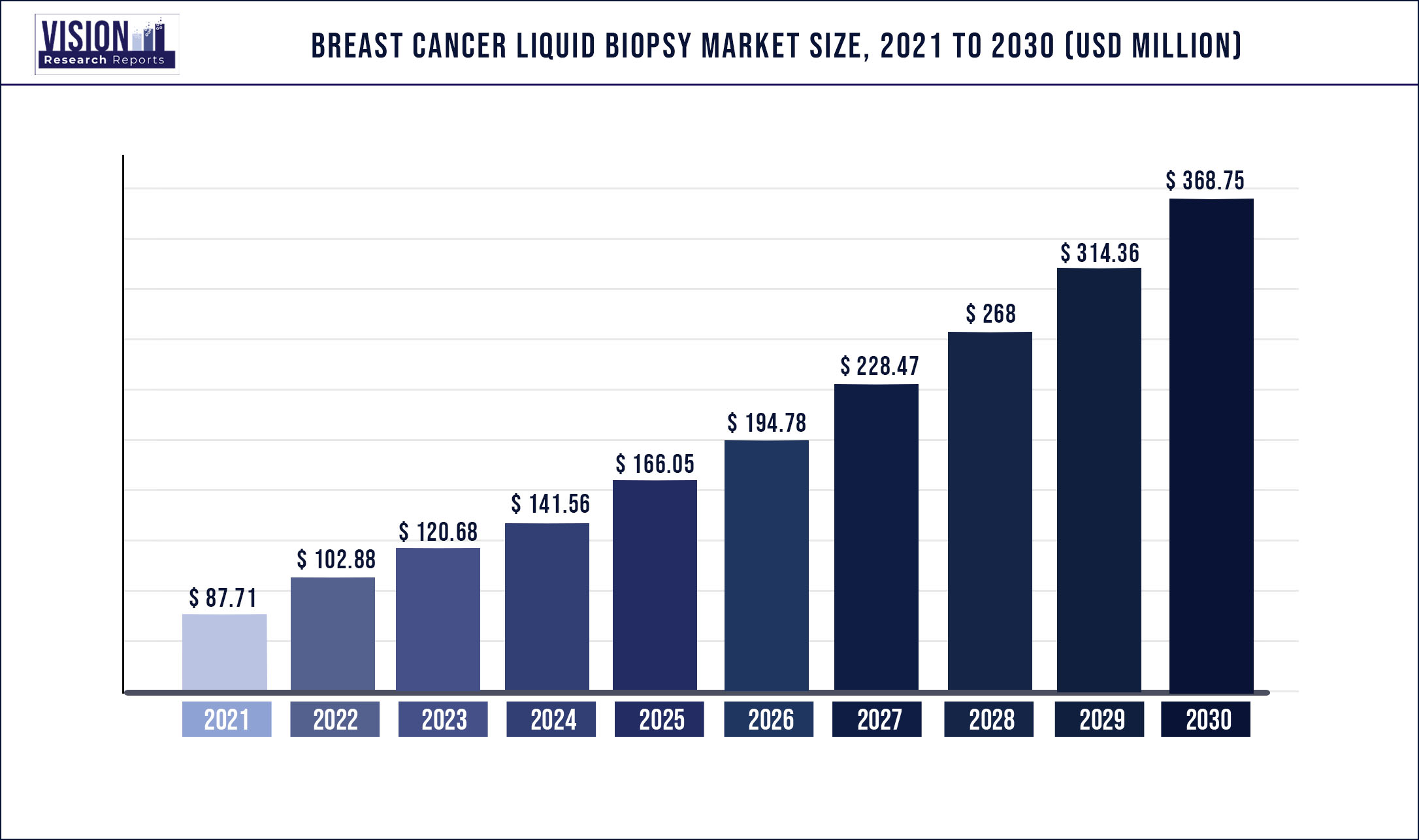
The global breast cancer liquid biopsy market was estimated at USD 87.71 million in 2021 and it is expected to surpass around USD 368.75 million by 2030, poised to grow at a CAGR of 17.3% from 2022 to 2030.
Breast cancer liquid biopsy market is growing as a result of the increasing prevalence of breast cancer among the population, growing awareness about early diagnosis, and treatment along with a rising preference for non-invasive procedures. For instance, according to WHO, in 2020, 2.3 million women were diagnosed with breast cancer which resulted in the death of 685,000 women globally. Moreover, liquid biopsy are been proposed as a novel method for early detection of breast cancer which further increases its adoption further propelling the growth.
Technological advancements in ongoing research on circulating biomarkers have led to the development of a lesser invasive method of detecting non-hematological cancers. Liquid biopsy is considered viable for real-time monitoring of patients in comparison with conventional tumor biopsy. Owing to continuous advancements in related areas, awareness about the clinical importance of liquid biopsy is growing. To make these technologies globally available, further interventional clinical trials are needed, along with the development of an algorithm to combine proper circulating biomarkers. Several companies are undertaking initiatives to boost the global adoption of biopsies which is contributing to growth. As per John Hopkins Medicine in June 2022, liquid biopsy can be used to detect advanced breast cancer biomarkers in 5 hours, commercialization of such tests in the upcoming years is projected to make the diagnosis of the condition easier.
North America dominated the overall market in 2021 which can be attributed to high disease prevalence, rapid technological advancements, and growing government initiatives for cancer diagnosis. The U.S. is the biggest market for liquid biopsy tests owing to a high prevalence rate of the condition in the country, there is significant usage of oncology diagnostic tests. Moreover, rapid technological advancements and recent FDA approvals for liquid biopsy tests coupled with intense competition between biotechnology companies are among the factors expected to boost market growth over the forecast period. Furthermore, major players operating in the region are focusing on collaboration and expansion to meet the escalating demand. For instance, in August 2022, Guardant and Merck KGaA collaborated to expand their precision therapeutics and focus on hard-to-treat cancer.
Scope of The Report
| Report Coverage | Details |
| Market Size in 2021 | USD 87.71 million |
| Revenue Forecast by 2030 | USD 368.75 million |
| Growth rate from 2022 to 2030 | CAGR of 17.3% |
| Base Year | 2021 |
| Forecast Period | 2022 to 2030 |
| Segmentation | Circulating Biomarkers, Application |
| Companies Covered |
Biocept, Inc., Fluxion Biosciences, Inc., F. Hoffmann-La Roche Ltd., Sysmex Corporation, Thermo Fisher Scientific Inc. Menarini Group, NeoGenomics Laboratories, Inc., Qiagen N.V., and Myriad Genetics, I |
Key Players
Market Segmentation
Chapter 1. Introduction
1.1. Research Objective
1.2. Scope of the Study
1.3. Definition
Chapter 2. Research Methodology
2.1. Research Approach
2.2. Data Sources
2.3. Assumptions & Limitations
Chapter 3. Executive Summary
3.1. Market Snapshot
Chapter 4. Market Variables and Scope
4.1. Introduction
4.2. Market Classification and Scope
4.3. Industry Value Chain Analysis
4.3.1. Raw Material Procurement Analysis
4.3.2. Sales and Distribution Channel Analysis
4.3.3. Downstream Buyer Analysis
Chapter 5. COVID 19 Impact on Breast Cancer Liquid Biopsy Market
5.1. COVID-19 Landscape: Breast Cancer Liquid Biopsy Industry Impact
5.2. COVID 19 - Impact Assessment for the Industry
5.3. COVID 19 Impact: Global Major Government Policy
5.4. Market Trends and Opportunities in the COVID-19 Landscape
Chapter 6. Market Dynamics Analysis and Trends
6.1. Market Dynamics
6.1.1. Market Drivers
6.1.2. Market Restraints
6.1.3. Market Opportunities
6.2. Porter’s Five Forces Analysis
6.2.1. Bargaining power of suppliers
6.2.2. Bargaining power of buyers
6.2.3. Threat of substitute
6.2.4. Threat of new entrants
6.2.5. Degree of competition
Chapter 7. Competitive Landscape
7.1.1. Company Market Share/Positioning Analysis
7.1.2. Key Strategies Adopted by Players
7.1.3. Vendor Landscape
7.1.3.1. List of Suppliers
7.1.3.2. List of Buyers
Chapter 8. Global Breast Cancer Liquid Biopsy Market, By Circulating Biomarkers
8.1. Breast Cancer Liquid Biopsy Market, by Circulating Biomarkers, 2022-2030
8.1.1. Circulating Tumor Cells
8.1.1.1. Market Revenue and Forecast (2017-2030)
8.1.2. Circulating Cell-free DNA
8.1.2.1. Market Revenue and Forecast (2017-2030)
8.1.3. Extracellular Vesicles
8.1.3.1. Market Revenue and Forecast (2017-2030)
8.1.4. Other Circulating Biomarkers
8.1.4.1. Market Revenue and Forecast (2017-2030)
Chapter 9. Global Breast Cancer Liquid Biopsy Market, By Application
9.1. Breast Cancer Liquid Biopsy Market, by Application, 2022-2030
9.1.1. Targeting Treatment
9.1.1.1. Market Revenue and Forecast (2017-2030)
9.1.2. Companion Diagnostics
9.1.2.1. Market Revenue and Forecast (2017-2030)
9.1.3. Molecular
9.1.3.1. Market Revenue and Forecast (2017-2030)
9.1.4. Others
9.1.4.1. Market Revenue and Forecast (2017-2030)
Chapter 10. Global Breast Cancer Liquid Biopsy Market, Regional Estimates and Trend Forecast
10.1. North America
10.1.1. Market Revenue and Forecast, by Circulating Biomarkers (2017-2030)
10.1.2. Market Revenue and Forecast, by Application (2017-2030)
10.1.3. U.S.
10.1.3.1. Market Revenue and Forecast, by Circulating Biomarkers (2017-2030)
10.1.3.2. Market Revenue and Forecast, by Application (2017-2030)
10.1.4. Rest of North America
10.1.4.1. Market Revenue and Forecast, by Circulating Biomarkers (2017-2030)
10.1.4.2. Market Revenue and Forecast, by Application (2017-2030)
10.2. Europe
10.2.1. Market Revenue and Forecast, by Circulating Biomarkers (2017-2030)
10.2.2. Market Revenue and Forecast, by Application (2017-2030)
10.2.3. UK
10.2.3.1. Market Revenue and Forecast, by Circulating Biomarkers (2017-2030)
10.2.3.2. Market Revenue and Forecast, by Application (2017-2030)
10.2.4. Germany
10.2.4.1. Market Revenue and Forecast, by Circulating Biomarkers (2017-2030)
10.2.4.2. Market Revenue and Forecast, by Application (2017-2030)
10.2.5. France
10.2.5.1. Market Revenue and Forecast, by Circulating Biomarkers (2017-2030)
10.2.5.2. Market Revenue and Forecast, by Application (2017-2030)
10.2.6. Rest of Europe
10.2.6.1. Market Revenue and Forecast, by Circulating Biomarkers (2017-2030)
10.2.6.2. Market Revenue and Forecast, by Application (2017-2030)
10.3. APAC
10.3.1. Market Revenue and Forecast, by Circulating Biomarkers (2017-2030)
10.3.2. Market Revenue and Forecast, by Application (2017-2030)
10.3.3. India
10.3.3.1. Market Revenue and Forecast, by Circulating Biomarkers (2017-2030)
10.3.3.2. Market Revenue and Forecast, by Application (2017-2030)
10.3.4. China
10.3.4.1. Market Revenue and Forecast, by Circulating Biomarkers (2017-2030)
10.3.4.2. Market Revenue and Forecast, by Application (2017-2030)
10.3.5. Japan
10.3.5.1. Market Revenue and Forecast, by Circulating Biomarkers (2017-2030)
10.3.5.2. Market Revenue and Forecast, by Application (2017-2030)
10.3.6. Rest of APAC
10.3.6.1. Market Revenue and Forecast, by Circulating Biomarkers (2017-2030)
10.3.6.2. Market Revenue and Forecast, by Application (2017-2030)
10.4. MEA
10.4.1. Market Revenue and Forecast, by Circulating Biomarkers (2017-2030)
10.4.2. Market Revenue and Forecast, by Application (2017-2030)
10.4.3. GCC
10.4.3.1. Market Revenue and Forecast, by Circulating Biomarkers (2017-2030)
10.4.3.2. Market Revenue and Forecast, by Application (2017-2030)
10.4.4. North Africa
10.4.4.1. Market Revenue and Forecast, by Circulating Biomarkers (2017-2030)
10.4.4.2. Market Revenue and Forecast, by Application (2017-2030)
10.4.5. South Africa
10.4.5.1. Market Revenue and Forecast, by Circulating Biomarkers (2017-2030)
10.4.5.2. Market Revenue and Forecast, by Application (2017-2030)
10.4.6. Rest of MEA
10.4.6.1. Market Revenue and Forecast, by Circulating Biomarkers (2017-2030)
10.4.6.2. Market Revenue and Forecast, by Application (2017-2030)
10.5. Latin America
10.5.1. Market Revenue and Forecast, by Circulating Biomarkers (2017-2030)
10.5.2. Market Revenue and Forecast, by Application (2017-2030)
10.5.3. Brazil
10.5.3.1. Market Revenue and Forecast, by Circulating Biomarkers (2017-2030)
10.5.3.2. Market Revenue and Forecast, by Application (2017-2030)
10.5.4. Rest of LATAM
10.5.4.1. Market Revenue and Forecast, by Circulating Biomarkers (2017-2030)
10.5.4.2. Market Revenue and Forecast, by Application (2017-2030)
Chapter 11. Company Profiles
11.1. Biocept, Inc.
11.1.1. Company Overview
11.1.2. Product Offerings
11.1.3. Financial Performance
11.1.4. Recent Initiatives
11.2. Fluxion Biosciences, Inc.
11.2.1. Company Overview
11.2.2. Product Offerings
11.2.3. Financial Performance
11.2.4. Recent Initiatives
11.3. F. Hoffmann-La Roche Ltd.
11.3.1. Company Overview
11.3.2. Product Offerings
11.3.3. Financial Performance
11.3.4. Recent Initiatives
11.4. Sysmex Corporation
11.4.1. Company Overview
11.4.2. Product Offerings
11.4.3. Financial Performance
11.4.4. LTE Scientific
11.5. Thermo Fisher Scientific Inc. Menarini Group
11.5.1. Company Overview
11.5.2. Product Offerings
11.5.3. Financial Performance
11.5.4. Recent Initiatives
11.6. NeoGenomics Laboratories, Inc.
11.6.1. Company Overview
11.6.2. Product Offerings
11.6.3. Financial Performance
11.6.4. Recent Initiatives
11.7. Qiagen N.V.
11.7.1. Company Overview
11.7.2. Product Offerings
11.7.3. Financial Performance
11.7.4. Recent Initiatives
11.8. Myriad Genetics, Inc.
11.8.1. Company Overview
11.8.2. Product Offerings
11.8.3. Financial Performance
11.8.4. Recent Initiatives
Chapter 12. Research Methodology
12.1. Primary Research
12.2. Secondary Research
12.3. Assumptions
Chapter 13. Appendix
13.1. About Us
13.2. Glossary of Terms
 Cross-segment Market Size and Analysis for
Mentioned Segments
Cross-segment Market Size and Analysis for
Mentioned Segments
 Additional Company Profiles (Upto 5 With No Cost)
Additional Company Profiles (Upto 5 With No Cost)
 Additional Countries (Apart From Mentioned Countries)
Additional Countries (Apart From Mentioned Countries)
 Country/Region-specific Report
Country/Region-specific Report
 Go To Market Strategy
Go To Market Strategy
 Region Specific Market Dynamics
Region Specific Market Dynamics Region Level Market Share
Region Level Market Share Import Export Analysis
Import Export Analysis Production Analysis
Production Analysis Others
Others