The global clinical trials market size was estimated at around USD 57.39 billion in 2023 and it is projected to hit around USD 92.77 billion by 2033, growing at a CAGR of 4.92% from 2024 to 2033.
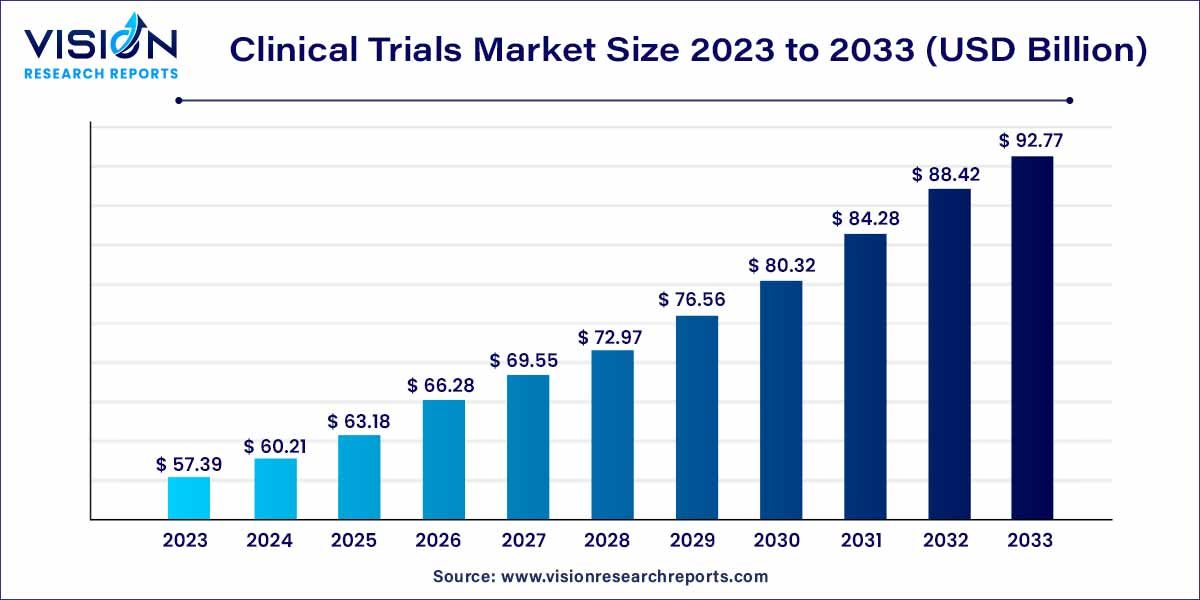
The global clinical trials market is witnessing significant growth and evolution, propelled by advancements in medical research, increasing prevalence of diseases, and a growing emphasis on personalized medicine. This comprehensive overview delves into the key facets of the clinical trials market, shedding light on its current landscape and future prospects.
The robust growth of the clinical trials market can be attributed to a confluence of factors propelling advancements in medical research and therapeutic innovation. Firstly, the escalating global burden of chronic diseases has intensified the demand for novel treatments, fostering a surge in clinical trial activities. Secondly, the continuous evolution of technology within the healthcare sector, including sophisticated data analytics and virtual trial capabilities, has significantly streamlined trial processes, enhancing efficiency and accelerating research timelines. Moreover, the increasing emphasis on personalized medicine, driven by a deeper understanding of genomic and molecular aspects, has spurred the exploration of targeted therapies through clinical trials. These factors collectively contribute to the dynamic expansion of the clinical trials market, positioning it as a pivotal arena for driving medical breakthroughs and shaping the future of healthcare.
The Phase III segment emerged as the market leader, constituting 54% of the total revenue share in 2023. The growth in clinical trial statistics underscores the increasing prevalence of Phase III trials, involving a substantial number of subjects. With a median cost exceeding USD 19.0 million for a single Phase III trial, this phase demands a higher patient count and often extends over a more prolonged treatment period. According to a clinical trial logistics survey conducted by Nice Insights, 35.0% of Phase III clinical trials are outsourced, a trend expected to rise due to the escalating number of investigational drugs advancing to the next phase.
Anticipated to experience significant growth throughout the analysis period, the Phase II segment stands as the second most expensive stage after Phase III studies. This two-part study involves exploring doses and efficacy in the initial phase and finalizing the dose in the subsequent part, playing a pivotal role, particularly in oncology-related studies. The FDA estimates that approximately 33.0% of investigational drugs typically undergo Phase II trials. Furthermore, the market is bolstered by numerous therapeutics and vaccines in Phase II targeting oncology treatment.
In 2023, the interventional studies segment emerged as the market leader, wielding significant influence. This method stands out as one of the most prominent approaches employed in clinical trials. Notably, interventional studies constituted over 75.0% of the total registered studies in 2022. Among these, the majority focused on drug or biologics, followed by clinical procedures, behavioral interventions, and device-related studies. A noteworthy trend is observed in the results, with interventional studies contributing to 94.0% of the total studies that posted outcomes. Within this subset, drug or biologics trials took the lead, followed by behavioral, device-related, and clinical procedure intervention studies.
The expanded access trials segment, also known as compassionate use trials, is poised to witness a significant Compound Annual Growth Rate (CAGR) during the forecast period. This approach serves as a crucial pathway for patients grappling with serious medical conditions, where satisfactory therapies are lacking, enabling them to undergo treatment outside the standard trial framework. The growth of the expanded access trials segment is expected to be fueled by increasing innovation in clinical trial methodologies. Notably, numerous oncology drugs are routinely administered to patients before receiving approval from the U.S. FDA, constituting a significant portion of the expanded access trial domain and contributing to accelerated revenue growth within this segment.
Oncology segment accounted for largest market revenue share in 2023. As per the U.S. FDA and various other sources, more than USD 38.0 billion is currently being spent by the pharmaceutical industry on the pre-clinical and clinical development of oncology therapy products. The cardiovascular condition segment is also anticipated to witness lucrative growth over the forecast period. The growing prevalence and increased demand for cost-effective medications worldwide have led to significant investment in R&D in this segment with more than 190 drugs in the pipeline. The majority of the drugs in the pipeline are indicated for heart failure, lipid disorders, vascular diseases, and stroke. Growing demand for cost-effective medicines in low- and middle-income countries is expected to boost the R&D investment by the government, thereby strengthening the market growth.
In 2023, pharmaceutical and biopharmaceutical companies secured the largest share of market revenue. This dominance is attributed to the heightened interest of the pharmaceutical industry in the research domain. Notably, there has been a discernible increase in the number of clinical trials funded by pharmaceutical and biopharmaceutical companies, underscoring their pivotal role in financing research for the development of new drugs.
The patient recruitment and retention segment also commanded a significant market share in 2023. This prominence can be attributed to the escalating number of clinical studies globally, driving the demand for clinical trial patient recruitment services. The market is further buoyed by the increasing funding for clinical research and the widespread adoption of digital technologies in this field. Substantial investments in Research and Development (R&D), coupled with government support for clinical trials, contribute to the segment's growth. The market expansion is additionally fueled by the presence of leading Contract Research Organizations (CROs) offering comprehensive support services, including patient recruitment. The active participation of multinational pharmaceutical and biopharmaceutical companies further enhances the overall market landscape.
In 2023, North America claimed a significant share of the global market at 51% and is projected to sustain its dominance throughout the forecast period. This dominance can be attributed to the region's increased Research and Development (R&D) investments and the widespread adoption of new technologies in clinical trials. Notably, market players such as IQVIA and PRA Health Sciences are implementing virtual services across various stages of clinical trials, contributing to the anticipated growth of the North American market.
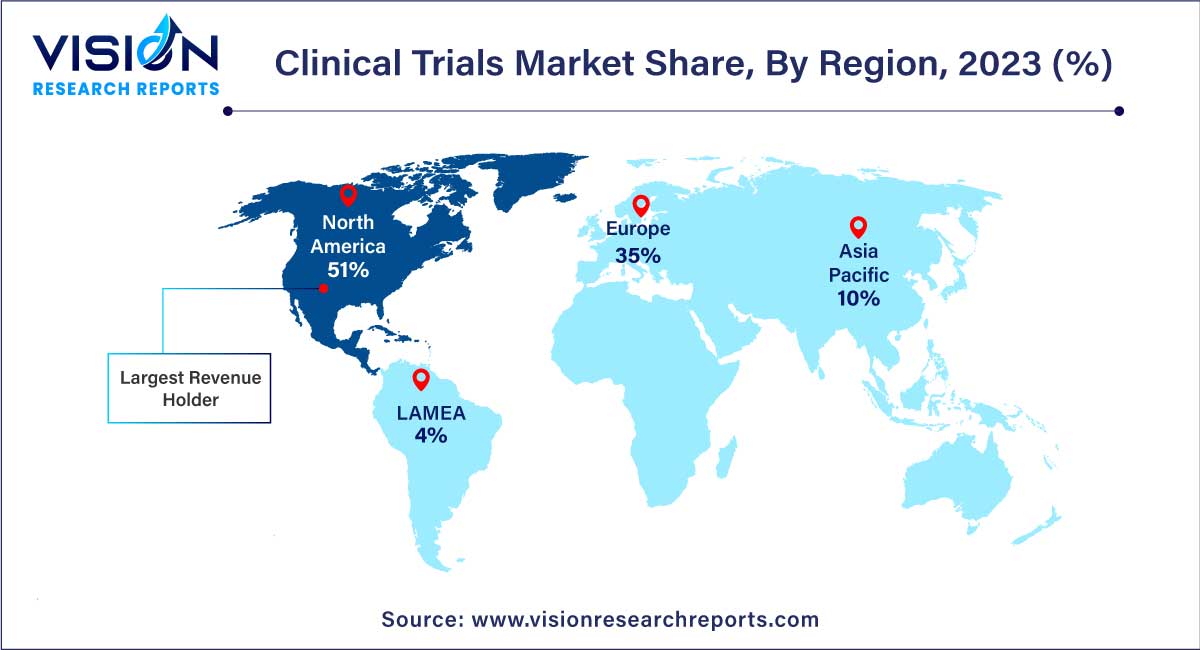
The Asia Pacific region is poised to experience the fastest Compound Annual Growth Rate (CAGR) over the forecast period, propelled by factors such as a large patient pool facilitating easy recruitment. The global impact of the COVID-19 pandemic stands out as a major driving force for the market in this region. Novotech, the largest expertized biotech Contract Research Organization (CRO) in Asia Pacific, has reported an upsurge in demand from biotechnology sponsors for studies due to high quality and quick turnaround. Biotechnology firms are increasingly favoring the Asia Pacific region for conducting COVID-19 trials, leveraging its large patient pool and expeditious procedures.
By Phase
By Study Design
By Indication
By Sponsor
By Service Type
By Region
 Cross-segment Market Size and Analysis for
Mentioned Segments
Cross-segment Market Size and Analysis for
Mentioned Segments
 Additional Company Profiles (Upto 5 With No Cost)
Additional Company Profiles (Upto 5 With No Cost)
 Additional Countries (Apart From Mentioned Countries)
Additional Countries (Apart From Mentioned Countries)
 Country/Region-specific Report
Country/Region-specific Report
 Go To Market Strategy
Go To Market Strategy
 Region Specific Market Dynamics
Region Specific Market Dynamics Region Level Market Share
Region Level Market Share Import Export Analysis
Import Export Analysis Production Analysis
Production Analysis Others
Others