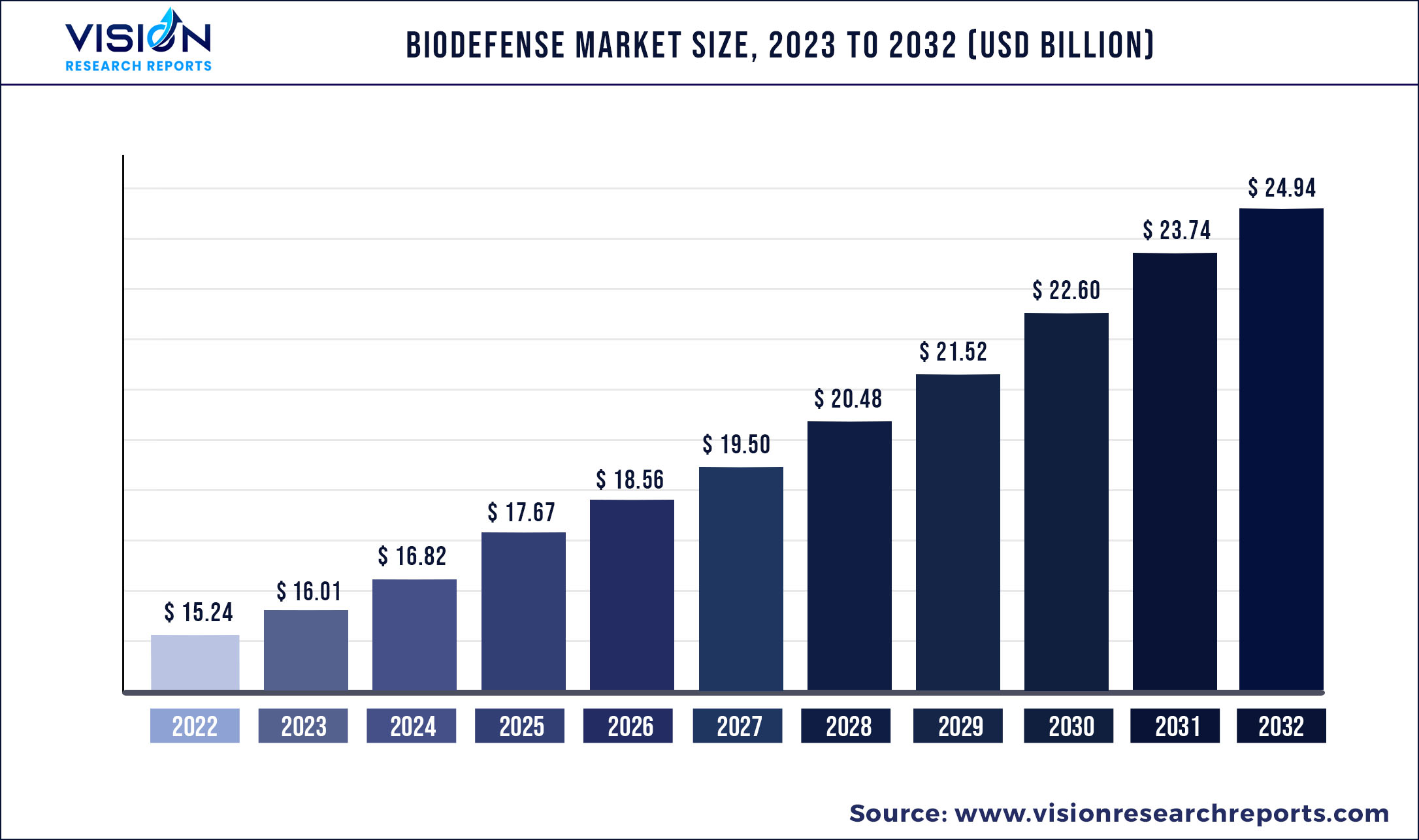
The global biodefense market was estimated at USD 15.24 billion in 2022 and it is expected to surpass around USD 24.94 billion by 2032, poised to grow at a CAGR of 5.05% from 2023 to 2032.
Key Pointers
Biodefense is a set of medical or military measures. These are taken in response to the demand for restoring the biosecurity of a country against biological toxins or infectious agents. The agents can be used with the intent to kill or infect humans, animals, or the environment and to instigate biological warfare. Biological agents used during bioterrorism are usually living organisms. These can include bacteria, viruses, fungi, or toxins that are deliberately used to sicken and kill creating social and economic turmoil. Factors such as the presence of favorable government initiatives in the U.S., growing investment from private players, increased government focus and funding towards biodefense strategies, and the growing prevalence of various agents such as the Ebola virus, Zika virus, and flu are driving the overall market growth.
The advent of technological advancements in the field of genetic engineering and biotechnology in the last decades has offered ease in modification of these fatal, and naturally occurring viruses which can be re-engineered to cause devastation. Moreover, these organisms can be easily obtained which makes biodefense a crucial aspect for countries around the world. Biological agents that have been used as a carrier for bioterrorism in the past include anthrax, botulism, and chemical & nuclear agent. This led to serious economic loss.
These instances drove governments around the world to engage in biodefense acts, treaties, and policies to counter biological threats, reduce risks, and prepare for, respond to, and recover from bioterrorism incidents. Since the anthrax bio-terrorism act that was carried out in 2001 through the mail, the National Institute of Infectious disease has played a vital role in developing medical products and strategies to counter bioterrorism and emerging and re-emerging infectious diseases by carrying out continuous research to diagnose, treat, and prevent them, whether deliberate or naturally occurring.
Governments of many countries remain committed to collaboration with several international partners to support response activities and increase preparedness. For instance, an experimental single-dose Ebola vaccine manufactured by Merck [rVSV-ZEBOV-GP] has been authorized for safe use by the World Health Organization and DRC MoH. As of November 17, 2019, approximately 250,000 persons at risk for Ebola have been vaccinated.
Developed economies are sending aid to countries with weak medical infra and low research capabilities many companies like Bavarian Nordic and Ology Bioservices are researching Ebola and Zika virus. The introduction of the Project BioShield Act was a strategic decision to increase funding for procuring, developing, and utilizing medical countermeasures against biological, chemical, radiological, and nuclear (CBRN) warfare agents. These above-mentioned factors and the rising risk of prevalence of the infectious disease is expected to boost the market growth.
Biodefense Market Segmentations and Key Players:
| By Product | Key Players | Regions Segmentations |
|
Anthrax Smallpox Botulism Radiation/Nuclear Others |
XOMA corporation Altimmune Inc. Emergent Biosolutions Inc. Dynavax Technologies Corporation. SIGA Technologies. Elusys Therapeutics Inc. Ichor Medical Systems. Dynport Vaccine Company. Cleveland Biolabs. Bavarian Nordic. Ology Bioservices. Alnylam Pharmaceuticals Inc |
North America Europe Asia Pacific Latin America Middle East & Africa |
Chapter 1. Introduction
1.1.Research Objective
1.2.Scope of the Study
1.3.Definition
Chapter 2. Research Methodology
2.1.Research Approach
2.2.Data Sources
2.3.Assumptions & Limitations
Chapter 3. Executive Summary
3.1.Market Snapshot
Chapter 4. Market Variables and Scope
4.1.Introduction
4.2.Market Classification and Scope
4.3.Industry Value Chain Analysis
4.3.1. Raw Material Procurement Analysis
4.3.2. Sales and Distribution Channel Analysis
4.3.3. Downstream Buyer Analysis
Chapter 5.COVID 19 Impact on Biodefense Market
5.1. COVID-19 Landscape: Biodefense Industry Impact
5.2. COVID 19 - Impact Assessment for the Industry
5.3. COVID 19 Impact: Major Government Policy
5.4.Market Trends and Opportunities in the COVID-19 Landscape
Chapter 6. Market Dynamics Analysis and Trends
6.1.Market Dynamics
6.1.1. Market Drivers
6.1.2. Market Restraints
6.1.3. Market Opportunities
6.2.Porter’s Five Forces Analysis
6.2.1. Bargaining power of suppliers
6.2.2. Bargaining power of buyers
6.2.3. Threat of substitute
6.2.4. Threat of new entrants
6.2.5. Degree of competition
Chapter 7. Competitive Landscape
7.1.1. Company Market Share/Positioning Analysis
7.1.2. Key Strategies Adopted by Players
7.1.3. Vendor Landscape
7.1.3.1.List of Suppliers
7.1.3.2.List of Buyers
Chapter 8. Biodefense Market, By Product
8.1.Biodefense Market, by Product Type, 2023-2032
8.1.1. Anthrax
8.1.1.1.Market Revenue and Forecast (2019-2032)
8.1.2. Smallpox
8.1.2.1.Market Revenue and Forecast (2019-2032)
8.1.3. Botulism
8.1.3.1.Market Revenue and Forecast (2019-2032)
8.1.4. Radiation/Nuclear
8.1.4.1.Market Revenue and Forecast (2019-2032)
8.1.5. Others
8.1.5.1.Market Revenue and Forecast (2019-2032)
Chapter 9. Biodefense Market, Regional Estimates and Trend Forecast
9.1. North America
9.1.1. Market Revenue and Forecast, by Product (2019-2032)
9.1.2. U.S.
9.1.2.1. Market Revenue and Forecast, by Product (2019-2032)
9.1.3. Rest of North America
9.1.3.1. Market Revenue and Forecast, by Product (2019-2032)
9.2. Europe
9.2.1. Market Revenue and Forecast, by Product (2019-2032)
9.2.2. UK
9.2.2.1. Market Revenue and Forecast, by Product (2019-2032)
9.2.3. Germany
9.2.3.1. Market Revenue and Forecast, by Product (2019-2032)
9.2.4. France
9.2.4.1. Market Revenue and Forecast, by Product (2019-2032)
9.2.5. Rest of Europe
9.2.5.1. Market Revenue and Forecast, by Product (2019-2032)
9.3. APAC
9.3.1. Market Revenue and Forecast, by Product (2019-2032)
9.3.2. India
9.3.2.1. Market Revenue and Forecast, by Product (2019-2032)
9.3.3. China
9.3.3.1. Market Revenue and Forecast, by Product (2019-2032)
9.3.4. Japan
9.3.4.1. Market Revenue and Forecast, by Product (2019-2032)
9.3.5. Rest of APAC
9.3.5.1. Market Revenue and Forecast, by Product (2019-2032)
9.4. MEA
9.4.1. Market Revenue and Forecast, by Product (2019-2032)
9.4.2. GCC
9.4.2.1. Market Revenue and Forecast, by Product (2019-2032)
9.4.3. North Africa
9.4.3.1. Market Revenue and Forecast, by Product (2019-2032)
9.4.4. South Africa
9.4.4.1. Market Revenue and Forecast, by Product (2019-2032)
9.4.5. Rest of MEA
9.4.5.1. Market Revenue and Forecast, by Product (2019-2032)
9.5. Latin America
9.5.1. Market Revenue and Forecast, by Product (2019-2032)
9.5.2. Brazil
9.5.2.1. Market Revenue and Forecast, by Product (2019-2032)
9.5.3. Rest of LATAM
9.5.3.1. Market Revenue and Forecast, by Product (2019-2032)
Chapter 10.Company Profiles
10.1. XOMA corporation
10.1.1.Company Overview
10.1.2.Product Offerings
10.1.3.Financial Performance
10.1.4.Recent Initiatives
10.2. Altimmune Inc.
10.2.1.Company Overview
10.2.2.Product Offerings
10.2.3.Financial Performance
10.2.4.Recent Initiatives
10.3. Emergent Biosolutions Inc.
10.3.1.Company Overview
10.3.2.Product Offerings
10.3.3.Financial Performance
10.3.4.Recent Initiatives
10.4. Dynavax Technologies Corporation.
10.4.1.Company Overview
10.4.2.Product Offerings
10.4.3.Financial Performance
10.4.4.Recent Initiatives
10.5. SIGA Technologies.
10.5.1.Company Overview
10.5.2.Product Offerings
10.5.3.Financial Performance
10.5.4.Recent Initiatives
10.6. Elusys Therapeutics Inc.
10.6.1.Company Overview
10.6.2.Product Offerings
10.6.3.Financial Performance
10.6.4.Recent Initiatives
10.7. Ichor Medical Systems.
10.7.1.Company Overview
10.7.2.Product Offerings
10.7.3.Financial Performance
10.7.4.Recent Initiatives
10.8. Dynport Vaccine Company.
10.8.1.Company Overview
10.8.2.Product Offerings
10.8.3.Financial Performance
10.8.4.Recent Initiatives
10.9. Cleveland Biolabs.
10.9.1.Company Overview
10.9.2.Product Offerings
10.9.3.Financial Performance
10.9.4.Recent Initiatives
10.10. Bavarian Nordic.
10.10.1. Company Overview
10.10.2. Product Offerings
10.10.3. Financial Performance
10.10.4. Recent Initiatives
Chapter 11.Research Methodology
11.1.Primary Research
11.2.Secondary Research
11.3.Assumptions
Chapter 12.Appendix
12.1. About Us
12.2. Glossary of Terms
 Cross-segment Market Size and Analysis for
Mentioned Segments
Cross-segment Market Size and Analysis for
Mentioned Segments
 Additional Company Profiles (Upto 5 With No Cost)
Additional Company Profiles (Upto 5 With No Cost)
 Additional Countries (Apart From Mentioned Countries)
Additional Countries (Apart From Mentioned Countries)
 Country/Region-specific Report
Country/Region-specific Report
 Go To Market Strategy
Go To Market Strategy
 Region Specific Market Dynamics
Region Specific Market Dynamics Region Level Market Share
Region Level Market Share Import Export Analysis
Import Export Analysis Production Analysis
Production Analysis Others
Others