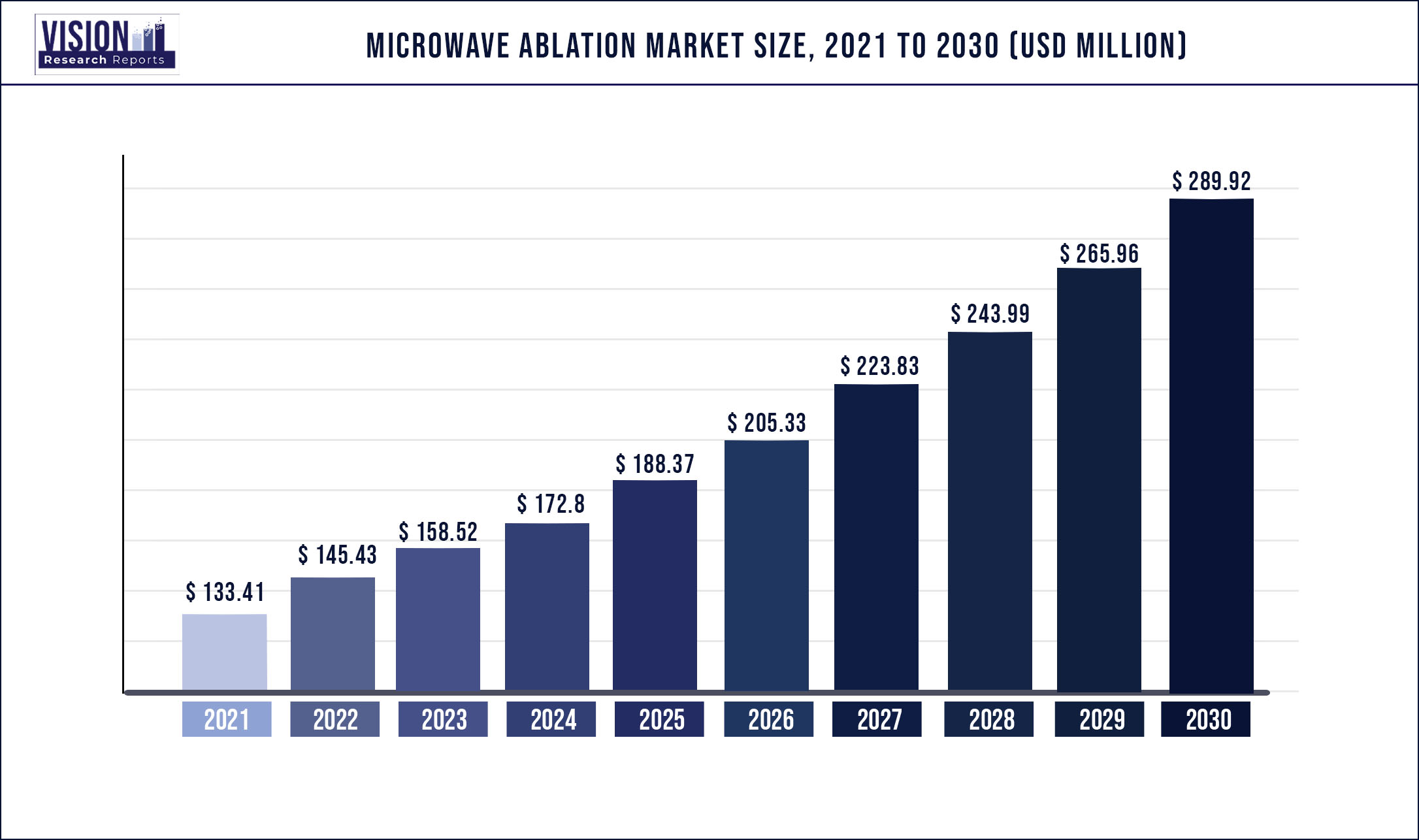
The global microwave ablation market was estimated at USD 133.41 million in 2021 and it is expected to surpass around USD 289.92 million by 2030, poised to grow at a CAGR of 9.01% from 2022 to 2030.
The microwave ablation market is growing rapidly due to its increasing incidences of tumors of the liver, lung, bone, and kidney globally. Additionally, a growing number of processes with the image-guided device is also expected to fuel the growth of the market. Technical advancements and a rising preference for minimally invasive surgeries is also expected to witness the growth of microwave ablation devices in near future. However, the high cost of devices used during the microwave ablation procedures and the preference for radiofrequency ablation devices over microwave ablation (MWA) devices by the respective surgeons is hindering the market growth.
Microwave ablation (MWA) is a thermal technology, that incorporates MRI and radio-frequency equipment for the treatment of various benign and malignant tumors. Microwave ablation is being explored for an assortment of restorative treatments including treatment of endometrial bleeding, uterine myomas, atrial fibrillation, and cancerous tumors within the liver, kidneys, pancreas, lungs, and breast. MWA offers a less-invasive elective to surgical resection of tumors, allowing shortened recovery times and offering a rational treatment choice for cancer patients who are not candidates for surgery. The microwave ablation procedure uses the most technically advanced devices that provide short ablation time, higher temperature for the ablation, weakened heat-sink effect than that of radiofrequency ablations, and large ablation zones. These factors are proven to improve tumor ablation efficacy using the MWA process.
The microwave ablation market is segmented based on component type into the generator, power distribution system, and accessories. Based on application, the microwave ablation market is segmented into oncology, cardiovascular, urology, orthopedic, and others. Additionally, based on the end-user, the market is segmented into hospitals, ambulatory surgical centers, and research laboratories.
The companies are working on developing innovative microwave ablation systems with the help of their research and innovation. For instance, In July 2020, Ethicon (Subsidiary of Johnson & Johnson Medical Devices Company) announced that the U.S. Food and Drug Administration (FDA) has approved their NEUWAVE Microwave Ablation System. Additionally, Medtronic plc has declared the breakthrough device designation status grant for the Emprint ablation catheter kit from the U.S. Food and Drug Administration (FDA), in April 2021. However, the higher number of product recalls may also impede market growth. For instance, the Solero Microwave Tissue Ablation (MTA) System and accessories from Angiodynamics Inc. has been terminated in June 2020 due to a lack of updates from the manufacturers to decrease the potential risks to patients due to delay in performing the procedure.
Scope of The Report
| Report Coverage | Details |
| Market Size in 2021 | USD 133.41 million |
| Revenue Forecast by 2030 | USD 289.92 million |
| Growth rate from 2022 to 2030 | CAGR of 9.01% |
| Base Year | 2021 |
| Forecast Period | 2022 to 2030 |
| Segmentation | Component Type, Application, End-user, Region |
| Companies Covered |
Angio Dynamics, Boston Scientific Corporation, Emblation microwave, Huber+Suhner, Johnson & Johnson Services Inc., Med Waves, Medtronic PLC, MedWaves Inc., Miramar labs, Neu Wave Medical, Pearson Medical, Symple Surgical, Urologix, Vison Medical USA Inc. |
Key Players
Market Segmentation
Chapter 1. Introduction
1.1. Research Objective
1.2. Scope of the Study
1.3. Definition
Chapter 2. Research Methodology
2.1. Research Approach
2.2. Data Sources
2.3. Assumptions & Limitations
Chapter 3. Executive Summary
3.1. Market Snapshot
Chapter 4. Market Variables and Scope
4.1. Introduction
4.2. Market Classification and Scope
4.3. Industry Value Chain Analysis
4.3.1. Raw Material Procurement Analysis
4.3.2. Sales and Distribution Channel Analysis
4.3.3. Downstream Buyer Analysis
Chapter 5. COVID 19 Impact on Microwave Ablation Market
5.1. COVID-19 Landscape: Microwave Ablation Industry Impact
5.2. COVID 19 - Impact Assessment for the Industry
5.3. COVID 19 Impact: Global Major Government Policy
5.4. Market Trends and Opportunities in the COVID-19 Landscape
Chapter 6. Market Dynamics Analysis and Trends
6.1. Market Dynamics
6.1.1. Market Drivers
6.1.2. Market Restraints
6.1.3. Market Opportunities
6.2. Porter’s Five Forces Analysis
6.2.1. Bargaining power of suppliers
6.2.2. Bargaining power of buyers
6.2.3. Threat of substitute
6.2.4. Threat of new entrants
6.2.5. Degree of competition
Chapter 7. Competitive Landscape
7.1.1. Company Market Share/Positioning Analysis
7.1.2. Key Strategies Adopted by Players
7.1.3. Vendor Landscape
7.1.3.1. List of Suppliers
7.1.3.2. List of Buyers
Chapter 8. Global Microwave Ablation Market, By Component Type
8.1. Microwave Ablation Market, by Component Type, 2022-2030
8.1.1 Generator
8.1.1.1. Market Revenue and Forecast (2017-2030)
8.1.2. Power Distribution System
8.1.2.1. Market Revenue and Forecast (2017-2030)
8.1.3. Accessories
8.1.3.1. Market Revenue and Forecast (2017-2030)
Chapter 9. Global Microwave Ablation Market, By Application
9.1. Microwave Ablation Market, by Application, 2022-2030
9.1.1. Oncology
9.1.1.1. Market Revenue and Forecast (2017-2030)
9.1.2. Cardiovascular
9.1.2.1. Market Revenue and Forecast (2017-2030)
9.1.3. Urology
9.1.3.1. Market Revenue and Forecast (2017-2030)
9.1.4. Orthopedic
9.1.4.1. Market Revenue and Forecast (2017-2030)
9.1.5. Others
9.1.5.1. Market Revenue and Forecast (2017-2030)
Chapter 10. Global Microwave Ablation Market, By End-user
10.1. Microwave Ablation Market, by End-user, 2022-2030
10.1.1. Hospitals
10.1.1.1. Market Revenue and Forecast (2017-2030)
10.1.2. Ambulatory Surgical Centers
10.1.2.1. Market Revenue and Forecast (2017-2030)
10.1.3. Research Laboratories
10.1.3.1. Market Revenue and Forecast (2017-2030)
Chapter 11. Global Microwave Ablation Market, Regional Estimates and Trend Forecast
11.1. North America
11.1.1. Market Revenue and Forecast, by Component Type (2017-2030)
11.1.2. Market Revenue and Forecast, by Application (2017-2030)
11.1.3. Market Revenue and Forecast, by End-user (2017-2030)
11.1.4. U.S.
11.1.4.1. Market Revenue and Forecast, by Component Type (2017-2030)
11.1.4.2. Market Revenue and Forecast, by Application (2017-2030)
11.1.4.3. Market Revenue and Forecast, by End-user (2017-2030)
11.1.5. Rest of North America
11.1.5.1. Market Revenue and Forecast, by Component Type (2017-2030)
11.1.5.2. Market Revenue and Forecast, by Application (2017-2030)
11.1.5.3. Market Revenue and Forecast, by End-user (2017-2030)
11.2. Europe
11.2.1. Market Revenue and Forecast, by Component Type (2017-2030)
11.2.2. Market Revenue and Forecast, by Application (2017-2030)
11.2.3. Market Revenue and Forecast, by End-user (2017-2030)
11.2.4. UK
11.2.4.1. Market Revenue and Forecast, by Component Type (2017-2030)
11.2.4.2. Market Revenue and Forecast, by Application (2017-2030)
11.2.4.3. Market Revenue and Forecast, by End-user (2017-2030)
11.2.5. Germany
11.2.5.1. Market Revenue and Forecast, by Component Type (2017-2030)
11.2.5.2. Market Revenue and Forecast, by Application (2017-2030)
11.2.5.3. Market Revenue and Forecast, by End-user (2017-2030)
11.2.6. France
11.2.6.1. Market Revenue and Forecast, by Component Type (2017-2030)
11.2.6.2. Market Revenue and Forecast, by Application (2017-2030)
11.2.6.3. Market Revenue and Forecast, by End-user (2017-2030)
11.2.7. Rest of Europe
11.2.7.1. Market Revenue and Forecast, by Component Type (2017-2030)
11.2.7.2. Market Revenue and Forecast, by Application (2017-2030)
11.2.7.3. Market Revenue and Forecast, by End-user (2017-2030)
11.3. APAC
11.3.1. Market Revenue and Forecast, by Component Type (2017-2030)
11.3.2. Market Revenue and Forecast, by Application (2017-2030)
11.3.3. Market Revenue and Forecast, by End-user (2017-2030)
11.3.4. India
11.3.4.1. Market Revenue and Forecast, by Component Type (2017-2030)
11.3.4.2. Market Revenue and Forecast, by Application (2017-2030)
11.3.4.3. Market Revenue and Forecast, by End-user (2017-2030)
11.3.5. China
11.3.5.1. Market Revenue and Forecast, by Component Type (2017-2030)
11.3.5.2. Market Revenue and Forecast, by Application (2017-2030)
11.3.5.3. Market Revenue and Forecast, by End-user (2017-2030)
11.3.6. Japan
11.3.6.1. Market Revenue and Forecast, by Component Type (2017-2030)
11.3.6.2. Market Revenue and Forecast, by Application (2017-2030)
11.3.6.3. Market Revenue and Forecast, by End-user (2017-2030)
11.3.7. Rest of APAC
11.3.7.1. Market Revenue and Forecast, by Component Type (2017-2030)
11.3.7.2. Market Revenue and Forecast, by Application (2017-2030)
11.3.7.3. Market Revenue and Forecast, by End-user (2017-2030)
11.4. MEA
11.4.1. Market Revenue and Forecast, by Component Type (2017-2030)
11.4.2. Market Revenue and Forecast, by Application (2017-2030)
11.4.3. Market Revenue and Forecast, by End-user (2017-2030)
11.4.4. GCC
11.4.4.1. Market Revenue and Forecast, by Component Type (2017-2030)
11.4.4.2. Market Revenue and Forecast, by Application (2017-2030)
11.4.4.3. Market Revenue and Forecast, by End-user (2017-2030)
11.4.5. North Africa
11.4.5.1. Market Revenue and Forecast, by Component Type (2017-2030)
11.4.5.2. Market Revenue and Forecast, by Application (2017-2030)
11.4.5.3. Market Revenue and Forecast, by End-user (2017-2030)
11.4.6. South Africa
11.4.6.1. Market Revenue and Forecast, by Component Type (2017-2030)
11.4.6.2. Market Revenue and Forecast, by Application (2017-2030)
11.4.6.3. Market Revenue and Forecast, by End-user (2017-2030)
11.4.7. Rest of MEA
11.4.7.1. Market Revenue and Forecast, by Component Type (2017-2030)
11.4.7.2. Market Revenue and Forecast, by Application (2017-2030)
11.4.7.3. Market Revenue and Forecast, by End-user (2017-2030)
11.5. Latin America
11.5.1. Market Revenue and Forecast, by Component Type (2017-2030)
11.5.2. Market Revenue and Forecast, by Application (2017-2030)
11.5.3. Market Revenue and Forecast, by End-user (2017-2030)
11.5.4. Brazil
11.5.4.1. Market Revenue and Forecast, by Component Type (2017-2030)
11.5.4.2. Market Revenue and Forecast, by Application (2017-2030)
11.5.4.3. Market Revenue and Forecast, by End-user (2017-2030)
11.5.5. Rest of LATAM
11.5.5.1. Market Revenue and Forecast, by Component Type (2017-2030)
11.5.5.2. Market Revenue and Forecast, by Application (2017-2030)
11.5.5.3. Market Revenue and Forecast, by End-user (2017-2030)
Chapter 12. Company Profiles
12.1. Angio Dynamics
12.1.1. Company Overview
12.1.2. Product Offerings
12.1.3. Financial Performance
12.1.4. Recent Initiatives
12.2. Boston Scientific Corporation
12.2.1. Company Overview
12.2.2. Product Offerings
12.2.3. Financial Performance
12.2.4. Recent Initiatives
12.3. Emblation microwave
12.3.1. Company Overview
12.3.2. Product Offerings
12.3.3. Financial Performance
12.3.4. Recent Initiatives
12.4. Huber+Suhner
12.4.1. Company Overview
12.4.2. Product Offerings
12.4.3. Financial Performance
12.4.4. Recent Initiatives
12.5. Johnson & Johnson Services Inc.
12.5.1. Company Overview
12.5.2. Product Offerings
12.5.3. Financial Performance
12.5.4. Recent Initiatives
12.6. Med Waves
12.6.1. Company Overview
12.6.2. Product Offerings
12.6.3. Financial Performance
12.6.4. Recent Initiatives
12.7. Medtronic PLC
12.7.1. Company Overview
12.7.2. Product Offerings
12.7.3. Financial Performance
12.7.4. Recent Initiatives
12.8. MedWaves Inc.
12.8.1. Company Overview
12.8.2. Product Offerings
12.8.3. Financial Performance
12.8.4. Recent Initiatives
12.9. Miramar labs
12.9.1. Company Overview
12.9.2. Product Offerings
12.9.3. Financial Performance
12.9.4. Recent Initiatives
12.10. Neu Wave Medical
12.10.1. Company Overview
12.10.2. Product Offerings
12.10.3. Financial Performance
12.10.4. Recent Initiatives
Chapter 13. Research Methodology
13.1. Primary Research
13.2. Secondary Research
13.3. Assumptions
Chapter 14. Appendix
14.1. About Us
14.2. Glossary of Terms
 Cross-segment Market Size and Analysis for
Mentioned Segments
Cross-segment Market Size and Analysis for
Mentioned Segments
 Additional Company Profiles (Upto 5 With No Cost)
Additional Company Profiles (Upto 5 With No Cost)
 Additional Countries (Apart From Mentioned Countries)
Additional Countries (Apart From Mentioned Countries)
 Country/Region-specific Report
Country/Region-specific Report
 Go To Market Strategy
Go To Market Strategy
 Region Specific Market Dynamics
Region Specific Market Dynamics Region Level Market Share
Region Level Market Share Import Export Analysis
Import Export Analysis Production Analysis
Production Analysis Others
Others