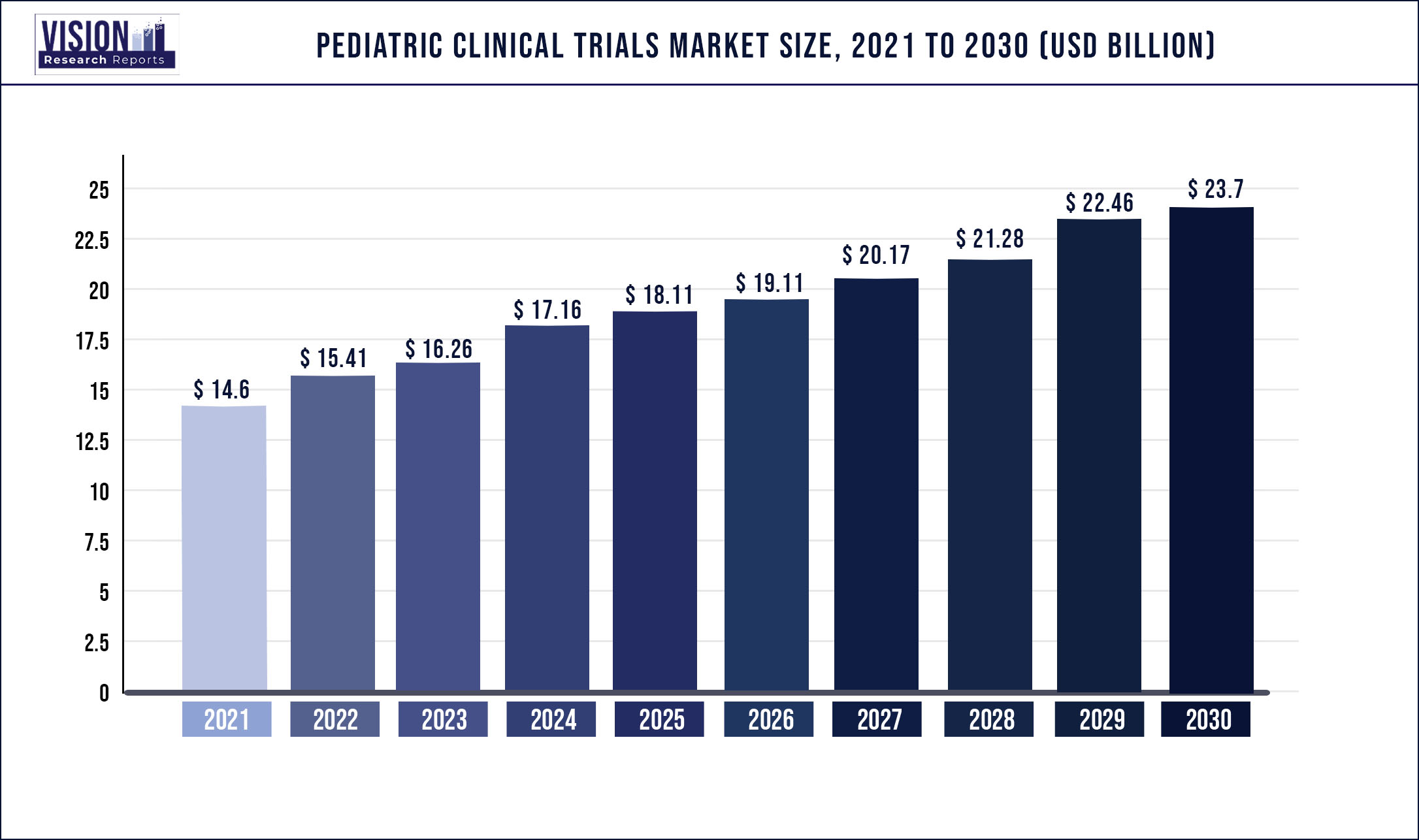
The global pediatric clinical trials market was estimated at USD 14.6 billion in 2021 and it is expected to surpass around USD 23.7 billion by 2030, poised to grow at a CAGR of 5.53% from 2022 to 2030.
The market is growing on account of the rise in the number of pediatric clinical studies globally. As per the BMC journal, between January 2008 and December 2010, 7,029 pediatric clinical trials were registered. Whereas, between January 2017 and December 2019, the number of registered pediatric clinical trials was 11,738, which certainly reflects a significant surge in the total number of trials across the industry. The treatment options for COVID-19 pediatric patients were limited at the start of the pandemic, owing to which, in May 2020, FDA granted an Emergency Use Authorization (EUA) for Veklury (remdesivir), which could be used for the treatment of COVID-19 in adults and children.
Similarly, Convalescent plasma was also granted an EUA from the FDA for the treatment of children and adults with COVID-19. Such actions by the regulatory agencies are likely to promote the demand for pediatric clinical trials. Furthermore, increasing efforts by several government bodies to support the clinical research pertaining to pediatric diseases has further supported the market growth. For instance, in March 2022, the National Cancer Institute launched the Molecular Characterization Initiative for pediatric tumors. The Initiative is offered through NCI’s Childhood Cancer Data Initiative, which was formed to promote data sharing and collection of new data among researchers who study pediatric oncology.
In addition, the increasing prevalence of pediatric cancer is another significant factor supporting the market as well as the oncology segment’s growth. For instance, as stated by the American Society of Clinical Oncology (ASCO)in February 2022, in the U.S., an estimated 10,470 children are younger than 15 years of age and about 5,480 teens aged 15 to 19 years will be diagnosed with cancer by the end of 2022. The WHO states that diarrhea, malaria, pneumonia, and sepsis are the leading causes of death among children between the age of 1 month and 9 years The high burden of these diseases is contributing to the demand for new treatment opinions. Thus, is likely to have a positive impact on the market growth. Apart from the high burden of infectious diseases, a significant number of children also suffer from diabetes.
For instance, according to the U.S. CDC, over 26.9 million in the U.S. were diagnosed with diabetes in 2018, which included 210,000 children and adolescents younger than age 20—or 25 years. Commercially, COVID19 vaccines were majorly focused on immunizing adults aged 18 years and above, this created a high risk of disease among children below the age of 18 years. The ClinicalTrial.GOV reports that as on 4th May 2022, over 1000 studies were in the active stage for treating and diagnosing COVID-19 for pediatric patients. The burden of COVID-19 among the kids is likely to promote the demand for COVID-19 vaccine trials for pediatrics from 2021.
Scope of The Report
| Report Coverage | Details |
| Market Size in 2021 | USD 14.6 Billion |
| Revenue Forecast by 2030 | USD 23.7 Billion |
| Growth rate from 2022 to 2030 | CAGR of 5.53% |
| Base Year | 2021 |
| Forecast Period | 2022 to 2030 |
| Segmentation | Phase, study design, therapeutic areas, region |
| Companies Covered | ICON plc,Syneos Health,Medpace, Inc.,PPD Inc.,Premier Research,LabCorp Drug Development ,QPS Holdings.,Pfizer Inc.,The Emmes Company, LLC,IQVIA Inc. |
Phase Insights
On the basis of phase, the global market has been further sub-segmented into phases I, II, III, and IV pediatric clinical trials. The phase II trials segment dominated the market in 2021 and accounted for the maximum share of more than 32.00% of the global revenue. Phase II trials are conducted on children to investigate the safety and efficacy of the intervention. Phase II studies consist of a high number of pediatric subjects; moreover, studies in this phase are more complex than those in other phases. Generally, medicinal product testing in children is deferred until the trials reach phase III, which examines the efficacy, acceptability, and adverse effects.
Although the purpose of the deferral is to safeguard children from undue injury, it also implies that children’s access to potentially valuable treatments would be delayed. The phase IV segment is expected to register the fastest growth rate of more than 5.00% during the forecast period. Phase IV post-marketing trials are infrequently conducted in children. However, the Food and Drug Administration (FDA) Pediatric Research Equity Act (PREA) requires pediatric trials of marketed medicines.
Study Design Insights
On the basis of study design, the global market has been further categorized into treatment studies and observational studies. Treatment studies have accounted for the maximum revenue share of the global market of more than 65.00% in 2021. According to Clinicaltrials.gov, from 2000 to 2019, a total of 53,060 pediatric trials were registered, with 36,136 (68.1%) including treatment studies. However, the majority of treatment pediatric trials were single site (61.4%), small-scale (58.9%), randomized (66.0%), and had no masking (56.9%), there were fewer drug trials conducted throughout time.
In terms of observational trial categories, drug studies were the most common (36.9%), while behavioral treatment trials increased from 12.6 to 20.4%. The treatment studies for pediatric trials are mostly funded by Institutions (78.8%), industry (19.1%), NIH (National Institute of Health) (1.6%) & U.S. federal agencies (0.5%). In addition, the industry-funded trials were completed more frequently and were more focused on treatment, whether they were single-site or multi-site trials. NIH-funded and other-funded trials, on the other hand, had a larger proportion of behavioral and "other" intervention trials.
Therapeutic Areas Insights
On the basis of therapeutic areas, the global market has been further segmented into infectious diseases, oncology, autoimmune/inflammation, respiratory disorders, mental health disorders, and others. The oncology therapeutic area segment accounted for the second-largest revenue share of more than 17.7% in the global market in 2021. Cancer is one of the major causes of death worldwide. It is estimated that cancer cases among children below the age of 19 years are likely to reach 291,000 by 2040. The Molecular Characterization Initiative is offered through NCI’s Childhood Cancer Data Initiative, which was launched in 2019 to promote data sharing and collection of new data among researchers who study childhood cancers.
The government has launched this to collect data on pediatric cancer for researchers. It’s a kind of indirect support that will boost the segment’s growth. The mental health disorders segment is expected to rise with the fastest growth rate over the forecast period. The high prevalence of CNS-related issues, such as autism hyperactivity, migraine, seizures, and others, in children, is one of the key reasons for the high demand for pediatric clinical trials for mental health disorders. The funding for CNS-related clinical studies from various public organizations is expected to further support the growth of this segment.
Regional Insights
On the basis of geographies, the market has been further divided into North America, Asia Pacific, Latin America, Europe, Middle East & Africa. North America dominated the global market in 2021 and accounted for the largest share of 44.0% of the global revenue. This is due to the presence of a significant number of pharmaceutical companies and CROs conducting pediatric trials in the region. Apart from this, public organizations are also undertaking active initiatives to support pediatric research. For instance, the current NIH funding for pediatric research accounts for USD 4 billion. Such initiatives are further contributing to the market growth in this region.
Asia Pacific, on the other hand, is projected to register the fastest CAGR during the forecast period. The Asia Pacific region has become a hotspot for conducting clinical trials on account of the ease of regulatory compliance, low study costs, and rapidly growing pediatric patient population. Moreover, supportive initiatives promoting pediatric research in the region, owing to the high prevalence of pediatric diseases, are driving the regional market. For instance, UNESCO, in March 2022, reported that researchers in China are focusing on making advances in neuroscience to treat children with autism in the country.
Key Players
ICON plc,Syneos Health,Medpace, Inc.,PPD Inc.,Premier Research,LabCorp Drug Development ,QPS Holdings.,Pfizer Inc.,The Emmes Company, LLC,IQVIA Inc.
Market Segmentation
Chapter 1. Introduction
1.1. Research Objective
1.2. Scope of the Study
1.3. Definition
Chapter 2. Research Methodology
2.1. Research Approach
2.2. Data Sources
2.3. Assumptions & Limitations
Chapter 3. Executive Summary
3.1. Market Snapshot
Chapter 4. Market Variables and Scope
4.1. Introduction
4.2. Market Classification and Scope
4.3. Industry Value Chain Analysis
4.3.1. Raw Material Procurement Analysis
4.3.2. Sales and Therapeutic Areas Analysis
4.3.3. Downstream Buyer Analysis
Chapter 5. COVID 19 Impact on Pediatric Clinical Trials Market
5.1. COVID-19 Landscape: Pediatric Clinical Trials Industry Impact
5.2. COVID 19 - Impact Assessment for the Industry
5.3. COVID 19 Impact: Global Major Government Policy
5.4. Market Trends and Opportunities in the COVID-19 Landscape
Chapter 6. Market Dynamics Analysis and Trends
6.1. Market Dynamics
6.1.1. Market Drivers
6.1.2. Market Restraints
6.1.3. Market Opportunities
6.2. Porter’s Five Forces Analysis
6.2.1. Bargaining power of suppliers
6.2.2. Bargaining power of buyers
6.2.3. Threat of substitute
6.2.4. Threat of new entrants
6.2.5. Degree of competition
Chapter 7. Competitive Landscape
7.1.1. Company Market Share/Positioning Analysis
7.1.2. Key Strategies Adopted by Players
7.1.3. Vendor Landscape
7.1.3.1. List of Suppliers
7.1.3.2. List of Buyers
Chapter 8. Global Pediatric Clinical Trials Market, By Phase
8.1. Pediatric Clinical Trials Market, by Phase Type, 2022-2030
8.1.1. Phase I
8.1.1.1. Market Revenue and Forecast (2017-2030)
8.1.2. Phase II
8.1.2.1. Market Revenue and Forecast (2017-2030)
8.1.3. Phase III
8.1.3.1. Market Revenue and Forecast (2017-2030)
8.1.4. Phase IV
8.1.4.1. Market Revenue and Forecast (2017-2030)
Chapter 9. Global Pediatric Clinical Trials Market, By Study Design
9.1. Pediatric Clinical Trials Market, by Study Design, 2022-2030
9.1.1. Treatment Studies
9.1.1.1. Market Revenue and Forecast (2017-2030)
9.1.2. Observational Studies
9.1.2.1. Market Revenue and Forecast (2017-2030)
Chapter 10. Global Pediatric Clinical Trials Market, By Therapeutic Areas
10.1. Pediatric Clinical Trials Market, by Therapeutic Areas, 2022-2030
10.1.1. Infectious Diseases
10.1.1.1. Market Revenue and Forecast (2017-2030)
10.1.2. Oncology
10.1.2.1. Market Revenue and Forecast (2017-2030)
10.1.3. Autoimmune/Inflammatory Diseases
10.1.3.1. Market Revenue and Forecast (2017-2030)
10.1.4. Respiratory Disorders
10.1.4.1. Market Revenue and Forecast (2017-2030)
10.1.5. Mental Health Disorders
10.1.5.1. Market Revenue and Forecast (2017-2030)
10.1.6. Others
10.1.6.1. Market Revenue and Forecast (2017-2030)
Chapter 11. Global Pediatric Clinical Trials Market, Regional Estimates and Trend Forecast
11.1. North America
11.1.1. Market Revenue and Forecast, by Phase (2017-2030)
11.1.2. Market Revenue and Forecast, by Study Design (2017-2030)
11.1.3. Market Revenue and Forecast, by Therapeutic Areas (2017-2030)
11.1.4. U.S.
11.1.4.1. Market Revenue and Forecast, by Phase (2017-2030)
11.1.4.2. Market Revenue and Forecast, by Study Design (2017-2030)
11.1.4.3. Market Revenue and Forecast, by Therapeutic Areas (2017-2030)
11.1.5. Rest of North America
11.1.5.1. Market Revenue and Forecast, by Phase (2017-2030)
11.1.5.2. Market Revenue and Forecast, by Study Design (2017-2030)
11.1.5.3. Market Revenue and Forecast, by Therapeutic Areas (2017-2030)
11.2. Europe
11.2.1. Market Revenue and Forecast, by Phase (2017-2030)
11.2.2. Market Revenue and Forecast, by Study Design (2017-2030)
11.2.3. Market Revenue and Forecast, by Therapeutic Areas (2017-2030)
11.2.4. UK
11.2.4.1. Market Revenue and Forecast, by Phase (2017-2030)
11.2.4.2. Market Revenue and Forecast, by Study Design (2017-2030)
11.2.4.3. Market Revenue and Forecast, by Therapeutic Areas (2017-2030)
11.2.5. Germany
11.2.5.1. Market Revenue and Forecast, by Phase (2017-2030)
11.2.5.2. Market Revenue and Forecast, by Study Design (2017-2030)
11.2.5.3. Market Revenue and Forecast, by Therapeutic Areas (2017-2030)
11.2.6. France
11.2.6.1. Market Revenue and Forecast, by Phase (2017-2030)
11.2.6.2. Market Revenue and Forecast, by Study Design (2017-2030)
11.2.6.3. Market Revenue and Forecast, by Therapeutic Areas (2017-2030)
11.2.7. Rest of Europe
11.2.7.1. Market Revenue and Forecast, by Phase (2017-2030)
11.2.7.2. Market Revenue and Forecast, by Study Design (2017-2030)
11.2.7.3. Market Revenue and Forecast, by Therapeutic Areas (2017-2030)
11.3. APAC
11.3.1. Market Revenue and Forecast, by Phase (2017-2030)
11.3.2. Market Revenue and Forecast, by Study Design (2017-2030)
11.3.3. Market Revenue and Forecast, by Therapeutic Areas (2017-2030)
11.3.4. India
11.3.4.1. Market Revenue and Forecast, by Phase (2017-2030)
11.3.4.2. Market Revenue and Forecast, by Study Design (2017-2030)
11.3.4.3. Market Revenue and Forecast, by Therapeutic Areas (2017-2030)
11.3.5. China
11.3.5.1. Market Revenue and Forecast, by Phase (2017-2030)
11.3.5.2. Market Revenue and Forecast, by Study Design (2017-2030)
11.3.5.3. Market Revenue and Forecast, by Therapeutic Areas (2017-2030)
11.3.6. Japan
11.3.6.1. Market Revenue and Forecast, by Phase (2017-2030)
11.3.6.2. Market Revenue and Forecast, by Study Design (2017-2030)
11.3.6.3. Market Revenue and Forecast, by Therapeutic Areas (2017-2030)
11.3.7. Rest of APAC
11.3.7.1. Market Revenue and Forecast, by Phase (2017-2030)
11.3.7.2. Market Revenue and Forecast, by Study Design (2017-2030)
11.3.7.3. Market Revenue and Forecast, by Therapeutic Areas (2017-2030)
11.4. MEA
11.4.1. Market Revenue and Forecast, by Phase (2017-2030)
11.4.2. Market Revenue and Forecast, by Study Design (2017-2030)
11.4.3. Market Revenue and Forecast, by Therapeutic Areas (2017-2030)
11.4.4. GCC
11.4.4.1. Market Revenue and Forecast, by Phase (2017-2030)
11.4.4.2. Market Revenue and Forecast, by Study Design (2017-2030)
11.4.4.3. Market Revenue and Forecast, by Therapeutic Areas (2017-2030)
11.4.5. North Africa
11.4.5.1. Market Revenue and Forecast, by Phase (2017-2030)
11.4.5.2. Market Revenue and Forecast, by Study Design (2017-2030)
11.4.5.3. Market Revenue and Forecast, by Therapeutic Areas (2017-2030)
11.4.6. South Africa
11.4.6.1. Market Revenue and Forecast, by Phase (2017-2030)
11.4.6.2. Market Revenue and Forecast, by Study Design (2017-2030)
11.4.6.3. Market Revenue and Forecast, by Therapeutic Areas (2017-2030)
11.4.7. Rest of MEA
11.4.7.1. Market Revenue and Forecast, by Phase (2017-2030)
11.4.7.2. Market Revenue and Forecast, by Study Design (2017-2030)
11.4.7.3. Market Revenue and Forecast, by Therapeutic Areas (2017-2030)
11.5. Latin America
11.5.1. Market Revenue and Forecast, by Phase (2017-2030)
11.5.2. Market Revenue and Forecast, by Study Design (2017-2030)
11.5.3. Market Revenue and Forecast, by Therapeutic Areas (2017-2030)
11.5.4. Brazil
11.5.4.1. Market Revenue and Forecast, by Phase (2017-2030)
11.5.4.2. Market Revenue and Forecast, by Study Design (2017-2030)
11.5.4.3. Market Revenue and Forecast, by Therapeutic Areas (2017-2030)
11.5.5. Rest of LATAM
11.5.5.1. Market Revenue and Forecast, by Phase (2017-2030)
11.5.5.2. Market Revenue and Forecast, by Study Design (2017-2030)
11.5.5.3. Market Revenue and Forecast, by Therapeutic Areas (2017-2030)
Chapter 12. Company Profiles
12.1. ICON plc
12.1.1. Company Overview
12.1.2. Product Offerings
12.1.3. Financial Performance
12.1.4. Recent Initiatives
12.2. Syneos Health
12.2.1. Company Overview
12.2.2. Product Offerings
12.2.3. Financial Performance
12.2.4. Recent Initiatives
12.3. Medpace, Inc.
12.3.1. Company Overview
12.3.2. Product Offerings
12.3.3. Financial Performance
12.3.4. Recent Initiatives
12.4. PPD Inc.
12.4.1. Company Overview
12.4.2. Product Offerings
12.4.3. Financial Performance
12.4.4. Recent Initiatives
12.5. Premier Research
12.5.1. Company Overview
12.5.2. Product Offerings
12.5.3. Financial Performance
12.5.4. Recent Initiatives
12.6. LabCorp Drug Development
12.6.1. Company Overview
12.6.2. Product Offerings
12.6.3. Financial Performance
12.6.4. Recent Initiatives
12.7. QPS Holdings.
12.7.1. Company Overview
12.7.2. Product Offerings
12.7.3. Financial Performance
12.7.4. Recent Initiatives
12.8. Pfizer Inc.
12.8.1. Company Overview
12.8.2. Product Offerings
12.8.3. Financial Performance
12.8.4. Recent Initiatives
12.9. The Emmes Company, LLC
12.9.1. Company Overview
12.9.2. Product Offerings
12.9.3. Financial Performance
12.9.4. Recent Initiatives
12.10. IQVIA Inc.
12.10.1. Company Overview
12.10.2. Product Offerings
12.10.3. Financial Performance
12.10.4. Recent Initiatives
Chapter 13. Research Methodology
13.1. Primary Research
13.2. Secondary Research
13.3. Assumptions
Chapter 14. Appendix
14.1. About Us
14.2. Glossary of Terms
 Cross-segment Market Size and Analysis for
Mentioned Segments
Cross-segment Market Size and Analysis for
Mentioned Segments
 Additional Company Profiles (Upto 5 With No Cost)
Additional Company Profiles (Upto 5 With No Cost)
 Additional Countries (Apart From Mentioned Countries)
Additional Countries (Apart From Mentioned Countries)
 Country/Region-specific Report
Country/Region-specific Report
 Go To Market Strategy
Go To Market Strategy
 Region Specific Market Dynamics
Region Specific Market Dynamics Region Level Market Share
Region Level Market Share Import Export Analysis
Import Export Analysis Production Analysis
Production Analysis Others
Others