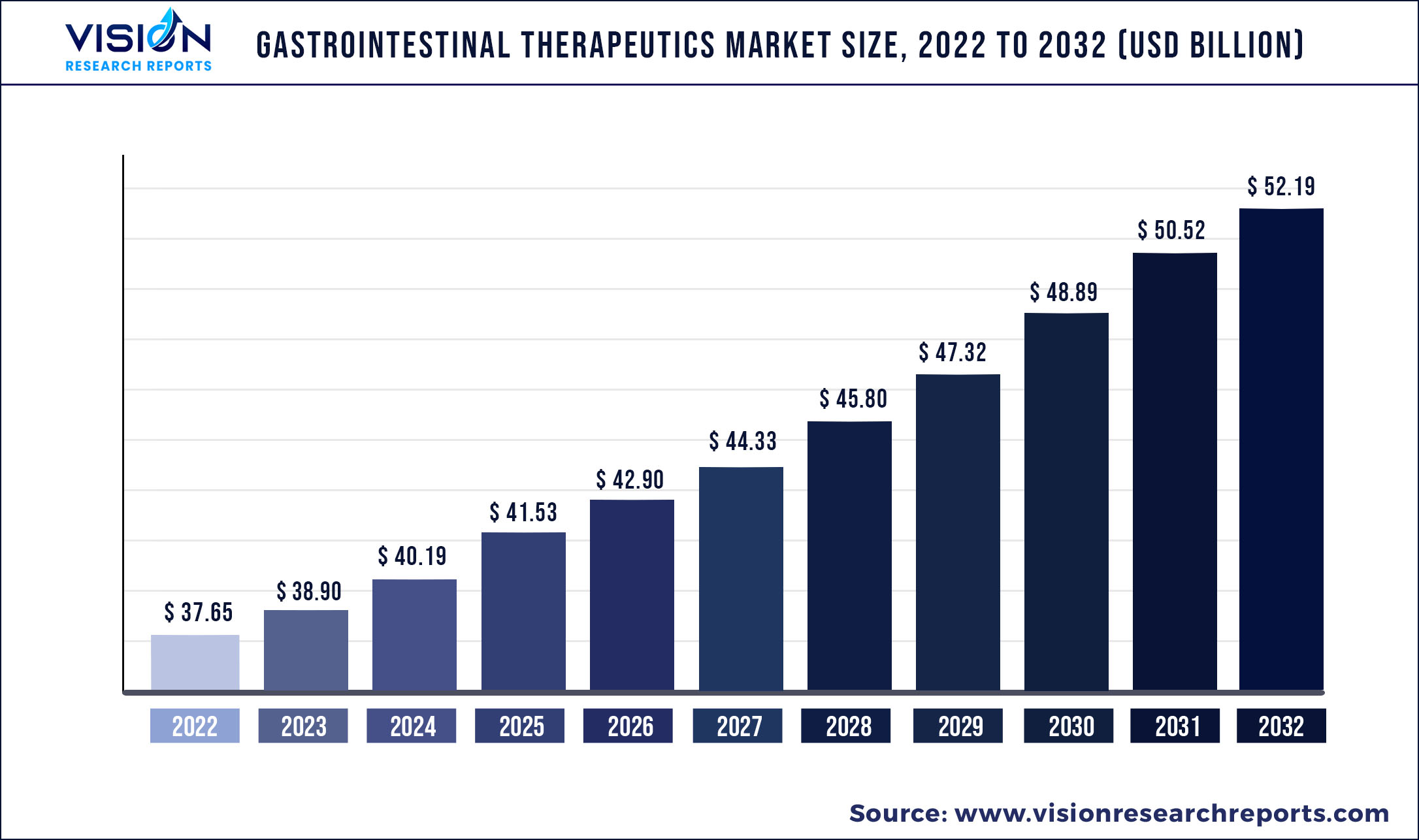
The global gastrointestinal therapeutics market size was estimated at around USD 37.65 billion in 2022 and it is projected to hit around USD 52.19 billion by 2032, growing at a CAGR of 3.32% from 2023 to 2032.
Key Pointers
| Report Coverage | Details |
| Market Size in 2022 | USD 37.65 billion |
| Revenue Forecast by 2032 | USD 52.19 billion |
| Growth rate from 2023 to 2032 | CAGR of 3.32% |
| Base Year | 2022 |
| Forecast Period | 2023 to 2032 |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
| Companies Covered | AbbVie Inc.; AstraZeneca; Janssen Pharmaceuticals NV; Sun Pharmaceutical Industries Ltd.; Takeda Pharmaceutical Company Limited; Salix Pharmaceuticals; Pfizer Inc.; Bayer AG; Abbott; Cipla Inc.; Gilead Sciences, Inc.; Biogen; Organon Group of Companies |
The increasing prevalence of gastrointestinal disorders such as IBS, Crohn’s disease, and ulcerative colitis, and the rising demand for gastrointestinal (GI) drugs are the major factors driving the market growth. Moreover, the surge in R&D efforts by pharmaceutical companies to develop novel GI drugs and increasing product approvals are expected to enhance market growth during the forecast period.
According to the GI Alliance data from February 2021, it is estimated that around 20 million Americans suffer from chronic digestive diseases and over 62 million Americans are diagnosed with gastrointestinal diseases annually, and the incidence of gastrointestinal disorders rises with age. Moreover, as per data published by the International Foundation for Gastrointestinal Disorders, Inc (IFFGD), the prevalence of GERD is 18-28% in North America, 23% in South America, and 9-26% in Europe. Thus, the high prevalence of gastrointestinal diseases is likely to drive market growth in the coming years.
Technological advancements and increasing R&D investments to develop novel therapies such as biologics & targeted drugs for the treatment of gastrointestinal diseases are expected to increase the market growth. A wide number of next-generation therapeutic targets are being investigated, including innovative small molecules and cellular therapy for gastrointestinal disorders. For the treatment of gastrointestinal diseases, the U.S. FDA-approved biological drugs are Amjevita, Cimzia, Entyvio, Inflectra, Remicade, Renflexis, and Tysabri, among others, which are available in the market.
Moreover, the increasing prescription of biological drugs and the presence of strong pipeline drugs such as ustekinumab, risankizumab, and ABBV-154 are expected to boost market growth during the projected period. The increasing regulatory approvals for novel gastrointestinal therapeutic drugs are anticipated to facilitate market expansion in the coming years.
However, high investment requirements and a long approval process for biological drugs are restraining market players’ opportunities to develop innovative therapeutic products. Moreover, patent expirations and stringent regulatory policies are likely to hamper growth. For instance, in April 2020, the U.S. FDA requested the removal of all ranitidine products (Zantac) from the market due to the identification of some contaminants in ranitidine preparation. Moreover, the introduction of biosimilars for Humira has reduced market growth.
Gastrointestinal Therapeutics Market Segmentations:
| By Type | By Drug Class | By Route of Administration | By Application | By Distribution Channel |
|
Branded Generics |
Aminosalicylates Digestive enzymes Proton Pump Inhibitors Laxatives Anti-Emetics H2 Antagonists Anti-Diarrheal Biologics/Biosimilar Others |
Oral Injectable Others |
Crohn's disease Ulcerative colitis GERD IBS Others |
Hospital Pharmacies Retail Pharmacies Online Pharmacies |
Chapter 1. Introduction
1.1. Research Objective
1.2. Scope of the Study
1.3. Definition
Chapter 2. Research Methodology
2.1. Research Approach
2.2. Data Sources
2.3. Assumptions & Limitations
Chapter 3. Executive Summary
3.1. Market Snapshot
Chapter 4. Market Variables and Scope
4.1. Introduction
4.2. Market Classification and Scope
4.3. Industry Value Chain Analysis
4.3.1. Raw Material Procurement Analysis
4.3.2. Sales and Distribution Channel Analysis
4.3.3. Downstream Buyer Analysis
Chapter 5. COVID 19 Impact on Gastrointestinal Therapeutics Market
5.1. COVID-19 Landscape: Gastrointestinal Therapeutics Industry Impact
5.2. COVID 19 - Impact Assessment for the Industry
5.3. COVID 19 Impact: Global Major Government Policy
5.4. Market Trends and Opportunities in the COVID-19 Landscape
Chapter 6. Market Dynamics Analysis and Trends
6.1. Market Dynamics
6.1.1. Market Drivers
6.1.2. Market Restraints
6.1.3. Market Opportunities
6.2. Porter’s Five Forces Analysis
6.2.1. Bargaining power of suppliers
6.2.2. Bargaining power of buyers
6.2.3. Threat of substitute
6.2.4. Threat of new entrants
6.2.5. Degree of competition
Chapter 7. Competitive Landscape
7.1.1. Company Market Share/Positioning Analysis
7.1.2. Key Strategies Adopted by Players
7.1.3. Vendor Landscape
7.1.3.1. List of Suppliers
7.1.3.2. List of Buyers
Chapter 8. Global Gastrointestinal Therapeutics Market, By Type
8.1. Gastrointestinal Therapeutics Market, by Type, 2023-2032
8.1.1. Branded
8.1.1.1. Market Revenue and Forecast (2019-2032)
8.1.2. Generics
8.1.2.1. Market Revenue and Forecast (2019-2032)
Chapter 9. Global Gastrointestinal Therapeutics Market, By Drug Class
9.1. Gastrointestinal Therapeutics Market, by Drug Class, 2023-2032
9.1.1. Aminosalicylates
9.1.1.1. Market Revenue and Forecast (2019-2032)
9.1.2. Digestive enzymes
9.1.2.1. Market Revenue and Forecast (2019-2032)
9.1.3. Proton Pump Inhibitors
9.1.3.1. Market Revenue and Forecast (2019-2032)
9.1.4. Laxatives
9.1.4.1. Market Revenue and Forecast (2019-2032)
9.1.5. Anti-Emetics
9.1.5.1. Market Revenue and Forecast (2019-2032)
9.1.6. H2 Antagonists
9.1.6.1. Market Revenue and Forecast (2019-2032)
9.1.7. Anti-Diarrheal
9.1.7.1. Market Revenue and Forecast (2019-2032)
9.1.8. Biologics/Biosimilar
9.1.8.1. Market Revenue and Forecast (2019-2032)
9.1.9. Others
9.1.9.1. Market Revenue and Forecast (2019-2032)
Chapter 10. Global Gastrointestinal Therapeutics Market, By Route of Administration
10.1. Gastrointestinal Therapeutics Market, by Route of Administration, 2023-2032
10.1.1. Oral
10.1.1.1. Market Revenue and Forecast (2019-2032)
10.1.2. Injectable
10.1.2.1. Market Revenue and Forecast (2019-2032)
10.1.3. Others
10.1.3.1. Market Revenue and Forecast (2019-2032)
Chapter 11. Global Gastrointestinal Therapeutics Market, By Application
11.1. Gastrointestinal Therapeutics Market, by Application, 2023-2032
11.1.1. Crohn's disease
11.1.1.1. Market Revenue and Forecast (2019-2032)
11.1.2. Ulcerative colitis
11.1.2.1. Market Revenue and Forecast (2019-2032)
11.1.3. GERD
11.1.3.1. Market Revenue and Forecast (2019-2032)
11.1.4. IBS
11.1.4.1. Market Revenue and Forecast (2019-2032)
11.1.5. Others
11.1.5.1. Market Revenue and Forecast (2019-2032)
Chapter 12. Global Gastrointestinal Therapeutics Market, By Distribution Channel
12.1. Gastrointestinal Therapeutics Market, by Distribution Channel, 2023-2032
12.1.1. Hospital Pharmacies
12.1.1.1. Market Revenue and Forecast (2019-2032)
12.1.2. Retail Pharmacies
12.1.2.1. Market Revenue and Forecast (2019-2032)
12.1.3. Online Pharmacies
12.1.3.1. Market Revenue and Forecast (2019-2032)
Chapter 13. Global Gastrointestinal Therapeutics Market, Regional Estimates and Trend Forecast
13.1. North America
13.1.1. Market Revenue and Forecast, by Type (2019-2032)
13.1.2. Market Revenue and Forecast, by Drug Class (2019-2032)
13.1.3. Market Revenue and Forecast, by Route of Administration (2019-2032)
13.1.4. Market Revenue and Forecast, by Application (2019-2032)
13.1.5. Market Revenue and Forecast, by Distribution Channel (2019-2032)
13.1.6. U.S.
13.1.6.1. Market Revenue and Forecast, by Type (2019-2032)
13.1.6.2. Market Revenue and Forecast, by Drug Class (2019-2032)
13.1.6.3. Market Revenue and Forecast, by Route of Administration (2019-2032)
13.1.6.4. Market Revenue and Forecast, by Application (2019-2032)
13.1.7. Market Revenue and Forecast, by Distribution Channel (2019-2032)
13.1.8. Rest of North America
13.1.8.1. Market Revenue and Forecast, by Type (2019-2032)
13.1.8.2. Market Revenue and Forecast, by Drug Class (2019-2032)
13.1.8.3. Market Revenue and Forecast, by Route of Administration (2019-2032)
13.1.8.4. Market Revenue and Forecast, by Application (2019-2032)
13.1.8.5. Market Revenue and Forecast, by Distribution Channel (2019-2032)
13.2. Europe
13.2.1. Market Revenue and Forecast, by Type (2019-2032)
13.2.2. Market Revenue and Forecast, by Drug Class (2019-2032)
13.2.3. Market Revenue and Forecast, by Route of Administration (2019-2032)
13.2.4. Market Revenue and Forecast, by Application (2019-2032)
13.2.5. Market Revenue and Forecast, by Distribution Channel (2019-2032)
13.2.6. UK
13.2.6.1. Market Revenue and Forecast, by Type (2019-2032)
13.2.6.2. Market Revenue and Forecast, by Drug Class (2019-2032)
13.2.6.3. Market Revenue and Forecast, by Route of Administration (2019-2032)
13.2.7. Market Revenue and Forecast, by Application (2019-2032)
13.2.8. Market Revenue and Forecast, by Distribution Channel (2019-2032)
13.2.9. Germany
13.2.9.1. Market Revenue and Forecast, by Type (2019-2032)
13.2.9.2. Market Revenue and Forecast, by Drug Class (2019-2032)
13.2.9.3. Market Revenue and Forecast, by Route of Administration (2019-2032)
13.2.10. Market Revenue and Forecast, by Application (2019-2032)
13.2.11. Market Revenue and Forecast, by Distribution Channel (2019-2032)
13.2.12. France
13.2.12.1. Market Revenue and Forecast, by Type (2019-2032)
13.2.12.2. Market Revenue and Forecast, by Drug Class (2019-2032)
13.2.12.3. Market Revenue and Forecast, by Route of Administration (2019-2032)
13.2.12.4. Market Revenue and Forecast, by Application (2019-2032)
13.2.13. Market Revenue and Forecast, by Distribution Channel (2019-2032)
13.2.14. Rest of Europe
13.2.14.1. Market Revenue and Forecast, by Type (2019-2032)
13.2.14.2. Market Revenue and Forecast, by Drug Class (2019-2032)
13.2.14.3. Market Revenue and Forecast, by Route of Administration (2019-2032)
13.2.14.4. Market Revenue and Forecast, by Application (2019-2032)
13.2.15. Market Revenue and Forecast, by Distribution Channel (2019-2032)
13.3. APAC
13.3.1. Market Revenue and Forecast, by Type (2019-2032)
13.3.2. Market Revenue and Forecast, by Drug Class (2019-2032)
13.3.3. Market Revenue and Forecast, by Route of Administration (2019-2032)
13.3.4. Market Revenue and Forecast, by Application (2019-2032)
13.3.5. Market Revenue and Forecast, by Distribution Channel (2019-2032)
13.3.6. India
13.3.6.1. Market Revenue and Forecast, by Type (2019-2032)
13.3.6.2. Market Revenue and Forecast, by Drug Class (2019-2032)
13.3.6.3. Market Revenue and Forecast, by Route of Administration (2019-2032)
13.3.6.4. Market Revenue and Forecast, by Application (2019-2032)
13.3.7. Market Revenue and Forecast, by Distribution Channel (2019-2032)
13.3.8. China
13.3.8.1. Market Revenue and Forecast, by Type (2019-2032)
13.3.8.2. Market Revenue and Forecast, by Drug Class (2019-2032)
13.3.8.3. Market Revenue and Forecast, by Route of Administration (2019-2032)
13.3.8.4. Market Revenue and Forecast, by Application (2019-2032)
13.3.9. Market Revenue and Forecast, by Distribution Channel (2019-2032)
13.3.10. Japan
13.3.10.1. Market Revenue and Forecast, by Type (2019-2032)
13.3.10.2. Market Revenue and Forecast, by Drug Class (2019-2032)
13.3.10.3. Market Revenue and Forecast, by Route of Administration (2019-2032)
13.3.10.4. Market Revenue and Forecast, by Application (2019-2032)
13.3.10.5. Market Revenue and Forecast, by Distribution Channel (2019-2032)
13.3.11. Rest of APAC
13.3.11.1. Market Revenue and Forecast, by Type (2019-2032)
13.3.11.2. Market Revenue and Forecast, by Drug Class (2019-2032)
13.3.11.3. Market Revenue and Forecast, by Route of Administration (2019-2032)
13.3.11.4. Market Revenue and Forecast, by Application (2019-2032)
13.3.11.5. Market Revenue and Forecast, by Distribution Channel (2019-2032)
13.4. MEA
13.4.1. Market Revenue and Forecast, by Type (2019-2032)
13.4.2. Market Revenue and Forecast, by Drug Class (2019-2032)
13.4.3. Market Revenue and Forecast, by Route of Administration (2019-2032)
13.4.4. Market Revenue and Forecast, by Application (2019-2032)
13.4.5. Market Revenue and Forecast, by Distribution Channel (2019-2032)
13.4.6. GCC
13.4.6.1. Market Revenue and Forecast, by Type (2019-2032)
13.4.6.2. Market Revenue and Forecast, by Drug Class (2019-2032)
13.4.6.3. Market Revenue and Forecast, by Route of Administration (2019-2032)
13.4.6.4. Market Revenue and Forecast, by Application (2019-2032)
13.4.7. Market Revenue and Forecast, by Distribution Channel (2019-2032)
13.4.8. North Africa
13.4.8.1. Market Revenue and Forecast, by Type (2019-2032)
13.4.8.2. Market Revenue and Forecast, by Drug Class (2019-2032)
13.4.8.3. Market Revenue and Forecast, by Route of Administration (2019-2032)
13.4.8.4. Market Revenue and Forecast, by Application (2019-2032)
13.4.9. Market Revenue and Forecast, by Distribution Channel (2019-2032)
13.4.10. South Africa
13.4.10.1. Market Revenue and Forecast, by Type (2019-2032)
13.4.10.2. Market Revenue and Forecast, by Drug Class (2019-2032)
13.4.10.3. Market Revenue and Forecast, by Route of Administration (2019-2032)
13.4.10.4. Market Revenue and Forecast, by Application (2019-2032)
13.4.10.5. Market Revenue and Forecast, by Distribution Channel (2019-2032)
13.4.11. Rest of MEA
13.4.11.1. Market Revenue and Forecast, by Type (2019-2032)
13.4.11.2. Market Revenue and Forecast, by Drug Class (2019-2032)
13.4.11.3. Market Revenue and Forecast, by Route of Administration (2019-2032)
13.4.11.4. Market Revenue and Forecast, by Application (2019-2032)
13.4.11.5. Market Revenue and Forecast, by Distribution Channel (2019-2032)
13.5. Latin America
13.5.1. Market Revenue and Forecast, by Type (2019-2032)
13.5.2. Market Revenue and Forecast, by Drug Class (2019-2032)
13.5.3. Market Revenue and Forecast, by Route of Administration (2019-2032)
13.5.4. Market Revenue and Forecast, by Application (2019-2032)
13.5.5. Market Revenue and Forecast, by Distribution Channel (2019-2032)
13.5.6. Brazil
13.5.6.1. Market Revenue and Forecast, by Type (2019-2032)
13.5.6.2. Market Revenue and Forecast, by Drug Class (2019-2032)
13.5.6.3. Market Revenue and Forecast, by Route of Administration (2019-2032)
13.5.6.4. Market Revenue and Forecast, by Application (2019-2032)
13.5.7. Market Revenue and Forecast, by Distribution Channel (2019-2032)
13.5.8. Rest of LATAM
13.5.8.1. Market Revenue and Forecast, by Type (2019-2032)
13.5.8.2. Market Revenue and Forecast, by Drug Class (2019-2032)
13.5.8.3. Market Revenue and Forecast, by Route of Administration (2019-2032)
13.5.8.4. Market Revenue and Forecast, by Application (2019-2032)
13.5.8.5. Market Revenue and Forecast, by Distribution Channel (2019-2032)
Chapter 14. Company Profiles
14.1. AbbVie Inc.
14.1.1. Company Overview
14.1.2. Product Offerings
14.1.3. Financial Performance
14.1.4. Recent Initiatives
14.2. AstraZeneca
14.2.1. Company Overview
14.2.2. Product Offerings
14.2.3. Financial Performance
14.2.4. Recent Initiatives
14.3. Janssen Pharmaceuticals NV
14.3.1. Company Overview
14.3.2. Product Offerings
14.3.3. Financial Performance
14.3.4. Recent Initiatives
14.4. Sun Pharmaceutical Industries Ltd.
14.4.1. Company Overview
14.4.2. Product Offerings
14.4.3. Financial Performance
14.4.4. Recent Initiatives
14.5. Takeda Pharmaceutical Company Limited
14.5.1. Company Overview
14.5.2. Product Offerings
14.5.3. Financial Performance
14.5.4. Recent Initiatives
14.6. Salix Pharmaceuticals
14.6.1. Company Overview
14.6.2. Product Offerings
14.6.3. Financial Performance
14.6.4. Recent Initiatives
14.7. Pfizer Inc.
14.7.1. Company Overview
14.7.2. Product Offerings
14.7.3. Financial Performance
14.7.4. Recent Initiatives
14.8. Bayer AG
14.8.1. Company Overview
14.8.2. Product Offerings
14.8.3. Financial Performance
14.8.4. Recent Initiatives
14.9. Abbott
14.9.1. Company Overview
14.9.2. Product Offerings
14.9.3. Financial Performance
14.9.4. Recent Initiatives
14.10. Cipla Inc.
14.10.1. Company Overview
14.10.2. Product Offerings
14.10.3. Financial Performance
14.10.4. Recent Initiatives
Chapter 15. Research Methodology
15.1. Primary Research
15.2. Secondary Research
15.3. Assumptions
Chapter 16. Appendix
16.1. About Us
16.2. Glossary of Terms
 Cross-segment Market Size and Analysis for
Mentioned Segments
Cross-segment Market Size and Analysis for
Mentioned Segments
 Additional Company Profiles (Upto 5 With No Cost)
Additional Company Profiles (Upto 5 With No Cost)
 Additional Countries (Apart From Mentioned Countries)
Additional Countries (Apart From Mentioned Countries)
 Country/Region-specific Report
Country/Region-specific Report
 Go To Market Strategy
Go To Market Strategy
 Region Specific Market Dynamics
Region Specific Market Dynamics Region Level Market Share
Region Level Market Share Import Export Analysis
Import Export Analysis Production Analysis
Production Analysis Others
Others