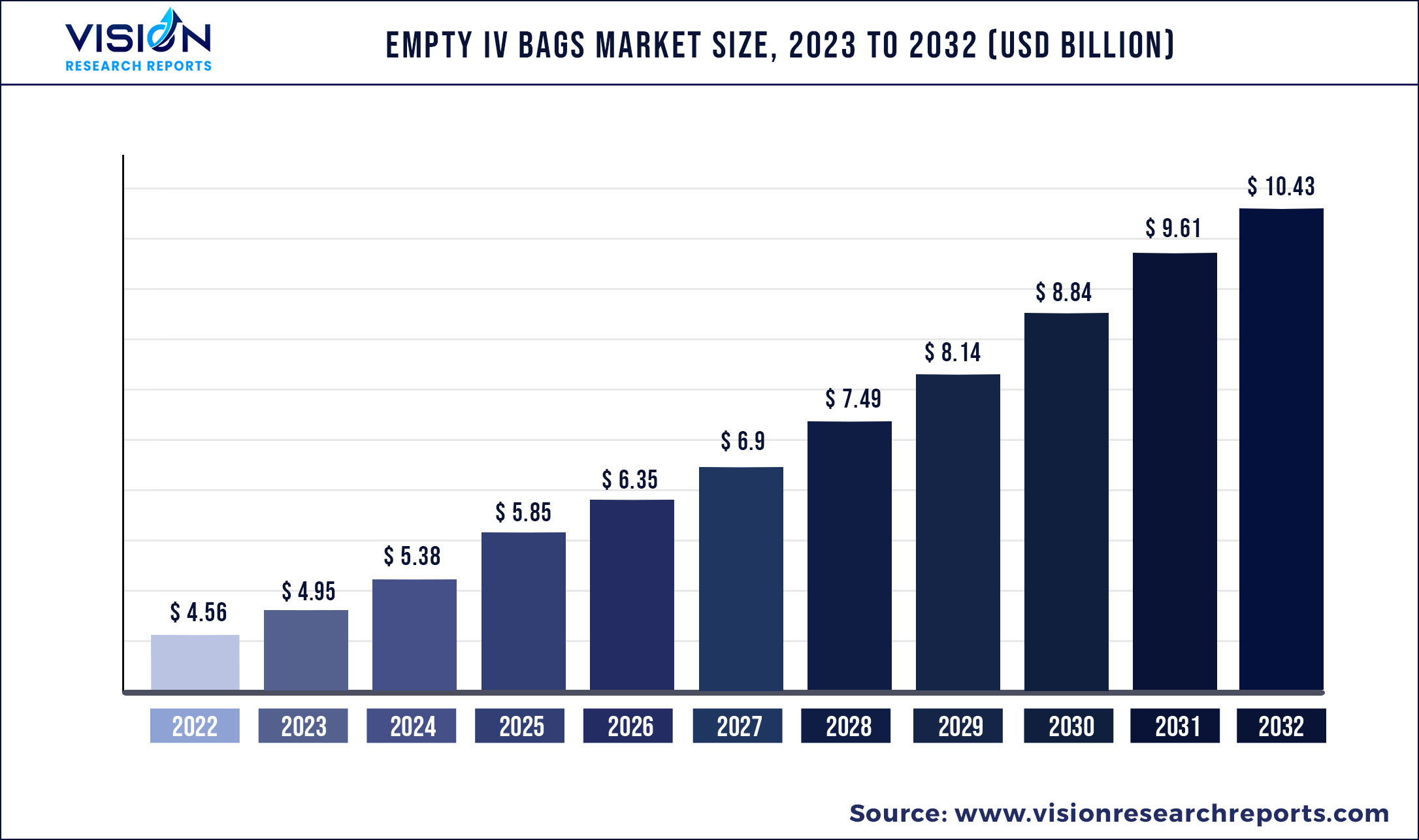
The global empty IV bags market was valued at USD 4.56 billion in 2022 and it is predicted to surpass around USD 10.43 billion by 2032 with a CAGR of 8.63% from 2023 to 2032.
Market Segmentation
Report Scope of the Empty IV Bags Market
| Report Coverage | Details |
| Market Size in 2022 | USD 4.56 billion |
| Revenue Forecast by 2032 | USD 10.43 billion |
| Growth rate from 2023 to 2032 | CAGR of 8.63% |
| Base Year | 2022 |
| Forecast Period | 2023 to 2032 |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
| Companies Covered | Baxter; B. Braun Medical Inc.; ICU MEDICAL, INC.; Wipak; RENOLIT SE; TECHNOFLEX; Sippex IV bags; JW Life science Corp; Fresenius Kabi AG; POLYCINE GmbH; BAUSCH Advanced Technology Group; BD |
The growth of the empty IV bags industry is attributed to technological advancements, the increasing prevalence of chronic conditions, the impact of COVID-19, and the growing risk of malnutrition. The COVID-19 pandemic has created a number of challenges for the healthcare industry worldwide. Restricted access to medical services has led to inadequate medical facilities for patients suffering from various conditions. However, the increase in hospital admission rates due to COVID-19 has led to a rise in the demand for intravenous bags used in IV therapy across all hospital settings globally. Hence, the market is expected to witness significant and continuous growth even after the pandemic ends.
The use of intravenous fluid treatment for COVID-19 patients in need was advised in interim guidelines that were issued by a number of organizations including the WHO, CDC, and NIH. Similarly, recommendations made by the U.K. Joint Anaesthetic and Intensive Care and International Fluid Academy recommended administering fluid therapy to critically ill patients to address their fluid and electrolyte needs.
As a result, these recommendations are helping to drive up the price of empty IV bags during the pandemic. Additionally, for patients who are COVID-19 positive, the European Society for Clinical Nutrition and Metabolism (ESPEN) is advocating the use of enteral and parenteral nutrition to meet their daily nutritional needs. The need for IV fluids is anticipated to increase dramatically as a result of these recommendations from numerous organizations, which will also increase demand for empty intravenous (IV) bags.
Significant research in the medical field has concluded that PVC-made empty intravenous bags are hazardous to human health and the environment due to phthalates leaching from equipment. In addition, the disposal of PVC products through the incineration process results in the production of toxic Hydrogen Chloride (HCL). This further contributes to acid rain.
Hence, companies such as Baxter, Hospira, and B. Braun Melsungen AG are manufacturing IV bags made of PVC-free material, which has thermal stability, moisture-barrier properties, inertness required for IV medications, and are environment friendly. For instance, Hospira launched the VISIV line of PVC/DEHP-free IV bags that have no overwrap, thereby reducing the amount of plastic that has to be discarded.
PVC-free empty IV bags are usually made of Ethylene-Vinyl Acetate (EVA), multilayer polyethylene, or polypropylene, which are compatible with the drug solution and have high seal strength. Pactiv LLC manufactures polypropylene-based bags, known as Propyflex bags, which have won the 2000 Medical Design Excellence Award in a program sponsored by Canon Communications LLC.
In addition, several firms are turning to plasticizer-free biologically inert materials to substitute the use of PVC. In the past five years, B. Braun spent around USD 500.00 million developing PVC- and DEHP-free products. The development and availability of modern materials in different designs are expected to boost growth through the replacement of glass & PVC containers.
Empty IV Bags Market Segmentations:
By Product
Chapter 1. Introduction
1.1.Research Objective
1.2.Scope of the Study
1.3.Definition
Chapter 2. Research Methodology
2.1.Research Approach
2.2.Data Sources
2.3.Assumptions & Limitations
Chapter 3. Executive Summary
3.1.Market Snapshot
Chapter 4. Market Variables and Scope
4.1.Introduction
4.2.Market Classification and Scope
4.3.Industry Value Chain Analysis
4.3.1. Raw Material Procurement Analysis
4.3.2. Sales and Distribution Channel Analysis
4.3.3. Downstream Buyer Analysis
Chapter 5.COVID 19 Impact on Empty IV Bags Market
5.1. COVID-19 Landscape: Empty IV Bags Industry Impact
5.2. COVID 19 - Impact Assessment for the Industry
5.3. COVID 19 Impact: Global Major Government Policy
5.4.Market Trends and Opportunities in the COVID-19 Landscape
Chapter 6. Market Dynamics Analysis and Trends
6.1.Market Dynamics
6.1.1. Market Drivers
6.1.2. Market Restraints
6.1.3. Market Opportunities
6.2.Porter’s Five Forces Analysis
6.2.1. Bargaining power of suppliers
6.2.2. Bargaining power of buyers
6.2.3. Threat of substitute
6.2.4. Threat of new entrants
6.2.5. Degree of competition
Chapter 7. Competitive Landscape
7.1.1. Company Market Share/Positioning Analysis
7.1.2. Key Strategies Adopted by Players
7.1.3. Vendor Landscape
7.1.3.1.List of Suppliers
7.1.3.2.List of Buyers
Chapter 8. Global Empty IV Bags Market, By Product
8.1.Empty IV Bags Market, by Product Type, 2023-2032
8.1.1. PVC
8.1.1.1.Market Revenue and Forecast (2020-2032)
8.1.2. Non-PVC (Material)
8.1.2.1.Market Revenue and Forecast (2020-2032)
8.1.3. Non-PVC (Type)
8.1.3.1.Market Revenue and Forecast (2020-2032)
Chapter 9. Global Empty IV Bags Market, Regional Estimates and Trend Forecast
9.1. North America
9.1.1. Market Revenue and Forecast, by Product (2020-2032)
9.1.2. U.S.
9.1.2.1. Market Revenue and Forecast, by Product (2020-2032)
9.1.3. Rest of North America
9.1.3.1. Market Revenue and Forecast, by Product (2020-2032)
9.2. Europe
9.2.1. Market Revenue and Forecast, by Product (2020-2032)
9.2.2. UK
9.2.2.1. Market Revenue and Forecast, by Product (2020-2032)
9.2.3. Germany
9.2.3.1. Market Revenue and Forecast, by Product (2020-2032)
9.2.4. France
9.2.4.1. Market Revenue and Forecast, by Product (2020-2032)
9.2.5. Rest of Europe
9.2.5.1. Market Revenue and Forecast, by Product (2020-2032)
9.3. APAC
9.3.1. Market Revenue and Forecast, by Product (2020-2032)
9.3.2. India
9.3.2.1. Market Revenue and Forecast, by Product (2020-2032)
9.3.3. China
9.3.3.1. Market Revenue and Forecast, by Product (2020-2032)
9.3.4. Japan
9.3.4.1. Market Revenue and Forecast, by Product (2020-2032)
9.3.5. Rest of APAC
9.3.5.1. Market Revenue and Forecast, by Product (2020-2032)
9.4. MEA
9.4.1. Market Revenue and Forecast, by Product (2020-2032)
9.4.2. GCC
9.4.2.1. Market Revenue and Forecast, by Product (2020-2032)
9.4.3. North Africa
9.4.3.1. Market Revenue and Forecast, by Product (2020-2032)
9.4.4. South Africa
9.4.4.1. Market Revenue and Forecast, by Product (2020-2032)
9.4.5. Rest of MEA
9.4.5.1. Market Revenue and Forecast, by Product (2020-2032)
9.5. Latin America
9.5.1. Market Revenue and Forecast, by Product (2020-2032)
9.5.2. Brazil
9.5.2.1. Market Revenue and Forecast, by Product (2020-2032)
9.5.3. Rest of LATAM
9.5.3.1. Market Revenue and Forecast, by Product (2020-2032)
Chapter 10.Company Profiles
10.1. Baxter
10.1.1.Company Overview
10.1.2.Product Offerings
10.1.3.Financial Performance
10.1.4.Recent Initiatives
10.2. B. Braun Medical Inc.
10.2.1.Company Overview
10.2.2.Product Offerings
10.2.3.Financial Performance
10.2.4.Recent Initiatives
10.3. ICU MEDICAL, INC.
10.3.1.Company Overview
10.3.2.Product Offerings
10.3.3.Financial Performance
10.3.4.Recent Initiatives
10.4. Wipak
10.4.1.Company Overview
10.4.2.Product Offerings
10.4.3.Financial Performance
10.4.4.Recent Initiatives
10.5. RENOLIT SE
10.5.1.Company Overview
10.5.2.Product Offerings
10.5.3.Financial Performance
10.5.4.Recent Initiatives
10.6. TECHNOFLEX
10.6.1.Company Overview
10.6.2.Product Offerings
10.6.3.Financial Performance
10.6.4.Recent Initiatives
10.7. Sippex IV bags
10.7.1.Company Overview
10.7.2.Product Offerings
10.7.3.Financial Performance
10.7.4.Recent Initiatives
10.8. JW Life science Corp
10.8.1.Company Overview
10.8.2.Product Offerings
10.8.3.Financial Performance
10.8.4.Recent Initiatives
10.9. Fresenius Kabi AG
10.9.1.Company Overview
10.9.2.Product Offerings
10.9.3.Financial Performance
10.9.4.Recent Initiatives
10.10. POLYCINE GmbH
10.10.1. Company Overview
10.10.2. Product Offerings
10.10.3. Financial Performance
10.10.4. Recent Initiatives
Chapter 11.Research Methodology
11.1.Primary Research
11.2.Secondary Research
11.3.Assumptions
Chapter 12.Appendix
12.1. About Us
12.2. Glossary of Terms
 Cross-segment Market Size and Analysis for
Mentioned Segments
Cross-segment Market Size and Analysis for
Mentioned Segments
 Additional Company Profiles (Upto 5 With No Cost)
Additional Company Profiles (Upto 5 With No Cost)
 Additional Countries (Apart From Mentioned Countries)
Additional Countries (Apart From Mentioned Countries)
 Country/Region-specific Report
Country/Region-specific Report
 Go To Market Strategy
Go To Market Strategy
 Region Specific Market Dynamics
Region Specific Market Dynamics Region Level Market Share
Region Level Market Share Import Export Analysis
Import Export Analysis Production Analysis
Production Analysis Others
Others