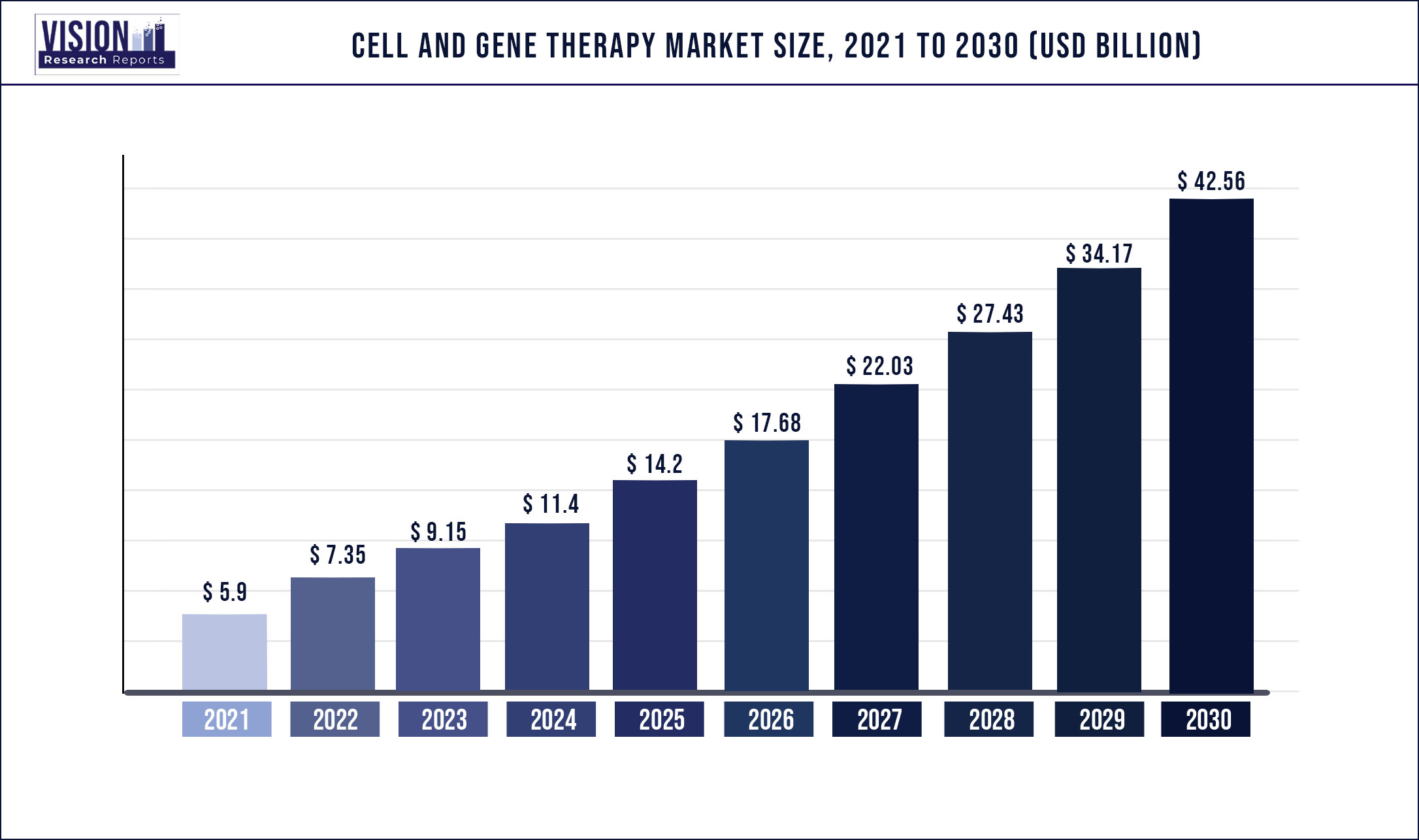
The cell and gene therapy market size was valued at USD 5.9 billion in 2021 and is expected to hit around USD 42.56 billion by 2030, growing at a CAGR of 39.42 % during the forecast period 2022 to 2030.
Cell and gene therapies (C>) continue to be at the very center of healthcare innovation and are the fastest-growing areas of therapeutics, having already contributed to some of the most significant disruptions in the pharmaceutical and biotech industries. Continuous advancements in C> are transforming how certain diseases are treated and potentially cured, and significantly changing healthcare outcomes. So, what’s next? Where is C> headed? The following explores the industry dynamics that will shape the C> space throughout the next decade.
COST VERSUS VALUE
The US Food and Drug Administration has approved seven cell and gene therapy drugs, but the new product pipeline has approximately 1,200 experimental therapies, with more than half of these in Phase 2 clinical trials. CAGR estimates are about 15% for cell therapies and nearly 30% for gene therapies, and the Alliance for Regenerative Medicine points to a rapid rise in cell and gene therapy developers, with 1,100 such ventures in 2020, an increase of about 10% from 2019.1-3 The industry is still transitioning out of the early development phase and sits at the very start of the rapid growth phase with a lot of opportunity ahead.
Showing the therapies are providing a lifelong cure and not just another type of ongoing treatment will be key to justifying the pricing and gaining acceptance from patients, providers, and payors, be they insurance or governmental. Unfortunately, when new cell and gene products hit the market, the discussion in media often focuses on the price tag instead of highlighting the availability of a new therapy with the potential to cure instead of simply managing what may have been a previously untreatable disease.
This is creating increased pressure to drive down manufacturing costs – a common challenge across the pharma and biotech industries. In C>, this effort will be focused on several fronts, such as increasing batch sizes, standardizing and streamlining processes, and reducing the cost of the materials used in manufacturing. This will go hand in hand with developing processes that provide high yields of consistent product.
The markets will also have an influence. Where there is a broader market upon which to spread the development costs, such as cancers, where there is usually a larger patient population, therapies can hit a lower price point. For the rare or orphan diseases, it will be harder to spread out costs. But there is potential to achieve lower costs in the long run, as in these cases, it may be fruitful to have a platform, such as a particular viral vector, upon which multiple therapeutics can be more easily developed, thereby reducing some of R&D costs.
The following factors are likely to contribute to the growth of the cell and gene therapy market
In the cell and gene therapy field, gene therapy gathered the pace last from 2 decades because of the discovery of several genes responsible for mutation in various diseases. The advancement in the cell & gene therapy field and innovative technologies give the new era for biological therapeutics. Also, PRIME Designation and marketing authorization for products provide a new opportunity for the manufacturer’s financing and revenue generation.
CHALLENGES AROUND CAPACITY, TECHNICAL CAPABILITIES & EXPERTISE
Physical capacity to produce gene and cell therapy products continues to be a bottleneck. Even as more capacity is added, the industry seems to be growing even faster. Some of the expansion is being hampered by the lack of availability of materials – the supply of items, such as plasmids, media, and the numerous disposables from pipettes to bioreactors, has continued to be tight.
The solution will, in part, come from increasing the production of these materials. There also has to be a greater focus on learning how to better utilize the supply so more product can be manufactured with the same amount of material.
The ability to scale up the manufacture of therapies in a cost-effective manner is also essential. The industry must find systems that can be run as a “small” version for the preclinical and early trial work but then be scaled up into larger equipment for commercial use, rather than simply scaling volumes by duplicating small systems that inherently uses more resources and physical space within facilities.
The availability of a workforce with appropriate skills is also going to continue to be a challenge. Currently, the industry is largely clustered in a few locations – in the US, there are a few key metropolitan areas on the east and west coasts that have very high concentrations of C> companies. As a result, the companies in these locations are running into problems finding the workers they need. This competition for the limited number of workers is driving up wages and that is anathema to the idea of reducing costs. It also means projects are delayed because the expertise and workers needed to do the work are not available. There may be several approaches to solving this. The industry may expand into locations other than the east and west coasts; a longer-term solution however would involve federal, state, and local governments supporting and funding dedicated life science workforces. The cities, states, and countries that invest in educating and training a workforce will attract the companies and the jobs and tax dollars they bring. Any country that wants to stay in the forefront of this industry must be able to supply the workers, otherwise they will see the industry and jobs move elsewhere.
REGULATORY APPROACHES
A decade ago, the new treatments in the G&CT space advancing to the clinic sparked excitement about the eventual path to commercialization. This spurred regulatory agencies to work with researchers and companies to move these treatments forward, but given the uncharted waters, even the questions to be asked were evolving. Now, some of these treatments have been approved, and parties on both sides of the regulatory equation have learned how to evaluate the development phase and the important regulatory factors and questions. For example, progress in regulatory perspectives has led to a preference for a single producer cell line clone for use through all development phases.
Regulators, such as the FDA and the European Medicines Agency (EMA), have published formal and informal guidelines to help viral vector companies produce products. Due to the evolving nature of the C> industry, regulations surrounding these products are updated to keep up with the rapidly growing field and its development – for example, the FDA has provided or updated nine guidance documents within the last 2 years.4
This will continue to evolve, and each new type of therapy will bring its own learning curve to the regulatory process. Moreover, it will continue to be important for gene therapy developers and manufacturers to work hand in hand with regulatory agencies as more open discussions around issues, challenges, and best practice will put the industry in a better position to collaborate on any future regulations and guidance.
DRIVING THE INDUSTRY FORWARD
If the industry wants to keep moving up the steep growth curve, then capacity and raw materials must be readily available. The innovations in therapies, platforms, and processes will all come with time, money and increasing availability of resources.
Investment into basic research at the university level and into small start-ups coming from venture capital, private equity, industry, and the capital markets will be essential – university labs and small start-ups are well-positioned for the needed innovation. Invariably, these companies start out with a small team that is willing to take risks and then grow into larger companies or go through a series of acquisitions that bring those innovations into larger biotechs and big pharma. These larger entities can use their resources and expertise to commercialize the product and get it to market.
Outsourcing to niche CDMOs with the smaller scale capabilities and expertise required to develop scalable processes at this stage will be essential to expediting a C>’s path to market.
KEY HIGHLIGHTS
WHAT’S NEXT?
Throughout the past 5 or 6 years, a lot of the excitement, especially for investors, has been around cancer therapies. That work has largely been on bloodborne cancers, but there are now approaches being applied to solid tumor therapies. There is likely to be a continued focus on LV and it being used in other CAR-T and Natural Killer cell therapies both for bloodborne cancers as well as solid tumors.
As previously noted, most LV vectors currently use the VSVG envelope, and one area of innovation will involve developing other envelopes to see if there are ways to use LV vectors in vivo and targeting specific organs or used in combination with some immune suppression to reduce the immune response to the virus.
Other platforms will likely develop over time. There has been interest in foamy viruses – a RV belonging to the Spumavirus genus – but they proved extremely difficult to make. Recently, there has been a great deal of interest in using bocavirus as a vector platform, it shows promise in systemic applications like an AAV, but with the added benefit of being capable of carrying a transgene almost as large as that of an LV vector. However, innovation in the space must be facilitated through investment and addressing several challenges that are currently hamstringing progress.
CELL AND GENE THERAPY MARKET SEGMENTATION
GEOGRAPHICAL OUTLOOK
North America: High economic status and high expenditure on healthcare services drive the cell and gene therapy market in North America. National Health Institutes, industries, academic institutes, and hospitals are the significant contributors of sponsorship and financial funding for cell and gene therapy products.
Europe: The increasing funding for cell and gene therapy drives the cell and gene therapy market growth consistently in Europe. Around USD 2.6 billion financings were reported in Europe for CGTs in 2020, which increased by 103% compared to previous years. In the cell therapy segment, USD 1.8 billion and in gene therapy, USD 2.3 billion funding accounted in 2020, which increased by 196% and 111% growth respectively
VENDOR LANDSCAPE
KEY QUESTIONS ANSWERED:
Market Segmentation
By Therapy Type
By Application
By End-Users
Key Vendors
Other Prominent Vendors
Upcoming Vendors
Chapter 1. Introduction
1.1. Research Objective
1.2. Scope of the Study
1.3. Definition
Chapter 2. Research Methodology
2.1. Research Approach
2.2. Data Sources
2.3. Assumptions & Limitations
Chapter 3. Executive Summary
3.1. Market Snapshot
Chapter 4. Market Variables and Scope
4.1. Introduction
4.2. Market Classification and Scope
4.3. Industry Value Chain Analysis
4.3.1. Raw Material Procurement Analysis
4.3.2. Sales and Distribution Channel Analysis
4.3.3. Downstream Buyer Analysis
Chapter 5. COVID 19 Impact on Cell and Gene Therapy Market
5.1. COVID-19 Landscape: Cell and Gene Therapy Industry Impact
5.2. COVID 19 - Impact Assessment for the Industry
5.3. COVID 19 Impact: Global Major Government Policy
5.4. Market Trends and Opportunities in the COVID-19 Landscape
Chapter 6. Market Dynamics Analysis and Trends
6.1. Market Dynamics
6.1.1. Market Drivers
6.1.2. Market Restraints
6.1.3. Market Opportunities
6.2. Porter’s Five Forces Analysis
6.2.1. Bargaining power of suppliers
6.2.2. Bargaining power of buyers
6.2.3. Threat of substitute
6.2.4. Threat of new entrants
6.2.5. Degree of competition
Chapter 7. Competitive Landscape
7.1.1. Company Market Share/Positioning Analysis
7.1.2. Key Strategies Adopted by Players
7.1.3. Vendor Landscape
7.1.3.1. List of Suppliers
7.1.3.2. List of Buyers
Chapter 8. Global Cell and Gene Therapy Market, By Therapy Type
8.1. Cell and Gene Therapy Market, by Therapy Type, 2022-2030
8.1.1 Gene Therapy
8.1.1.1. Market Revenue and Forecast (2017-2030)
8.1.2. Cell Therapy
8.1.2.1. Market Revenue and Forecast (2017-2030)
Chapter 9. Global Cell and Gene Therapy Market, By Application
9.1. Cell and Gene Therapy Market, by Application, 2022-2030
9.1.1. Oncology
9.1.1.1. Market Revenue and Forecast (2017-2030)
9.1.2. Genetic Disorders
9.1.2.1. Market Revenue and Forecast (2017-2030)
9.1.3. Dermatology Disorders
9.1.3.1. Market Revenue and Forecast (2017-2030)
9.1.4. Musculoskeletal Disorders
9.1.4.1. Market Revenue and Forecast (2017-2030)
9.1.5. Others
9.1.5.1. Market Revenue and Forecast (2017-2030)
Chapter 10. Global Cell and Gene Therapy Market, By End-Users
10.1. Cell and Gene Therapy Market, by End-Users, 2022-2030
10.1.1. Hospitals
10.1.1.1. Market Revenue and Forecast (2017-2030)
10.1.2. Cancer Care Centers
10.1.2.1. Market Revenue and Forecast (2017-2030)
10.1.3. Wound Care Centers
10.1.3.1. Market Revenue and Forecast (2017-2030)
10.1.4. Others
10.1.4.1. Market Revenue and Forecast (2017-2030)
Chapter 11. Global Cell and Gene Therapy Market, Regional Estimates and Trend Forecast
11.1. North America
11.1.1. Market Revenue and Forecast, by Therapy Type (2017-2030)
11.1.2. Market Revenue and Forecast, by Application (2017-2030)
11.1.3. Market Revenue and Forecast, by End-Users (2017-2030)
11.1.4. U.S.
11.1.4.1. Market Revenue and Forecast, by Therapy Type (2017-2030)
11.1.4.2. Market Revenue and Forecast, by Application (2017-2030)
11.1.4.3. Market Revenue and Forecast, by End-Users (2017-2030)
11.1.5. Rest of North America
11.1.5.1. Market Revenue and Forecast, by Therapy Type (2017-2030)
11.1.5.2. Market Revenue and Forecast, by Application (2017-2030)
11.1.5.3. Market Revenue and Forecast, by End-Users (2017-2030)
11.2. Europe
11.2.1. Market Revenue and Forecast, by Therapy Type (2017-2030)
11.2.2. Market Revenue and Forecast, by Application (2017-2030)
11.2.3. Market Revenue and Forecast, by End-Users (2017-2030)
11.2.4. UK
11.2.4.1. Market Revenue and Forecast, by Therapy Type (2017-2030)
11.2.4.2. Market Revenue and Forecast, by Application (2017-2030)
11.2.4.3. Market Revenue and Forecast, by End-Users (2017-2030)
11.2.5. Germany
11.2.5.1. Market Revenue and Forecast, by Therapy Type (2017-2030)
11.2.5.2. Market Revenue and Forecast, by Application (2017-2030)
11.2.5.3. Market Revenue and Forecast, by End-Users (2017-2030)
11.2.6. France
11.2.6.1. Market Revenue and Forecast, by Therapy Type (2017-2030)
11.2.6.2. Market Revenue and Forecast, by Application (2017-2030)
11.2.6.3. Market Revenue and Forecast, by End-Users (2017-2030)
11.2.7. Rest of Europe
11.2.7.1. Market Revenue and Forecast, by Therapy Type (2017-2030)
11.2.7.2. Market Revenue and Forecast, by Application (2017-2030)
11.2.7.3. Market Revenue and Forecast, by End-Users (2017-2030)
11.3. APAC
11.3.1. Market Revenue and Forecast, by Therapy Type (2017-2030)
11.3.2. Market Revenue and Forecast, by Application (2017-2030)
11.3.3. Market Revenue and Forecast, by End-Users (2017-2030)
11.3.4. India
11.3.4.1. Market Revenue and Forecast, by Therapy Type (2017-2030)
11.3.4.2. Market Revenue and Forecast, by Application (2017-2030)
11.3.4.3. Market Revenue and Forecast, by End-Users (2017-2030)
11.3.5. China
11.3.5.1. Market Revenue and Forecast, by Therapy Type (2017-2030)
11.3.5.2. Market Revenue and Forecast, by Application (2017-2030)
11.3.5.3. Market Revenue and Forecast, by End-Users (2017-2030)
11.3.6. Japan
11.3.6.1. Market Revenue and Forecast, by Therapy Type (2017-2030)
11.3.6.2. Market Revenue and Forecast, by Application (2017-2030)
11.3.6.3. Market Revenue and Forecast, by End-Users (2017-2030)
11.3.7. Rest of APAC
11.3.7.1. Market Revenue and Forecast, by Therapy Type (2017-2030)
11.3.7.2. Market Revenue and Forecast, by Application (2017-2030)
11.3.7.3. Market Revenue and Forecast, by End-Users (2017-2030)
11.4. MEA
11.4.1. Market Revenue and Forecast, by Therapy Type (2017-2030)
11.4.2. Market Revenue and Forecast, by Application (2017-2030)
11.4.3. Market Revenue and Forecast, by End-Users (2017-2030)
11.4.4. GCC
11.4.4.1. Market Revenue and Forecast, by Therapy Type (2017-2030)
11.4.4.2. Market Revenue and Forecast, by Application (2017-2030)
11.4.4.3. Market Revenue and Forecast, by End-Users (2017-2030)
11.4.5. North Africa
11.4.5.1. Market Revenue and Forecast, by Therapy Type (2017-2030)
11.4.5.2. Market Revenue and Forecast, by Application (2017-2030)
11.4.5.3. Market Revenue and Forecast, by End-Users (2017-2030)
11.4.6. South Africa
11.4.6.1. Market Revenue and Forecast, by Therapy Type (2017-2030)
11.4.6.2. Market Revenue and Forecast, by Application (2017-2030)
11.4.6.3. Market Revenue and Forecast, by End-Users (2017-2030)
11.4.7. Rest of MEA
11.4.7.1. Market Revenue and Forecast, by Therapy Type (2017-2030)
11.4.7.2. Market Revenue and Forecast, by Application (2017-2030)
11.4.7.3. Market Revenue and Forecast, by End-Users (2017-2030)
11.5. Latin America
11.5.1. Market Revenue and Forecast, by Therapy Type (2017-2030)
11.5.2. Market Revenue and Forecast, by Application (2017-2030)
11.5.3. Market Revenue and Forecast, by End-Users (2017-2030)
11.5.4. Brazil
11.5.4.1. Market Revenue and Forecast, by Therapy Type (2017-2030)
11.5.4.2. Market Revenue and Forecast, by Application (2017-2030)
11.5.4.3. Market Revenue and Forecast, by End-Users (2017-2030)
11.5.5. Rest of LATAM
11.5.5.1. Market Revenue and Forecast, by Therapy Type (2017-2030)
11.5.5.2. Market Revenue and Forecast, by Application (2017-2030)
11.5.5.3. Market Revenue and Forecast, by End-Users (2017-2030)
Chapter 12. Company Profiles
12.1. Gilead sciences
12.1.1. Company Overview
12.1.2. Product Offerings
12.1.3. Financial Performance
12.1.4. Recent Initiatives
12.2. Novartis
12.2.1. Company Overview
12.2.2. Product Offerings
12.2.3. Financial Performance
12.2.4. Recent Initiatives
12.3. Smith Nephew (Osiris Therapeutic)
12.3.1. Company Overview
12.3.2. Product Offerings
12.3.3. Financial Performance
12.3.4. Recent Initiatives
12.4. Amgen
12.4.1. Company Overview
12.4.2. Product Offerings
12.4.3. Financial Performance
12.4.4. Recent Initiatives
12.5. Organogenesis
12.5.1. Company Overview
12.5.2. Product Offerings
12.5.3. Financial Performance
12.5.4. Recent Initiatives
12.6. Roche (Spark Therapeutics)
12.6.1. Company Overview
12.6.2. Product Offerings
12.6.3. Financial Performance
12.6.4. Recent Initiatives
12.7. Dendreon
12.7.1. Company Overview
12.7.2. Product Offerings
12.7.3. Financial Performance
12.7.4. Recent Initiatives
12.8. Vericel
12.8.1. Company Overview
12.8.2. Product Offerings
12.8.3. Financial Performance
12.8.4. Recent Initiatives
12.9. Bristol-Myers Squibb
12.9.1. Company Overview
12.9.2. Product Offerings
12.9.3. Financial Performance
12.9.4. Recent Initiatives
Chapter 13. Research Methodology
13.1. Primary Research
13.2. Secondary Research
13.3. Assumptions
Chapter 14. Appendix
14.1. About Us
14.2. Glossary of Terms
 Cross-segment Market Size and Analysis for
Mentioned Segments
Cross-segment Market Size and Analysis for
Mentioned Segments
 Additional Company Profiles (Upto 5 With No Cost)
Additional Company Profiles (Upto 5 With No Cost)
 Additional Countries (Apart From Mentioned Countries)
Additional Countries (Apart From Mentioned Countries)
 Country/Region-specific Report
Country/Region-specific Report
 Go To Market Strategy
Go To Market Strategy
 Region Specific Market Dynamics
Region Specific Market Dynamics Region Level Market Share
Region Level Market Share Import Export Analysis
Import Export Analysis Production Analysis
Production Analysis Others
Others