecast (2017-2030)
9.1.3. E-commerce
9.1.3.1. Market Revenue and Forecast (2017-2030)
Chapter 10. Global Alzheimer’s Therapeutics Market, Regional Estimates and Trend Forecast
10.1. North America
10.1.1. Market Revenue and Forecast, by Product (2017-2030)
10.1.2. Market Revenue and Forecast, by End-user (2017-2030)
10.1.3. U.S.
10.1.3.1. Market Revenue and Forecast, by Product (2017-2030)
10.1.3.2. Market Revenue and Forecast, by End-user (2017-2030)
10.1.4. Rest of North America
10.1.4.1. Market Revenue and Forecast, by Product (2017-2030)
10.1.4.2. Market Revenue and Forecast, by End-user (2017-2030)
10.2. Europe
10.2.1. Market Revenue and Forecast, by Product (2017-2030)
10.2.2. Market Revenue and Forecast, by End-user (2017-2030)
10.2.3. UK
10.2.3.1. Market Revenue and Forecast, by Product (2017-2030)
10.2.3.2. Market Revenue and Forecast, by End-user (2017-2030)
10.2.4. Germany
10.2.4.1. Market Revenue and Forecast, by Product (2017-2030)
10.2.4.2. Market Revenue and Forecast, by End-user (2017-2030)
10.2.5. France
10.2.5.1. Market Revenue and Forecast, by Product (2017-2030)
10.2.5.2. Market Revenue and Forecast, by End-user (2017-2030)
10.2.6. Rest of Europe
10.2.6.1. Market Revenue and Forecast, by Product (2017-2030)
10.2.6.2. Market Revenue and Forecast, by End-user (2017-2030)
10.3. APAC
10.3.1. Market Revenue and Forecast, by Product (2017-2030)
10.3.2. Market Revenue and Forecast, by End-user (2017-2030)
10.3.3. India
10.3.3.1. Market Revenue and Forecast, by Product (2017-2030)
10.3.3.2. Market Revenue and Forecast, by End-user (2017-2030)
10.3.4. China
10.3.4.1. Market Revenue and Forecast, by Product (2017-2030)
10.3.4.2. Market Revenue and Forecast, by End-user (2017-2030)
10.3.5. Japan
10.3.5.1. Market Revenue and Forecast, by Product (2017-2030)
10.3.5.2. Market Revenue and Forecast, by End-user (2017-2030)
10.3.6. Rest of APAC
10.3.6.1. Market Revenue and Forecast, by Product (2017-2030)
10.3.6.2. Market Revenue and Forecast, by End-user (2017-2030)
10.4. MEA
10.4.1. Market Revenue and Forecast, by Product (2017-2030)
10.4.2. Market Revenue and Forecast, by End-user (2017-2030)
10.4.3. GCC
10.4.3.1. Market Revenue and Forecast, by Product (2017-2030)
10.4.3.2. Market Revenue and Forecast, by End-user (2017-2030)
10.4.4. North Africa
10.4.4.1. Market Revenue and Forecast, by Product (2017-2030)
10.4.4.2. Market Revenue and Forecast, by End-user (2017-2030)
10.4.5. South Africa
10.4.5.1. Market Revenue and Forecast, by Product (2017-2030)
10.4.5.2. Market Revenue and Forecast, by End-user (2017-2030)
10.4.6. Rest of MEA
10.4.6.1. Market Revenue and Forecast, by Product (2017-2030)
10.4.6.2. Market Revenue and Forecast, by End-user (2017-2030)
10.5. Latin America
10.5.1. Market Revenue and Forecast, by Product (2017-2030)
10.5.2. Market Revenue and Forecast, by End-user (2017-2030)
10.5.3. Brazil
10.5.3.1. Market Revenue and Forecast, by Product (2017-2030)
10.5.3.2. Market Revenue and Forecast, by End-user (2017-2030)
10.5.4. Rest of LATAM
10.5.4.1. Market Revenue and Forecast, by Product (2017-2030)
10.5.4.2. Market Revenue and Forecast, by End-user (2017-2030)
Chapter 11. Company Profiles
11.1. Eisai Co., Ltd.
11.1.1. Company Overview
11.1.2. Product Offerings
11.1.3. Financial Performance
11.1.4. Recent Initiatives
11.2. Novartis AG
11.2.1. Company Overview
11.2.2. Product Offerings
11.2.3. Financial Performance
11.2.4. Recent Initiatives
11.3. AbbVie Inc. (Allergan Plc.)
11.3.1. Company Overview
11.3.2. Product Offerings
11.3.3. Financial Performance
11.3.4. Recent Initiatives
11.4. Adamas Pharmaceuticals, Inc.
11.4.1. Company Overview
11.4.2. Product Offerings
11.4.3. Financial Performance
11.4.4. LTE Scientific
11.5. H. Lundbeck A/S
11.5.1. Company Overview
11.5.2. Product Offerings
11.5.3. Financial Performance
11.5.4. Recent Initiatives
11.6. Biogen
11.6.1. Company Overview
11.6.2. Product Offerings
11.6.3. Financial Performance
11.6.4. Recent Initiatives
11.7. AC Immune
11.7.1. Company Overview
11.7.2. Product Offerings
11.7.3. Financial Performance
11.7.4. Recent Initiatives
11.8. F. Hoffmann La Roche Ltd.
11.8.1. Company Overview
11.8.2. Product Offerings
11.8.3. Financial Performance
11.8.4. Recent Initiatives
11.9. Daiichi Sankyo Company, Limited
11.9.1. Company Overview
11.9.2. Product Offerings
11.9.3. Financial Performance
11.9.4. Recent Initiatives
11.10. Johnson & Johnson Services, Inc.
11.10.1. Company Overview
11.10.2. Product Offerings
11.10.3. Financial Performance
11.10.4. Recent Initiatives
Chapter 12. Research Methodology
12.1. Primary Research
12.2. Secondary Research
12.3. Assumptions
Chapter 13. Appendix
13.1. About Us
13.2. Glossary of Terms
 Cross-segment Market Size and Analysis for
Mentioned Segments
Cross-segment Market Size and Analysis for
Mentioned Segments
 Additional Company Profiles (Upto 5 With No Cost)
Additional Company Profiles (Upto 5 With No Cost)
 Additional Countries (Apart From Mentioned Countries)
Additional Countries (Apart From Mentioned Countries)
 Country/Region-specific Report
Country/Region-specific Report
 Go To Market Strategy
Go To Market Strategy
 Region Specific Market Dynamics
Region Specific Market Dynamics Region Level Market Share
Region Level Market Share Import Export Analysis
Import Export Analysis Production Analysis
Production Analysis Others
Others