The global cardiac resynchronization therapy market was surpassed at USD 4.30 billion in 2022 and is expected to hit around USD 6.89 billion by 2032, growing at a CAGR of 4.82% from 2023 to 2032.
Key Pointers
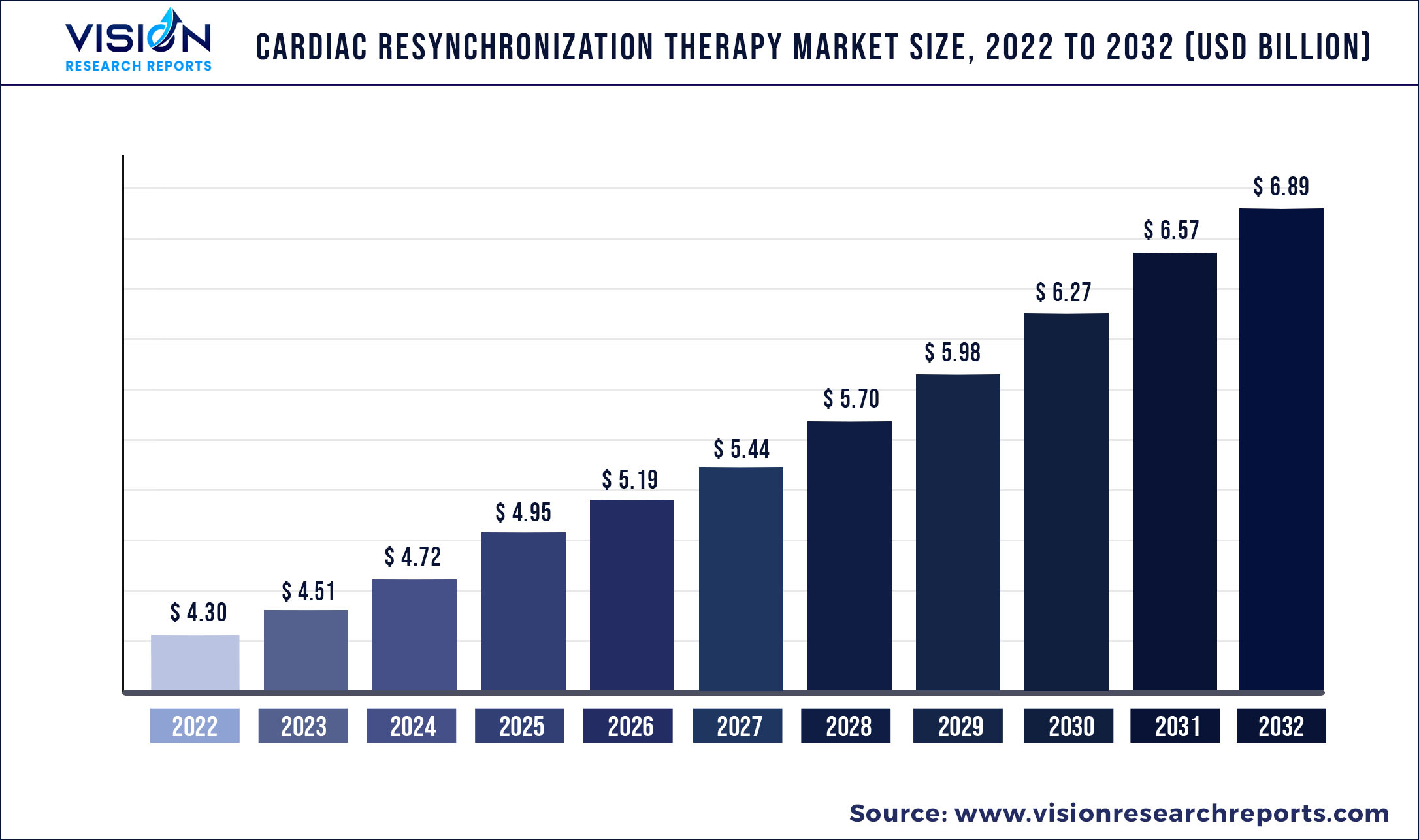
Growing product developments, an aging population, initiatives by key companies, and the prevalence of cardiovascular diseases are some of the key drivers of this market. Boston Scientific, for instance, reported cumulative distribution of around 389,000 CRT-Ds globally as of January 2021. During 2020, the company sold around 33,000 CRT-Ds; out of which, about 16,500 were registered in the U.S., which is indicative of high demand.
The aging population is a key factor estimated to fuel market growth. Statistics Korea estimated that about 8.53 million individuals are aged 65 years and older in 2021. This number is expected to reach 12.98 million by 2030. The aging population indicates a growing number of people at risk of chronic diseases, such as congestive heart failure, hypertension, and arrhythmia. This is anticipated to contribute to market growth in the country. According to a survey conducted by the European Society of Cardiology and published in the Archives of Cardiovascular Diseases in 2019, French patients were found to be more likely to receive a CRT-P implant compared to the rest of Europe. The patients were identified to be comparatively older, with about 44.7% aged 75 years and above, had fewer co-morbidities, and less severe heart failure symptoms.
Rising product improvements, R&D initiatives by market players, and the prevalence of cardiovascular diseases are expected to fuel market growth. For instance, in February 2020, Abbott received CE Marking for its Gallant ICDs and CRT-D devices. These technologies enhance the company's solutions by enabling remote monitoring through Abbott's myMerlinPuls App. MultiPoint Pacing and SyncAV feature further enhance the Gallant CRT-D system to improve patient response to CRT therapy. This supported the company’s growth objectives. The Spanish Pacemaker Registry reported that, in 2020, about 3,850 CRT devices were implanted in Spain. Out of the implanted devices, 2,387 were CRT-D devices and 1,463 were CRT-P devices. It was reported that recipients were 78.8 years old on average. Syncope was found to be the symptom that led to implantation in 41.8% of cases. Dizziness and heart failure were found to be the other symptoms.
Cardiac Resynchronization Therapy Market Segmentations:
| By Product | By End-use |
|
CRT-Defibrillator CRT-Pacemaker |
Hospital Cardiac Center Others |
Cardiac Resynchronization Therapy Market Key Players and Regions Segmentations:
| Key Players | Regions |
|
Abbott Boston Scientific Corporation Biotronik SE & Co., KG MicroPort Scientific Corporation Livanova plc. |
Europe Asia Pacific Latin America MEA |
Chapter 1. Introduction
1.1. Research Objective
1.2. Scope of the Study
1.3. Definition
Chapter 2. Research Methodology
2.1. Research Approach
2.2. Data Sources
2.3. Assumptions & Limitations
Chapter 3. Executive Summary
3.1. Market Snapshot
Chapter 4. Market Variables and Scope
4.1. Introduction
4.2. Market Classification and Scope
4.3. Industry Value Chain Analysis
4.3.1. Raw Material Procurement Analysis
4.3.2. Sales and Distribution Channel Analysis
4.3.3. Downstream Buyer Analysis
Chapter 5. COVID 19 Impact on Cardiac Resynchronization Therapy Market
5.1. COVID-19 Landscape: Cardiac Resynchronization Therapy Industry Impact
5.2. COVID 19 - Impact Assessment for the Industry
5.3. COVID 19 Impact: Global Major Government Policy
5.4. Market Trends and Opportunities in the COVID-19 Landscape
Chapter 6. Market Dynamics Analysis and Trends
6.1. Market Dynamics
6.1.1. Market Drivers
6.1.2. Market Restraints
6.1.3. Market Opportunities
6.2. Porter’s Five Forces Analysis
6.2.1. Bargaining power of suppliers
6.2.2. Bargaining power of buyers
6.2.3. Threat of substitute
6.2.4. Threat of new entrants
6.2.5. Degree of competition
Chapter 7. Competitive Landscape
7.1.1. Company Market Share/Positioning Analysis
7.1.2. Key Strategies Adopted by Players
7.1.3. Vendor Landscape
7.1.3.1. List of Suppliers
7.1.3.2. List of Buyers
Chapter 8. Global Cardiac Resynchronization Therapy Market, By Product
8.1. Cardiac Resynchronization Therapy Market, by Product, 2023-2032
8.1.1. CRT-Defibrillator
8.1.1.1. Market Revenue and Forecast (2019-2032)
8.1.2. CRT-Pacemaker
8.1.2.1. Market Revenue and Forecast (2019-2032)
Chapter 9. Global Cardiac Resynchronization Therapy Market, By End-use
9.1. Cardiac Resynchronization Therapy Market, by End-use, 2023-2032
9.1.1. Hospital
9.1.1.1. Market Revenue and Forecast (2019-2032)
9.1.2. Cardiac Center
9.1.2.1. Market Revenue and Forecast (2019-2032)
9.1.3. Others
9.1.3.1. Market Revenue and Forecast (2019-2032)
Chapter 10. Global Cardiac Resynchronization Therapy Market, Regional Estimates and Trend Forecast
10.1. North America
10.1.1. Market Revenue and Forecast, by Product (2019-2032)
10.1.2. Market Revenue and Forecast, by End-use (2019-2032)
10.1.3. U.S.
10.1.3.1. Market Revenue and Forecast, by Product (2019-2032)
10.1.3.2. Market Revenue and Forecast, by End-use (2019-2032)
10.1.4. Rest of North America
10.1.4.1. Market Revenue and Forecast, by Product (2019-2032)
10.1.4.2. Market Revenue and Forecast, by End-use (2019-2032)
10.2. Europe
10.2.1. Market Revenue and Forecast, by Product (2019-2032)
10.2.2. Market Revenue and Forecast, by End-use (2019-2032)
10.2.3. UK
10.2.3.1. Market Revenue and Forecast, by Product (2019-2032)
10.2.3.2. Market Revenue and Forecast, by End-use (2019-2032)
10.2.4. Germany
10.2.4.1. Market Revenue and Forecast, by Product (2019-2032)
10.2.4.2. Market Revenue and Forecast, by End-use (2019-2032)
10.2.5. France
10.2.5.1. Market Revenue and Forecast, by Product (2019-2032)
10.2.5.2. Market Revenue and Forecast, by End-use (2019-2032)
10.2.6. Rest of Europe
10.2.6.1. Market Revenue and Forecast, by Product (2019-2032)
10.2.6.2. Market Revenue and Forecast, by End-use (2019-2032)
10.3. APAC
10.3.1. Market Revenue and Forecast, by Product (2019-2032)
10.3.2. Market Revenue and Forecast, by End-use (2019-2032)
10.3.3. India
10.3.3.1. Market Revenue and Forecast, by Product (2019-2032)
10.3.3.2. Market Revenue and Forecast, by End-use (2019-2032)
10.3.4. China
10.3.4.1. Market Revenue and Forecast, by Product (2019-2032)
10.3.4.2. Market Revenue and Forecast, by End-use (2019-2032)
10.3.5. Japan
10.3.5.1. Market Revenue and Forecast, by Product (2019-2032)
10.3.5.2. Market Revenue and Forecast, by End-use (2019-2032)
10.3.6. Rest of APAC
10.3.6.1. Market Revenue and Forecast, by Product (2019-2032)
10.3.6.2. Market Revenue and Forecast, by End-use (2019-2032)
10.4. MEA
10.4.1. Market Revenue and Forecast, by Product (2019-2032)
10.4.2. Market Revenue and Forecast, by End-use (2019-2032)
10.4.3. GCC
10.4.3.1. Market Revenue and Forecast, by Product (2019-2032)
10.4.3.2. Market Revenue and Forecast, by End-use (2019-2032)
10.4.4. North Africa
10.4.4.1. Market Revenue and Forecast, by Product (2019-2032)
10.4.4.2. Market Revenue and Forecast, by End-use (2019-2032)
10.4.5. South Africa
10.4.5.1. Market Revenue and Forecast, by Product (2019-2032)
10.4.5.2. Market Revenue and Forecast, by End-use (2019-2032)
10.4.6. Rest of MEA
10.4.6.1. Market Revenue and Forecast, by Product (2019-2032)
10.4.6.2. Market Revenue and Forecast, by End-use (2019-2032)
10.5. Latin America
10.5.1. Market Revenue and Forecast, by Product (2019-2032)
10.5.2. Market Revenue and Forecast, by End-use (2019-2032)
10.5.3. Brazil
10.5.3.1. Market Revenue and Forecast, by Product (2019-2032)
10.5.3.2. Market Revenue and Forecast, by End-use (2019-2032)
10.5.4. Rest of LATAM
10.5.4.1. Market Revenue and Forecast, by Product (2019-2032)
10.5.4.2. Market Revenue and Forecast, by End-use (2019-2032)
Chapter 11. Company Profiles
11.1. Abbott
11.1.1. Company Overview
11.1.2. Product Offerings
11.1.3. Financial Performance
11.1.4. Recent Initiatives
11.2. Boston Scientific Corporation
11.2.1. Company Overview
11.2.2. Product Offerings
11.2.3. Financial Performance
11.2.4. Recent Initiatives
11.3. Biotronik SE & Co., KG
11.3.1. Company Overview
11.3.2. Product Offerings
11.3.3. Financial Performance
11.3.4. Recent Initiatives
11.4. MicroPort Scientific Corporation
11.4.1. Company Overview
11.4.2. Product Offerings
11.4.3. Financial Performance
11.4.4. LTE Scientific
11.5. Livanova plc.
11.5.1. Company Overview
11.5.2. Product Offerings
11.5.3. Financial Performance
11.5.4. Recent Initiatives
Chapter 12. Research Methodology
12.1. Primary Research
12.2. Secondary Research
12.3. Assumptions
Chapter 13. Appendix
13.1. About Us
13.2. Glossary of Terms
 Cross-segment Market Size and Analysis for
Mentioned Segments
Cross-segment Market Size and Analysis for
Mentioned Segments
 Additional Company Profiles (Upto 5 With No Cost)
Additional Company Profiles (Upto 5 With No Cost)
 Additional Countries (Apart From Mentioned Countries)
Additional Countries (Apart From Mentioned Countries)
 Country/Region-specific Report
Country/Region-specific Report
 Go To Market Strategy
Go To Market Strategy
 Region Specific Market Dynamics
Region Specific Market Dynamics Region Level Market Share
Region Level Market Share Import Export Analysis
Import Export Analysis Production Analysis
Production Analysis Others
Others