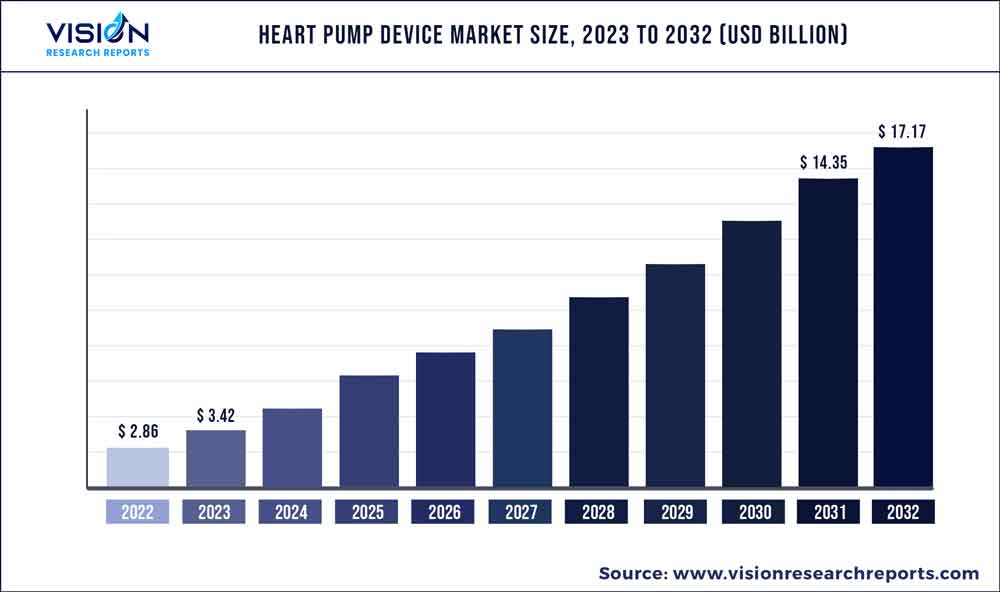
The global heart pump device market was surpassed at USD 2.86 billion in 2022 and is expected to hit around USD 17.17 billion by 2032, growing at a CAGR of 19.63% from 2023 to 2032. The heart pump device market in the United States was accounted for USD 1.2 billion in 2022.
Key Pointers
Report Scope of the Heart Pump Device Market
| Report Coverage | Details |
| Revenue Share of North America in 2022 | 48% |
| Revenue Forecast by 2032 | USD 17.17 billion |
| Growth Rate from 2023 to 2032 | CAGR of 19.63% |
| Base Year | 2022 |
| Forecast Period | 2023 to 2032 |
| Market Analysis (Terms Used) | Value (US$ Million/Billion) or (Volume/Units) |
| Companies Covered | Abbott; ABIOMED; CorWave SA; LivaNova PLC; Berlin Heart; SynCardia Systems, LLC; Jarvik Heart; BiVACOR Inc.; Leviticus Cardio; Evaheart, Inc.; Teleflex Incorporated; Getinge AB.; CARMAT; Fresenius SE & Co. KgaA |
The growing number of individuals suffering from cardiovascular diseases (CVD), and the growing geriatric population are driving the demand for heart pump devices. However, as the pandemic progressed, there was an increased demand for heart pump products due to the higher risk of severe illness in patients with pre-existing heart conditions who contract COVID-19. This led to an increase in the adoption of these devices as a treatment option for HF patients who were also diagnosed with COVID-19.
The COVID-19 pandemic had a significant impact on the market. During the pandemic, there was a decrease in patients seeking medical attention for heart failure (HF) due to fear of exposure to the virus in hospitals and clinics. This led to a decline in the revenue of key market players during the initial phase of the pandemic.
According to the Chinese Center for Disease Control and Prevention, the case-fatality rate for COVID-19 was found to be 11% for patients with CVD, compared to the overall rate of 2.3%. As the prevalence of HF and the aging population increase, the demand for heart pumps is expected to increase over the forecast period.
The increasing prevalence of CVD and associated conditions are expected to drive market growth over the forecast period. According to an article by NCBI in May 2022, 92.1 million Americans are affected by some form of CVD, and it is predicted that 43.9% will have some type of CVD by 2030. The rising number of end-stage HF patients, coupled with the shortage of donor organs, has fueled a surge in demand for heart pump devices. The growing incidence of HF is driving market growth.
The rising investment in R&D for heart pump products is expected to drive market growth by promoting increased adoption and usage of these devices. For example, BiVACOR received a grant of USD 750,000 from the Australian government through the Targeted Translation Research Accelerator (TTRA) and Medical Research Future Fund (MRFF) program in October 2022. This initiative aims to enhance patient experience and improve quality of life while at home, thereby driving growth in the market.
Type Insights
Based on type, the implanted heart pump devices segment accounted for the largest share of 76% of the market in 2022. Implanted heart pump devices, also known as VADs, are becoming popular for patients suffering from HF who are not eligible for a heart transplant. These products are surgically implanted into the chest and designed to take over the heart's pumping function by continuously pumping blood from the ventricles to the rest of the body. Furthermore, advancements in technology and innovation in implantable heart pump products are expected to drive this segment's growth further. Manufacturers are developing smaller, more efficient products that require less invasive procedures, which are expected to increase the adoption of VADs as a treatment option for HF patients.
The extracorporeal heart pump devices segment is also expected to grow significantly over the forecast period. Extracorporeal heart pump products are external devices that temporarily support the heart by pumping blood outside the body and returning it to the circulatory system. They effectively treat acute HF and are used as a bridge to transplantation. The rising prevalence of heart disease and the shortage of donor hearts have driven the demand for extracorporeal products.
Product Insights
Based on product type, the ventricular assist devices (VADs) segment accounted for the largest share of the heart pump devices market in 2022. It is estimated to register the fastest CAGR of 19.82% during the forecast period. VADs are mechanical devices that assist with pumping blood from the heart's ventricles to the rest of the body. They are typically used as a temporary solution while awaiting a heart transplant but are also increasingly being used as a long-term treatment option for HF patients who are not candidates for transplantation.
Furthermore, market players' introduction of new and advanced VADs is another factor driving the segment's growth. These products are designed to be more effective, user-friendly, and affordable, which is expected to increase the demand for VADs even further. For instance, in August 2022, Abbott Laboratories disclosed data revealing that patients with advanced heart failure who received its HeartMate 3 LVAD had higher survival rates after five years than those who received the older HeartMate II LVAD. The HeartMate 3 demonstrated a 58% survival rate over the five years, compared to the HeartMate II's 44% survival rate. This is mainly due to decreased deaths related to stroke, clotting, and bleeding associated with the newer device.
End-use Insights
Based on end-use, the hospital segment accounted for the largest share of 82% in 2022. This growth can be attributed to the increasing number of surgical procedures being performed globally due to the rising incidence of cardiovascular diseases and the availability of skilled professionals and novel heart pump products.
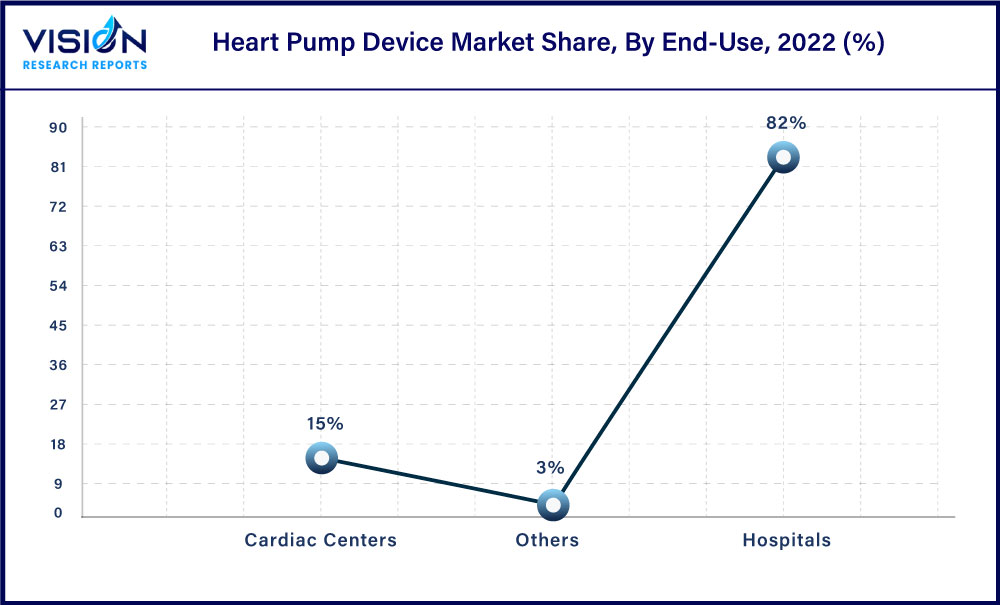
The cardiac center’s segment is also expected to grow significantly over the forecast period owing to the increasing preference of individuals to undergo surgical interventions in cardiac centers. Moreover, the increasing prevalence of cardiac disease is driving demand for specialized care, and cardiac centers are well-positioned to meet this demand.
Regional Insights
In 2022, North America was anticipated to be the dominant region, with a revenue share of over 48%. This is mainly due to the high incidences of heart disease in the region, a large aging population, and strong healthcare infrastructure. These key factors are expected to fuel regional growth. In addition, according to the CDC, the prevalence of atrial fibrillation is estimated to rise to 12.1 million by the year 2030. This highlights a rising burden of CVD in the country, which is expected to increase the demand for heart pump devices during the forecast period.
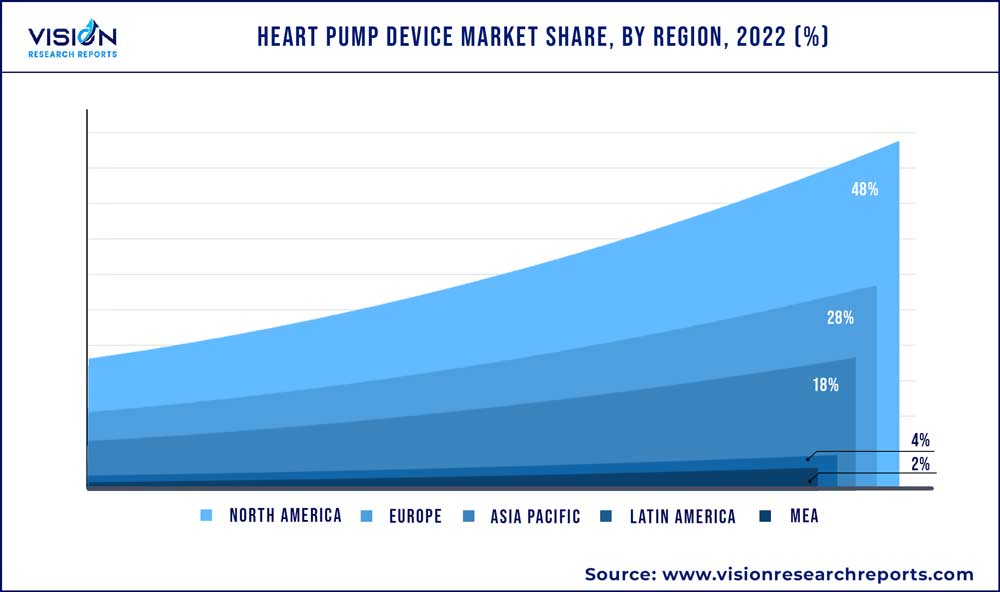
Europe is estimated to grow at the fastest rate during the forecast period. The growth is attributed to the aging population, high prevalence of cardiovascular diseases, and advanced healthcare infrastructure. In addition, favorable government policies, increased awareness of heart pump devices, and significant investment in research and development contribute to the market's expansion, leading to wider adoption of these life-saving products in the European region.
Heart Pump Device Market Segmentations:
By Type
By Product
By End-Use
By Regional
Chapter 1. Introduction
1.1. Research Objective
1.2. Scope of the Study
1.3. Definition
Chapter 2. Research Methodology
2.1. Research Approach
2.2. Data Sources
2.3. Assumptions & Limitations
Chapter 3. Executive Summary
3.1. Market Snapshot
Chapter 4. Market Variables and Scope
4.1. Introduction
4.2. Market Classification and Scope
4.3. Industry Value Chain Analysis
4.3.1. Raw Material Procurement Analysis
4.3.2. Sales and Distribution Type Analysis
4.3.3. Downstream Buyer Analysis
Chapter 5. COVID 19 Impact on Heart Pump Device Market
5.1. COVID-19 Landscape: Heart Pump Device Industry Impact
5.2. COVID 19 - Impact Assessment for the Industry
5.3. COVID 19 Impact: Global Major Government Policy
5.4. Market Trends and Opportunities in the COVID-19 Landscape
Chapter 6. Market Dynamics Analysis and Trends
6.1. Market Dynamics
6.1.1. Market Drivers
6.1.2. Market Restraints
6.1.3. Market Opportunities
6.2. Porter’s Five Forces Analysis
6.2.1. Bargaining power of suppliers
6.2.2. Bargaining power of buyers
6.2.3. Threat of substitute
6.2.4. Threat of new entrants
6.2.5. Degree of competition
Chapter 7. Competitive Landscape
7.1.1. Company Market Share/Positioning Analysis
7.1.2. Key Strategies Adopted by Players
7.1.3. Vendor Landscape
7.1.3.1. List of Suppliers
7.1.3.2. List of Buyers
Chapter 8. Global Heart Pump Device Market, By Type
8.1. Heart Pump Device Market, by Type, 2023-2032
8.1.1 Implanted Heart Pump Devices
8.1.1.1. Market Revenue and Forecast (2020-2032)
8.1.2. Extracorporeal Heart Pump Devices
8.1.2.1. Market Revenue and Forecast (2020-2032)
Chapter 9. Global Heart Pump Device Market, By Product
9.1. Heart Pump Device Market, by Product, 2023-2032
9.1.1. Ventricular Assist Devices (VADs)
9.1.1.1. Market Revenue and Forecast (2020-2032)
9.1.2. Total Artificial Hearts (TAHs)
9.1.2.1. Market Revenue and Forecast (2020-2032)
9.1.3. Intra-Aortic Balloon Pumps (IABPs)
9.1.3.1. Market Revenue and Forecast (2020-2032)
9.1.4. Extracorporeal Membrane Oxygenation (ECMO)
9.1.4.1. Market Revenue and Forecast (2020-2032)
Chapter 10. Global Heart Pump Device Market, By End-Use
10.1. Heart Pump Device Market, by End-Use, 2023-2032
10.1.1. Hospitals
10.1.1.1. Market Revenue and Forecast (2020-2032)
10.1.2. Cardiac Centers
10.1.2.1. Market Revenue and Forecast (2020-2032)
10.1.3. Others
10.1.3.1. Market Revenue and Forecast (2020-2032)
Chapter 11. Global Heart Pump Device Market, Regional Estimates and Trend Forecast
11.1. North America
11.1.1. Market Revenue and Forecast, by Type (2020-2032)
11.1.2. Market Revenue and Forecast, by Product (2020-2032)
11.1.3. Market Revenue and Forecast, by End-Use (2020-2032)
11.1.4. U.S.
11.1.4.1. Market Revenue and Forecast, by Type (2020-2032)
11.1.4.2. Market Revenue and Forecast, by Product (2020-2032)
11.1.4.3. Market Revenue and Forecast, by End-Use (2020-2032)
11.1.5. Rest of North America
11.1.5.1. Market Revenue and Forecast, by Type (2020-2032)
11.1.5.2. Market Revenue and Forecast, by Product (2020-2032)
11.1.5.3. Market Revenue and Forecast, by End-Use (2020-2032)
11.2. Europe
11.2.1. Market Revenue and Forecast, by Type (2020-2032)
11.2.2. Market Revenue and Forecast, by Product (2020-2032)
11.2.3. Market Revenue and Forecast, by End-Use (2020-2032)
11.2.4. UK
11.2.4.1. Market Revenue and Forecast, by Type (2020-2032)
11.2.4.2. Market Revenue and Forecast, by Product (2020-2032)
11.2.4.3. Market Revenue and Forecast, by End-Use (2020-2032)
11.2.5. Germany
11.2.5.1. Market Revenue and Forecast, by Type (2020-2032)
11.2.5.2. Market Revenue and Forecast, by Product (2020-2032)
11.2.5.3. Market Revenue and Forecast, by End-Use (2020-2032)
11.2.6. France
11.2.6.1. Market Revenue and Forecast, by Type (2020-2032)
11.2.6.2. Market Revenue and Forecast, by Product (2020-2032)
11.2.6.3. Market Revenue and Forecast, by End-Use (2020-2032)
11.2.7. Rest of Europe
11.2.7.1. Market Revenue and Forecast, by Type (2020-2032)
11.2.7.2. Market Revenue and Forecast, by Product (2020-2032)
11.2.7.3. Market Revenue and Forecast, by End-Use (2020-2032)
11.3. APAC
11.3.1. Market Revenue and Forecast, by Type (2020-2032)
11.3.2. Market Revenue and Forecast, by Product (2020-2032)
11.3.3. Market Revenue and Forecast, by End-Use (2020-2032)
11.3.4. India
11.3.4.1. Market Revenue and Forecast, by Type (2020-2032)
11.3.4.2. Market Revenue and Forecast, by Product (2020-2032)
11.3.4.3. Market Revenue and Forecast, by End-Use (2020-2032)
11.3.5. China
11.3.5.1. Market Revenue and Forecast, by Type (2020-2032)
11.3.5.2. Market Revenue and Forecast, by Product (2020-2032)
11.3.5.3. Market Revenue and Forecast, by End-Use (2020-2032)
11.3.6. Japan
11.3.6.1. Market Revenue and Forecast, by Type (2020-2032)
11.3.6.2. Market Revenue and Forecast, by Product (2020-2032)
11.3.6.3. Market Revenue and Forecast, by End-Use (2020-2032)
11.3.7. Rest of APAC
11.3.7.1. Market Revenue and Forecast, by Type (2020-2032)
11.3.7.2. Market Revenue and Forecast, by Product (2020-2032)
11.3.7.3. Market Revenue and Forecast, by End-Use (2020-2032)
11.4. MEA
11.4.1. Market Revenue and Forecast, by Type (2020-2032)
11.4.2. Market Revenue and Forecast, by Product (2020-2032)
11.4.3. Market Revenue and Forecast, by End-Use (2020-2032)
11.4.4. GCC
11.4.4.1. Market Revenue and Forecast, by Type (2020-2032)
11.4.4.2. Market Revenue and Forecast, by Product (2020-2032)
11.4.4.3. Market Revenue and Forecast, by End-Use (2020-2032)
11.4.5. North Africa
11.4.5.1. Market Revenue and Forecast, by Type (2020-2032)
11.4.5.2. Market Revenue and Forecast, by Product (2020-2032)
11.4.5.3. Market Revenue and Forecast, by End-Use (2020-2032)
11.4.6. South Africa
11.4.6.1. Market Revenue and Forecast, by Type (2020-2032)
11.4.6.2. Market Revenue and Forecast, by Product (2020-2032)
11.4.6.3. Market Revenue and Forecast, by End-Use (2020-2032)
11.4.7. Rest of MEA
11.4.7.1. Market Revenue and Forecast, by Type (2020-2032)
11.4.7.2. Market Revenue and Forecast, by Product (2020-2032)
11.4.7.3. Market Revenue and Forecast, by End-Use (2020-2032)
11.5. Latin America
11.5.1. Market Revenue and Forecast, by Type (2020-2032)
11.5.2. Market Revenue and Forecast, by Product (2020-2032)
11.5.3. Market Revenue and Forecast, by End-Use (2020-2032)
11.5.4. Brazil
11.5.4.1. Market Revenue and Forecast, by Type (2020-2032)
11.5.4.2. Market Revenue and Forecast, by Product (2020-2032)
11.5.4.3. Market Revenue and Forecast, by End-Use (2020-2032)
11.5.5. Rest of LATAM
11.5.5.1. Market Revenue and Forecast, by Type (2020-2032)
11.5.5.2. Market Revenue and Forecast, by Product (2020-2032)
11.5.5.3. Market Revenue and Forecast, by End-Use (2020-2032)
Chapter 12. Company Profiles
12.1. Abbott
12.1.1. Company Overview
12.1.2. Product Offerings
12.1.3. Financial Performance
12.1.4. Recent Initiatives
12.2. ABIOMED
12.2.1. Company Overview
12.2.2. Product Offerings
12.2.3. Financial Performance
12.2.4. Recent Initiatives
12.3. CorWave SA
12.3.1. Company Overview
12.3.2. Product Offerings
12.3.3. Financial Performance
12.3.4. Recent Initiatives
12.4. LivaNova PLC
12.4.1. Company Overview
12.4.2. Product Offerings
12.4.3. Financial Performance
12.4.4. Recent Initiatives
12.5. Berlin Heart
12.5.1. Company Overview
12.5.2. Product Offerings
12.5.3. Financial Performance
12.5.4. Recent Initiatives
12.6. SynCardia Systems, LLC
12.6.1. Company Overview
12.6.2. Product Offerings
12.6.3. Financial Performance
12.6.4. Recent Initiatives
12.7. Jarvik Heart
12.7.1. Company Overview
12.7.2. Product Offerings
12.7.3. Financial Performance
12.7.4. Recent Initiatives
12.8. BiVACOR Inc.
12.8.1. Company Overview
12.8.2. Product Offerings
12.8.3. Financial Performance
12.8.4. Recent Initiatives
12.9. Leviticus Cardio
12.9.1. Company Overview
12.9.2. Product Offerings
12.9.3. Financial Performance
12.9.4. Recent Initiatives
12.10. Evaheart, Inc.
12.10.1. Company Overview
12.10.2. Product Offerings
12.10.3. Financial Performance
12.10.4. Recent Initiatives
Chapter 13. Research Methodology
13.1. Primary Research
13.2. Secondary Research
13.3. Assumptions
Chapter 14. Appendix
14.1. About Us
14.2. Glossary of Terms
 Cross-segment Market Size and Analysis for
Mentioned Segments
Cross-segment Market Size and Analysis for
Mentioned Segments
 Additional Company Profiles (Upto 5 With No Cost)
Additional Company Profiles (Upto 5 With No Cost)
 Additional Countries (Apart From Mentioned Countries)
Additional Countries (Apart From Mentioned Countries)
 Country/Region-specific Report
Country/Region-specific Report
 Go To Market Strategy
Go To Market Strategy
 Region Specific Market Dynamics
Region Specific Market Dynamics Region Level Market Share
Region Level Market Share Import Export Analysis
Import Export Analysis Production Analysis
Production Analysis Others
Others