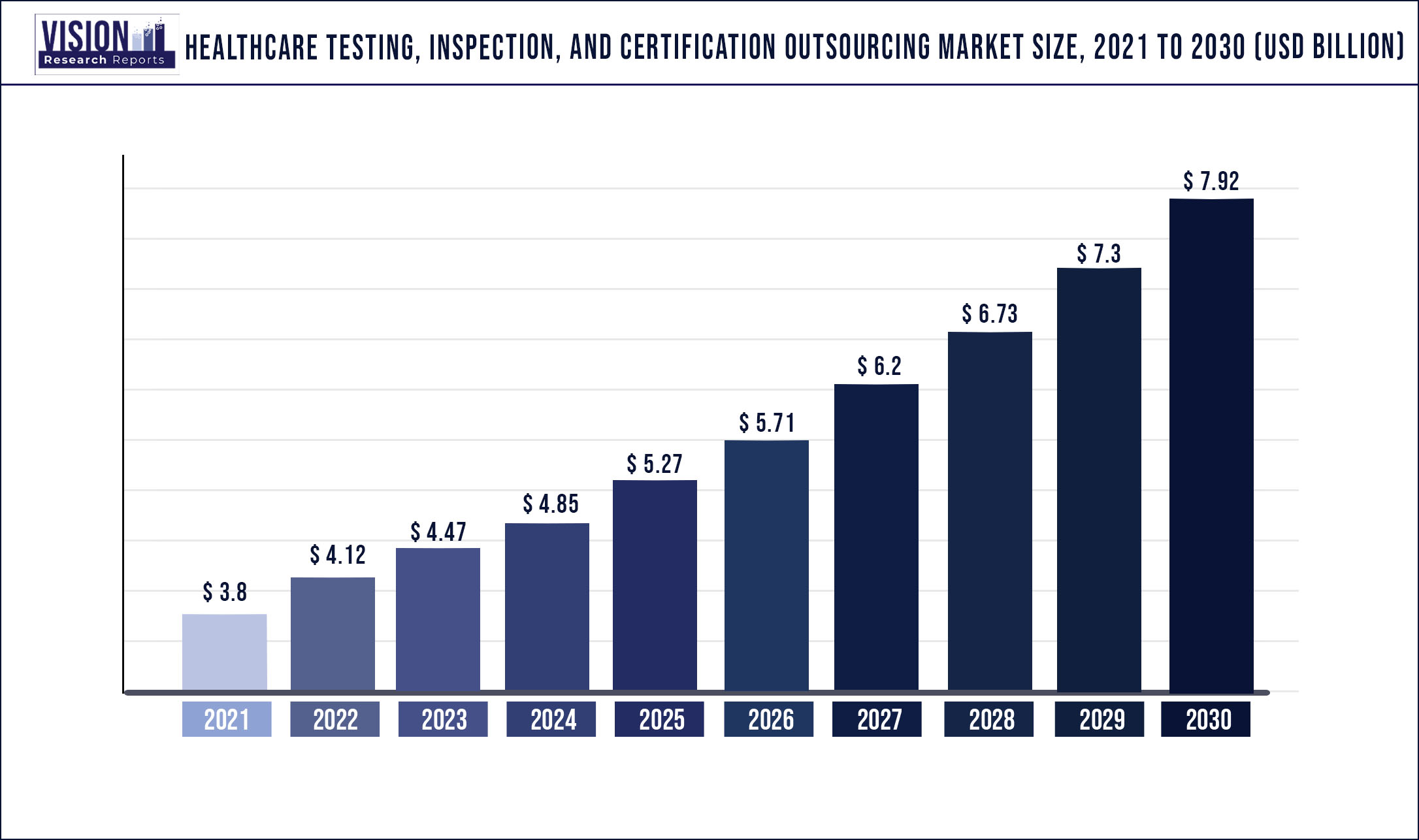
The global healthcare testing, inspection, and certification outsourcing market was estimated at USD 3.8 billion in 2021 and it is expected to surpass around USD 7.92 billion by 2030, poised to grow at a CAGR of 8.5% from 2022 to 2030
Report Highlights
The main drivers of this market are that it helps in improving safety standards of medical devices and pharmaceuticals, there is the rise in strict regulations imposed by the government to make sure that the product is safe, and the increasing use of advanced technology for TIC services.
The COVID-19 pandemic significantly impacted the market. The healthcare testing, inspection, and certification companies are collaborating to reduce the prices to provide safe, quick, and efficient products that will be used to fight the coronavirus. Also, medical devices are producing a lot of opportunities for the healthcare TIC market. For instance, in October 2020, CSA announced the testing, inspection, and certification of PPE kits that are used to fight against COVID-19 in Canada.
As healthcare TIC service providers are capitalizing on the promise of new technologies to revolutionize the industry, frequently by forming partnerships with specialized providers, capital investment is expected to rise even more. Market expansion is being aided by the growth of the middle class in emerging nations, a rise in the need for safety, and rising infrastructure spending. For instance, in December 2021, SGS SA acquired Quay Pharma, a pharma R&D organization, to expand its capability in biopharmaceutical analytical testing.
Scope of The Report
| Report Coverage | Details |
| Market Size in 2021 | USD 3.8 billion |
| Revenue Forecast by 2030 | USD 7.92 billion |
| Growth rate from 2022 to 2030 | CAGR of 8.5% |
| Base Year | 2021 |
| Forecast Period | 2022 to 2030 |
| Segmentation | Services, type, region |
| Companies Covered | SGS SA, Intertek Group plc, Eurofins Scientific, DEKRA CERTIFICATION B.V., UL LLC, Nemko, TÜV SÜD, Element Materials Technology, CSA Group Testing & Certification Inc., ALS, Applus+ |
Services Insights
The services segment is segmented into testing, inspection, and certification. The testing services segment accounted for the largest share of 52.4% in 2021 during the forecast period. The growth of the market is owing to the increasing influence of strict regulations by the government that has led to a rise in verification, validation, and certification. This also helped companies in maintaining the quality parameters and fulfilling the demands of customers, promoting the expansion of testing facilities and equipment by various companies. For instance, in December 2021, Labcorp completely acquired Toxikon Corp. (TC) to expand Labcorp’s development testing capabilities for biotech, pharmaceutical, and medical device clients.
The inspection services are anticipated to register the second fastest CAGR during the forecast period. This is a very important regulatory practice in gaining market approval and checking the safety of the product. Nowadays, inspection services have collaborated along with the prequalification application (laboratory information file, product dossier, and product streams) for promising and organizing inspections.
Type Insights
The medical devices segment dominated the market with a share of 79.3% during the forecast period. This is due to an increase in the manufacturing of medical device equipment that requires TIC services, which will create new opportunities for TIC services. Also, due to COVID-19, there is an increase in the production of medical devices for the prevention and treatment of diseases. For instance, in April 2020, Eurofins announced that its testing facility would support and ensure the safety of medical devices used in COVID-19.
The pharmaceutical segment accounted for the fastest growth rate during the forecast period. TIC services are in high demand because of increased demand for low-cost drugs, increased regulatory pressure, and consumer awareness about pharmaceutical safety. These are some of the driving factors in this market.
Regional Insights
Europe dominated the market with a revenue share of 44.5% in 2021. The high revenue share can be attributed to the majority of healthcare testing, inspection, and certification players in the market. Also, there is a rise in mergers and acquisitions among the market players in this region, leading to the growth of the market. For instance, in February 2022, Eurofins BPT Italy announced an expansion of its biologics testing capacity. The total laboratory space that has increased is 1000 m2 of BSL2 GMP laboratory space.
The Asia-Pacific region is expected to grow at a lucrative rate with a CAGR of 10.08% during the forecast period owing to the rising economic development in countries such as China, India, Japan, and South Korea. This has led to an increase in manufacturing activity in the region, which in turn has created new opportunities for testing, inspection, and certification. For instance, in November 2021, Labcorp expanded its footprint in the Asia-Pacific with the addition of a new integrated bioanalytical laboratory in Singapore.
Key Players
Market Segmentation
Chapter 1. Introduction
1.1. Research Objective
1.2. Scope of the Study
1.3. Definition
Chapter 2. Research Methodology
2.1. Research Approach
2.2. Data Sources
2.3. Assumptions & Limitations
Chapter 3. Executive Summary
3.1. Market Snapshot
Chapter 4. Market Variables and Scope
4.1. Introduction
4.2. Market Classification and Scope
4.3. Industry Value Chain Analysis
4.3.1. Raw Material Procurement Analysis
4.3.2. Sales and Distribution Channel Analysis
4.3.3. Downstream Buyer Analysis
Chapter 5. COVID 19 Impact on Healthcare Testing, Inspection, And Certification Outsourcing Market
5.1. COVID-19 Landscape: Healthcare Testing, Inspection, And Certification Outsourcing Industry Impact
5.2. COVID 19 - Impact Assessment for the Industry
5.3. COVID 19 Impact: Global Major Government Policy
5.4. Market Trends and Opportunities in the COVID-19 Landscape
Chapter 6. Market Dynamics Analysis and Trends
6.1. Market Dynamics
6.1.1. Market Drivers
6.1.2. Market Restraints
6.1.3. Market Opportunities
6.2. Porter’s Five Forces Analysis
6.2.1. Bargaining power of suppliers
6.2.2. Bargaining power of buyers
6.2.3. Threat of substitute
6.2.4. Threat of new entrants
6.2.5. Degree of competition
Chapter 7. Competitive Landscape
7.1.1. Company Market Share/Positioning Analysis
7.1.2. Key Strategies Adopted by Players
7.1.3. Vendor Landscape
7.1.3.1. List of Suppliers
7.1.3.2. List of Buyers
Chapter 8. Global Healthcare Testing, Inspection, And Certification Outsourcing Market, By Services
8.1. Healthcare Testing, Inspection, And Certification Outsourcing Market, by Services, 2022-2030
8.1.1. Testing
8.1.1.1. Market Revenue and Forecast (2017-2030)
8.1.2. Inspection
8.1.2.1. Market Revenue and Forecast (2017-2030)
8.1.3. Certification
8.1.3.1. Market Revenue and Forecast (2017-2030)
Chapter 9. Global Healthcare Testing, Inspection, And Certification Outsourcing Market, By Type
9.1. Healthcare Testing, Inspection, And Certification Outsourcing Market, by Type, 2022-2030
9.1.1. Medical Devices
9.1.1.1. Market Revenue and Forecast (2017-2030)
9.1.2. Pharmaceutical
9.1.2.1. Market Revenue and Forecast (2017-2030)
Chapter 10. Global Healthcare Testing, Inspection, And Certification Outsourcing Market, Regional Estimates and Trend Forecast
10.1. North America
10.1.1. Market Revenue and Forecast, by Services (2017-2030)
10.1.2. Market Revenue and Forecast, by Type (2017-2030)
10.1.3. U.S.
10.1.3.1. Market Revenue and Forecast, by Services (2017-2030)
10.1.3.2. Market Revenue and Forecast, by Type (2017-2030)
10.1.4. Rest of North America
10.1.4.1. Market Revenue and Forecast, by Services (2017-2030)
10.1.4.2. Market Revenue and Forecast, by Type (2017-2030)
10.2. Europe
10.2.1. Market Revenue and Forecast, by Services (2017-2030)
10.2.2. Market Revenue and Forecast, by Type (2017-2030)
10.2.3. UK
10.2.3.1. Market Revenue and Forecast, by Services (2017-2030)
10.2.3.2. Market Revenue and Forecast, by Type (2017-2030)
10.2.4. Germany
10.2.4.1. Market Revenue and Forecast, by Services (2017-2030)
10.2.4.2. Market Revenue and Forecast, by Type (2017-2030)
10.2.5. France
10.2.5.1. Market Revenue and Forecast, by Services (2017-2030)
10.2.5.2. Market Revenue and Forecast, by Type (2017-2030)
10.2.6. Rest of Europe
10.2.6.1. Market Revenue and Forecast, by Services (2017-2030)
10.2.6.2. Market Revenue and Forecast, by Type (2017-2030)
10.3. APAC
10.3.1. Market Revenue and Forecast, by Services (2017-2030)
10.3.2. Market Revenue and Forecast, by Type (2017-2030)
10.3.3. India
10.3.3.1. Market Revenue and Forecast, by Services (2017-2030)
10.3.3.2. Market Revenue and Forecast, by Type (2017-2030)
10.3.4. China
10.3.4.1. Market Revenue and Forecast, by Services (2017-2030)
10.3.4.2. Market Revenue and Forecast, by Type (2017-2030)
10.3.5. Japan
10.3.5.1. Market Revenue and Forecast, by Services (2017-2030)
10.3.5.2. Market Revenue and Forecast, by Type (2017-2030)
10.3.6. Rest of APAC
10.3.6.1. Market Revenue and Forecast, by Services (2017-2030)
10.3.6.2. Market Revenue and Forecast, by Type (2017-2030)
10.4. MEA
10.4.1. Market Revenue and Forecast, by Services (2017-2030)
10.4.2. Market Revenue and Forecast, by Type (2017-2030)
10.4.3. GCC
10.4.3.1. Market Revenue and Forecast, by Services (2017-2030)
10.4.3.2. Market Revenue and Forecast, by Type (2017-2030)
10.4.4. North Africa
10.4.4.1. Market Revenue and Forecast, by Services (2017-2030)
10.4.4.2. Market Revenue and Forecast, by Type (2017-2030)
10.4.5. South Africa
10.4.5.1. Market Revenue and Forecast, by Services (2017-2030)
10.4.5.2. Market Revenue and Forecast, by Type (2017-2030)
10.4.6. Rest of MEA
10.4.6.1. Market Revenue and Forecast, by Services (2017-2030)
10.4.6.2. Market Revenue and Forecast, by Type (2017-2030)
10.5. Latin America
10.5.1. Market Revenue and Forecast, by Services (2017-2030)
10.5.2. Market Revenue and Forecast, by Type (2017-2030)
10.5.3. Brazil
10.5.3.1. Market Revenue and Forecast, by Services (2017-2030)
10.5.3.2. Market Revenue and Forecast, by Type (2017-2030)
10.5.4. Rest of LATAM
10.5.4.1. Market Revenue and Forecast, by Services (2017-2030)
10.5.4.2. Market Revenue and Forecast, by Type (2017-2030)
Chapter 11. Company Profiles
11.1. SGS SA
11.1.1. Company Overview
11.1.2. Product Offerings
11.1.3. Financial Performance
11.1.4. Recent Initiatives
11.2. Intertek Group plc
11.2.1. Company Overview
11.2.2. Product Offerings
11.2.3. Financial Performance
11.2.4. Recent Initiatives
11.3. Eurofins Scientific
11.3.1. Company Overview
11.3.2. Product Offerings
11.3.3. Financial Performance
11.3.4. Recent Initiatives
11.4. DEKRA CERTIFICATION B.V.
11.4.1. Company Overview
11.4.2. Product Offerings
11.4.3. Financial Performance
11.4.4. LTE Scientific
11.5. UL LLC
11.5.1. Company Overview
11.5.2. Product Offerings
11.5.3. Financial Performance
11.5.4. Recent Initiatives
11.6. Nemko
11.6.1. Company Overview
11.6.2. Product Offerings
11.6.3. Financial Performance
11.6.4. Recent Initiatives
11.7. TÜV SÜD
11.7.1. Company Overview
11.7.2. Product Offerings
11.7.3. Financial Performance
11.7.4. Recent Initiatives
11.8. Element Materials Technology
11.8.1. Company Overview
11.8.2. Product Offerings
11.8.3. Financial Performance
11.8.4. Recent Initiatives
11.9. CSA Group Testing & Certification Inc.
11.9.1. Company Overview
11.9.2. Product Offerings
11.9.3. Financial Performance
11.9.4. Recent Initiatives
11.10. ALS
11.10.1. Company Overview
11.10.2. Product Offerings
11.10.3. Financial Performance
11.10.4. Recent Initiatives
Chapter 12. Research Methodology
12.1. Primary Research
12.2. Secondary Research
12.3. Assumptions
Chapter 13. Appendix
13.1. About Us
13.2. Glossary of Terms
 Cross-segment Market Size and Analysis for
Mentioned Segments
Cross-segment Market Size and Analysis for
Mentioned Segments
 Additional Company Profiles (Upto 5 With No Cost)
Additional Company Profiles (Upto 5 With No Cost)
 Additional Countries (Apart From Mentioned Countries)
Additional Countries (Apart From Mentioned Countries)
 Country/Region-specific Report
Country/Region-specific Report
 Go To Market Strategy
Go To Market Strategy
 Region Specific Market Dynamics
Region Specific Market Dynamics Region Level Market Share
Region Level Market Share Import Export Analysis
Import Export Analysis Production Analysis
Production Analysis Others
Others