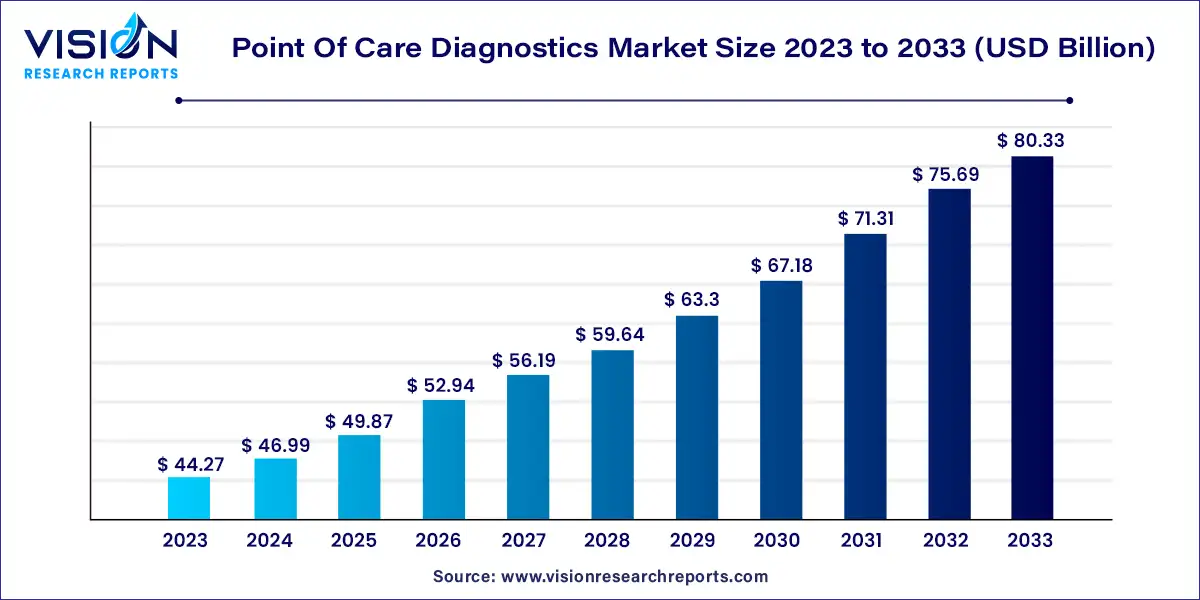
The global point of care diagnostics market was estimated at USD 44.27 billion in 2023 and it is expected to surpass around USD 80.33 billion by 2033, poised to grow at a CAGR of 6.14% from 2024 to 2033.
Point of care diagnostics, also known as bedside testing or near-patient testing, refers to medical diagnostic testing performed outside of a traditional laboratory setting. This approach enables rapid and convenient testing, providing immediate results that aid in clinical decision-making.
The point of care diagnostics market has witnessed significant growth in recent years, driven by factors such as increasing prevalence of infectious diseases, rising demand for decentralized healthcare solutions, and advancements in technology. These diagnostics encompass a wide range of tests, including blood glucose monitoring, pregnancy testing, infectious disease screening, cardiac markers testing, and more.
The growth of the point of care diagnostics market is driven by an advancement in technology have led to the development of portable and user-friendly diagnostic devices, enabling rapid testing and immediate results. Additionally, the increasing prevalence of infectious diseases worldwide has heightened the demand for decentralized healthcare solutions, further propelling the market growth. Moreover, the COVID-19 pandemic has underscored the importance of point of care diagnostics in pandemic management, accelerating the adoption of rapid antigen tests and molecular assays. Furthermore, the emphasis on accessibility, efficiency, and cost-effectiveness in healthcare systems has fueled the demand for point of care diagnostics, driving market expansion.
Infectious diseases dominated the market, comprising 28% of global revenue share in 2023. The segment's growth is fueled by the increasing demand for rapid tests, prompting industry players to offer point of care solutions in decentralized regions and introduce innovative products. For instance, Sensible Diagnostics revealed plans in May 2023 to launch a POC PCR instrument capable of performing PCR tests within 10 minutes by 2024, with a primary focus on infectious diseases. Additionally, Abbott announced the release of ID NOW, the world's fastest molecular POC test, delivering COVID-19 results in just 13 minutes and suitable for various dispersed healthcare settings like doctor offices and urgent care clinics.
The cancer segment is projected to experience the highest CAGR during the forecast period. The increased use of markers in cancer diagnostics is the primary factor driving growth in the cancer markers segment. The rising global prevalence of cancer underscores the importance of early disease detection and is anticipated to fuel market growth. For instance, Cancer.org reported approximately 1.9 million new cancer cases diagnosed in the U.S. in 2021, resulting in 608,570 cancer-related deaths. Moreover, key market players are introducing innovative products to capitalize on untapped opportunities.
The clinic end-use segment emerged as the market leader, holding the highest revenue share in 2023. Point of care diagnostics in primary care settings encompass a wide range of tests, from simple glucose testing to complex coagulation testing. Many clinics are transitioning to point of care diagnostics from traditional lab testing, which helps expedite decision-making by reducing delays in specimen preparation and transport, thereby determining the need for further tests more swiftly. Point of care diagnostics offer several advantages, including rapid availability of results, cost-effectiveness, and improved outcomes. The market has experienced rapid growth in recent years, particularly in the U.S. and European countries, driven by increased awareness of medical and organizational benefits, as well as the economic advantages of point of care diagnostics.
The home sector is poised to witness the highest growth rate over the forecast period, owing to the convenience and cost-effectiveness of point of care diagnostics delivered directly to patients at home. Point of care diagnostics in the home healthcare sector enable patients to address healthcare needs at home and make informed decisions instantly. Additionally, with the aging population being more susceptible to chronic diseases, the demand for healthcare services is expected to steadily rise in the coming years. This trend will further boost the home healthcare sector, enhancing overall access to medical care while reducing unnecessary visits, hospital admissions, readmissions, as well as the time and costs associated with traveling to healthcare providers.
North America asserted its dominance in the market, capturing a significant 43% share in 2023. This strong presence can be attributed to several key factors, including the growing geriatric population and the region's higher healthcare expenditure. Additionally, technological advancements, such as the introduction of miniaturized diagnostic equipment delivering accurate and rapid results, coupled with the increasing adoption of Picture Archiving and Communication Systems (PACS) and Electronic Medical Records (EMRs), are expected to further propel market growth in the region during the forecast period. Furthermore, the market in North America benefits from the presence of key players like Abbott, BIOMERIEUX, BD, Siemens Healthineers AG, QIAGEN, Quidel Corporation, and Quest Diagnostics, whose innovations and developments contribute positively to market expansion. For instance, in April 2023, Abbott announced the U.S. FDA clearance for the reader for FreeStyle Libre 3 integrated continuous glucose monitoring system, demonstrating the region's commitment to advancing healthcare technology.
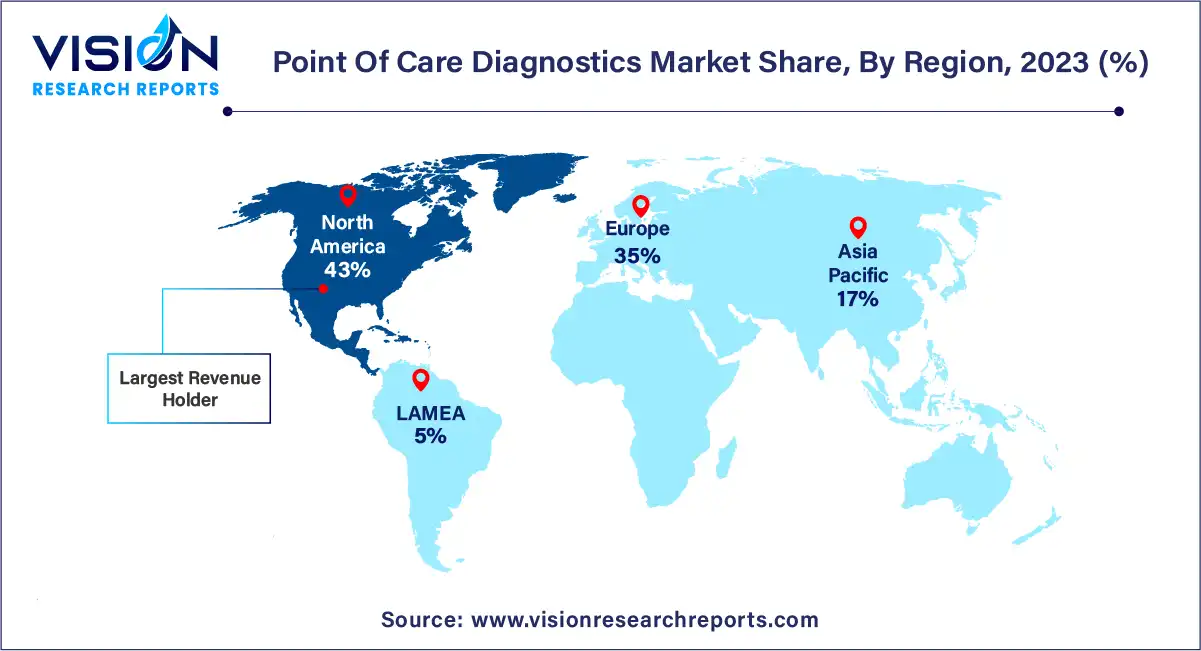
On the other hand, Asia Pacific is poised to witness significant growth in the point of care diagnostics market. Factors driving this growth include the ongoing development of healthcare infrastructure and the higher prevalence of chronic and targeted diseases such as diabetes, cancer, and infectious conditions like HIV, syphilis, and RSV across Asia Pacific countries. The adoption of point of care diagnostics in various healthcare settings such as operating rooms, emergency rooms, intensive care units, path labs, and hospitals is expected to increase due to the promise of early and efficient results. Additionally, the region benefits from a robust product pipeline, particularly in the infectious and cardiac markers segment, which is anticipated to further bolster market growth through to 2030.
By Product
By End-use
By Region
Chapter 1. Introduction
1.1. Research Objective
1.2. Scope of the Study
1.3. Definition
Chapter 2. Research Methodology
2.1. Research Approach
2.2. Data Sources
2.3. Assumptions & Limitations
Chapter 3. Executive Summary
3.1. Market Snapshot
Chapter 4. Market Variables and Scope
4.1. Introduction
4.2. Market Classification and Scope
4.3. Industry Value Chain Analysis
4.3.1. Raw Material Procurement Analysis
4.3.2. Sales and Distribution Channel Analysis
4.3.3. Downstream Buyer Analysis
Chapter 5. COVID 19 Impact on Point Of Care Diagnostics Market
5.1. COVID-19 Landscape: Point Of Care Diagnostics Industry Impact
5.2. COVID 19 - Impact Assessment for the Industry
5.3. COVID 19 Impact: Global Major Government Policy
5.4. Market Trends and Opportunities in the COVID-19 Landscape
Chapter 6. Market Dynamics Analysis and Trends
6.1. Market Dynamics
6.1.1. Market Drivers
6.1.2. Market Restraints
6.1.3. Market Opportunities
6.2. Porter’s Five Forces Analysis
6.2.1. Bargaining power of suppliers
6.2.2. Bargaining power of buyers
6.2.3. Threat of substitute
6.2.4. Threat of new entrants
6.2.5. Degree of competition
Chapter 7. Competitive Landscape
7.1.1. Company Market Share/Positioning Analysis
7.1.2. Key Strategies Adopted by Players
7.1.3. Vendor Landscape
7.1.3.1. List of Suppliers
7.1.3.2. List of Buyers
Chapter 8. Global Point Of Care Diagnostics Market, By Product
8.1. Point Of Care Diagnostics Market, by Product, 2024-2033
8.1.1. Glucose Testing
8.1.1.1. Market Revenue and Forecast (2021-2033)
8.1.2. Hb1Ac Testing
8.1.2.1. Market Revenue and Forecast (2021-2033)
8.1.3. Coagulation Testing
8.1.3.1. Market Revenue and Forecast (2021-2033)
8.1.4. Fertility/Pregnancy
8.1.4.1. Market Revenue and Forecast (2021-2033)
8.1.5. Infectious Disease
8.1.5.1. Market Revenue and Forecast (2021-2033)
8.1.6. Cardiac Markers
8.1.6.1. Market Revenue and Forecast (2021-2033)
8.1.7. Thyroid Stimulating Hormone
8.1.7.1. Market Revenue and Forecast (2021-2033)
8.1.8. Hematology
8.1.8.1. Market Revenue and Forecast (2021-2033)
8.1.9. Primary Care Systems
8.1.9.1. Market Revenue and Forecast (2021-2033)
8.1.10. Decentralized Clinical Chemistry
8.1.10.1. Market Revenue and Forecast (2021-2033)
8.1.11. Feces
8.1.11.1. Market Revenue and Forecast (2021-2033)
8.1.2. Lipid Testing
8.1.12.1. Market Revenue and Forecast (2021-2033)
8.1.13. Cancer Marker
8.1.13.1. Market Revenue and Forecast (2021-2033)
8.1.14. Blood Gas/Electrolytes
8.1.14.1. Market Revenue and Forecast (2021-2033)
8.1.15. Ambulatory Chemistry
8.1.15.1. Market Revenue and Forecast (2021-2033)
8.1.16. Drug of Abuse (DOA) Testing
8.1.16.1. Market Revenue and Forecast (2021-2033)
8.1.17. Urinalysis/Nephrology
8.1.17.1. Market Revenue and Forecast (2021-2033)
Chapter 9. Global Point Of Care Diagnostics Market, By End-use
9.1. Point Of Care Diagnostics Market, by End-use, 2024-2033
9.1.1. Clinics
9.1.1.1. Market Revenue and Forecast (2021-2033)
9.1.2. Hospitals
9.1.2.1. Market Revenue and Forecast (2021-2033)
9.1.3. Home
9.1.3.1. Market Revenue and Forecast (2021-2033)
9.1.4. Assisted Living Healthcare Facilities
9.1.4.1. Market Revenue and Forecast (2021-2033)
9.1.5. Laboratory
9.1.5.1. Market Revenue and Forecast (2021-2033)
Chapter 10. Global Point Of Care Diagnostics Market, Regional Estimates and Trend Forecast
10.1. North America
10.1.1. Market Revenue and Forecast, by Product (2021-2033)
10.1.2. Market Revenue and Forecast, by End-use (2021-2033)
10.1.3. U.S.
10.1.3.1. Market Revenue and Forecast, by Product (2021-2033)
10.1.3.2. Market Revenue and Forecast, by End-use (2021-2033)
10.1.4. Rest of North America
10.1.4.1. Market Revenue and Forecast, by Product (2021-2033)
10.1.4.2. Market Revenue and Forecast, by End-use (2021-2033)
10.2. Europe
10.2.1. Market Revenue and Forecast, by Product (2021-2033)
10.2.2. Market Revenue and Forecast, by End-use (2021-2033)
10.2.3. UK
10.2.3.1. Market Revenue and Forecast, by Product (2021-2033)
10.2.3.2. Market Revenue and Forecast, by End-use (2021-2033)
10.2.4. Germany
10.2.4.1. Market Revenue and Forecast, by Product (2021-2033)
10.2.4.2. Market Revenue and Forecast, by End-use (2021-2033)
10.2.5. France
10.2.5.1. Market Revenue and Forecast, by Product (2021-2033)
10.2.5.2. Market Revenue and Forecast, by End-use (2021-2033)
10.2.6. Rest of Europe
10.2.6.1. Market Revenue and Forecast, by Product (2021-2033)
10.2.6.2. Market Revenue and Forecast, by End-use (2021-2033)
10.3. APAC
10.3.1. Market Revenue and Forecast, by Product (2021-2033)
10.3.2. Market Revenue and Forecast, by End-use (2021-2033)
10.3.3. India
10.3.3.1. Market Revenue and Forecast, by Product (2021-2033)
10.3.3.2. Market Revenue and Forecast, by End-use (2021-2033)
10.3.4. China
10.3.4.1. Market Revenue and Forecast, by Product (2021-2033)
10.3.4.2. Market Revenue and Forecast, by End-use (2021-2033)
10.3.5. Japan
10.3.5.1. Market Revenue and Forecast, by Product (2021-2033)
10.3.5.2. Market Revenue and Forecast, by End-use (2021-2033)
10.3.6. Rest of APAC
10.3.6.1. Market Revenue and Forecast, by Product (2021-2033)
10.3.6.2. Market Revenue and Forecast, by End-use (2021-2033)
10.4. MEA
10.4.1. Market Revenue and Forecast, by Product (2021-2033)
10.4.2. Market Revenue and Forecast, by End-use (2021-2033)
10.4.3. GCC
10.4.3.1. Market Revenue and Forecast, by Product (2021-2033)
10.4.3.2. Market Revenue and Forecast, by End-use (2021-2033)
10.4.4. North Africa
10.4.4.1. Market Revenue and Forecast, by Product (2021-2033)
10.4.4.2. Market Revenue and Forecast, by End-use (2021-2033)
10.4.5. South Africa
10.4.5.1. Market Revenue and Forecast, by Product (2021-2033)
10.4.5.2. Market Revenue and Forecast, by End-use (2021-2033)
10.4.6. Rest of MEA
10.4.6.1. Market Revenue and Forecast, by Product (2021-2033)
10.4.6.2. Market Revenue and Forecast, by End-use (2021-2033)
10.5. Latin America
10.5.1. Market Revenue and Forecast, by Product (2021-2033)
10.5.2. Market Revenue and Forecast, by End-use (2021-2033)
10.5.3. Brazil
10.5.3.1. Market Revenue and Forecast, by Product (2021-2033)
10.5.3.2. Market Revenue and Forecast, by End-use (2021-2033)
10.5.4. Rest of LATAM
10.5.4.1. Market Revenue and Forecast, by Product (2021-2033)
10.5.4.2. Market Revenue and Forecast, by End-use (2021-2033)
Chapter 11. Company Profiles
11.1. Danaher
11.1.1. Company Overview
11.1.2. Product Offerings
11.1.3. Financial Performance
11.1.4. Recent Initiatives
11.2. BD
11.2.1. Company Overview
11.2.2. Product Offerings
11.2.3. Financial Performance
11.2.4. Recent Initiatives
11.3. Qiagen
11.3.1. Company Overview
11.3.2. Product Offerings
11.3.3. Financial Performance
11.3.4. Recent Initiatives
11.4. Abbott
11.4.1. Company Overview
11.4.2. Product Offerings
11.4.3. Financial Performance
11.4.4. Recent Initiatives
11.5. Siemens Healthcare AG
11.5.1. Company Overview
11.5.2. Product Offerings
11.5.3. Financial Performance
11.5.4. Recent Initiatives
11.6. bioMerieux SA
11.6.1. Company Overview
11.6.2. Product Offerings
11.6.3. Financial Performance
11.6.4. Recent Initiatives
11.7. Zoetis, Inc.
11.7.1. Company Overview
11.7.2. Product Offerings
11.7.3. Financial Performance
11.7.4. Recent Initiatives
11.8. Instrumentation Laboratory
11.8.1. Company Overview
11.8.2. Product Offerings
11.8.3. Financial Performance
11.8.4. Recent Initiatives
11.9. Nova Biomedical
11.9.1. Company Overview
11.9.2. Product Offerings
11.9.3. Financial Performance
11.9.4. Recent Initiatives
11.10. Quidel Corp.
11.10.1. Company Overview
11.10.2. Product Offerings
11.10.3. Financial Performance
11.10.4. Recent Initiatives
Chapter 12. Research Methodology
12.1. Primary Research
12.2. Secondary Research
12.3. Assumptions
Chapter 13. Appendix
13.1. About Us
13.2. Glossary of Terms
 Cross-segment Market Size and Analysis for
Mentioned Segments
Cross-segment Market Size and Analysis for
Mentioned Segments
 Additional Company Profiles (Upto 5 With No Cost)
Additional Company Profiles (Upto 5 With No Cost)
 Additional Countries (Apart From Mentioned Countries)
Additional Countries (Apart From Mentioned Countries)
 Country/Region-specific Report
Country/Region-specific Report
 Go To Market Strategy
Go To Market Strategy
 Region Specific Market Dynamics
Region Specific Market Dynamics Region Level Market Share
Region Level Market Share Import Export Analysis
Import Export Analysis Production Analysis
Production Analysis Others
Others