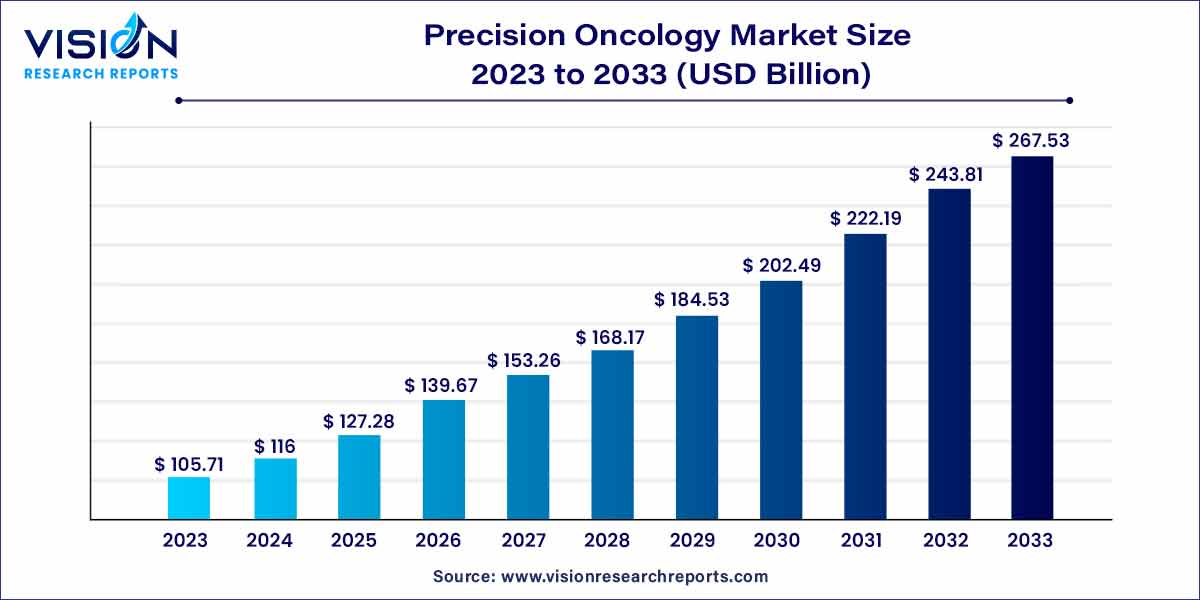
The global precision oncology market size was estimated at around USD 105.71 billion in 2023 and it is projected to hit around USD 267.53 billion by 2033, growing at a CAGR of 9.73% from 2024 to 2033.
Precision oncology is a rapidly evolving field at the forefront of cancer treatment, characterized by a personalized and targeted approach to patient care. This innovative approach involves tailoring medical interventions based on the specific genetic characteristics of an individual's cancer cells. The market overview of precision oncology encompasses various crucial components. In terms of product type, the market is segmented into diagnostics and therapeutics, with the therapeutics segment dominating in 2022, driven by the increasing approvals for tumor-agnostic therapies. Cancer type segmentation, including breast, colorectal, cervical, lung, prostate, and others, reflects the diverse applications of precision oncology across different malignancies.
The growth of the precision oncology market is propelled by several key factors. First and foremost, the increasing approvals for tumor-agnostic therapies within the therapeutics segment have played a pivotal role, contributing to a dominant market share. Additionally, the market benefits from the unique features of therapeutics tailored to specific proteins or mutations driving cancer's growth. Precision oncology-based therapies, emphasizing patient-specific cancer treatments based on individual DNA fingerprints, further drive market expansion. In the diagnostic tools segment, the market is stimulated by advancements in technology, leading to effective decision-making, optimized clinical outcomes, and a reduction in unnecessary therapies. The segmentation based on cancer type, such as breast cancer and cervical cancer, showcases a strategic alignment with the growing attention to screening, diagnosis, and treatment techniques. Furthermore, end-use segments like hospitals & diagnostic laboratories and pharmaceutical & biotechnology companies contribute significantly to market dominance, emphasizing the importance of precision oncology in these settings. Geographically, North America's dominance, driven by competitive dynamics and a burgeoning cancer population, and the anticipated rapid growth in Asia Pacific, fueled by a developed biotechnology sector and increasing awareness, further accentuate the overall positive trajectory of the precision oncology market.
| Report Coverage | Details |
| Market Revenue by 2033 | USD 267.53 billion |
| Growth Rate from 2024 to 2033 | CAGR of 9.73% |
| Revenue Share of North America in 2023 | 43% |
| Base Year | 2023 |
| Forecast Period | 2024 to 2033 |
| Market Analysis (Terms Used) | Value (US$ Million/Billion) or (Volume/Units) |
Due to increased technological emergence, the adaptive use of artificial intelligence and machine learning (AI & ML) in oncology research using genomic databases offers deeper understanding and ease for devising a therapeutic strategy, in comparison to the normal laboratory-based biochemical tests. For instance, in February 2022, Curesponse and Rafael entered into a collaboration for the utilization of artificial intelligence technology attributes on the precision oncology interface. The collaboration is intended to integrate Rafael's combat-proven machine learning capabilities with Curesponse's personalized oncology expertise to derive effective drug Response and Tumor Microenvironment (TME) to thereby yield a combined genomic-functional drug sensitivity profile.
Effective diagnostics is pivotal to oncology healthcare and clinical decisions. The shortfall of skilled researchers, outdated technology, and increased incidence of cancer cases, make the diagnostic procedures expensive and time consuming. For instance, in December 2020, Philips declared the introduction of advancements in its digital diagnostics at the European Congress of Pathology to support oncology clinical decision-making, by utilizing the power of genomics and imaging to yield individually predictable outcomes.
The potency profile of a drug candidate on potential tumor targets is crucial to gauge tumor inhibition effects on a broad range of cancer types that are highly aggressive. Pharmaceutical giants collaborate to study the efficacy of a drug candidate on the specific target group of cells. For instance, in February 2022, SEngine Precision Medicine Inc., and Oncodesign signed into a research collaboration to advance therapeutics for aggressively progressing tumors by using SEngine’s PARIS Test to pre-screen drugs on tumor cells extracted from biopsies of cancer survivors.
Cancer therapeutics are effectively designed based on the characteristics of cancer progression in an individual. Cancer conditions like breast cancer can be better personalized by prognosis and specific gene-targeted therapeutics. Numerous clinical trials are driving the need for precision oncology attributes to yield favorable outcomes. For instance, in April 2022, Reveal Genomics collaborated with Dana-Farber Cancer Institute for the evaluation of the HER2DX genomic test with three clinical trials to seek prospective clinical evidence so as to personalize the therapy and devise better clinical decisions to treat early-stage HER2+ breast cancer cases.
Precision oncology is increasingly trending due to numerous research studies that are supported by government and the research institutes in collaboration with pharmaceutical giants in the U.S. For instance, Lantern Pharma signed into an agreement with the Developmental Therapeutics Branch of the National Cancer Institute (NCI) to advance the identification of genetic markers for the prediction of a potential responses to Lantern's drug candidates, majorly in pancreatic cancers.
The precision oncology market is categorized into diagnostics and therapeutics based on product type. In 2023, the therapeutics segment held the largest revenue share of 72%. This growth can be attributed to various factors, including the increasing approvals for tumor-agnostic therapies. Furthermore, the unique attributes of therapeutics tailored to specific proteins or mutations driving cancer's progression have played a pivotal role. Precision oncology-based therapeutics offer personalized cancer treatments, aligning with the DNA fingerprint of each patient's cancer.
Conversely, the diagnostic tools segment is expected to expand at the highest CAGR of 10.42% from 2024 to 2033. Diagnostic products within this market assume a crucial role in verification and validation activities, with results intricately linked to the efficacy of the tests conducted. Advancements in technology have significantly enhanced the capabilities of diagnostic products, facilitating more informed decisions. This optimization of clinical outcomes not only reduces the reliance on unnecessary therapies but also minimizes side effects and mitigates the risk of drug resistance.
The precision oncology market is divided based on cancer type, encompassing breast cancer, colorectal cancer, cervical cancer, lung cancer, prostate cancer, and other types. As of 2023, breast cancer emerged as the leading segment, commanding the largest market share at 42%. This can be attributed to the escalating focus on breast cancer screening, diagnosis, as well as advancements in surgical and radiation techniques. The heightened research activities in early breast cancer detection and the overall prognosis of patients contribute to the development of innovative methods like precision oncology. Moreover, the rapid progress in the treatment of breast cancer patients is facilitated by new advanced technologies in DNA sequencing.
Concurrently, the cervical cancer segment is anticipated to grow at the noteworthy CAGR of 10.57% over the forecast period. This surge is fueled by increased research projects, funding initiatives, and product launches within the cervical cancer segment. For instance, the CDC's announcement of USD 215 million in funding awards across three national cancer programs has further stimulated market growth in cervical cancer research and development.
The market is categorized based on end-use into hospitals & diagnostic laboratories, pharmaceutical & biotechnology companies, and healthcare data companies. In 2023, the hospitals & diagnostic laboratories segment had the largest market share of 47%. Precision oncology products have been garnering increasing interest in hospitals and diagnostic laboratories globally, primarily due to their emphasis on molecular profiling of tumors to identify gene alterations. The market's significance in hospitals and diagnostic laboratories is underscored by its contributions to precision planning, accurate diagnosis, monitoring, and precise treatment, coupled with specialized expertise.
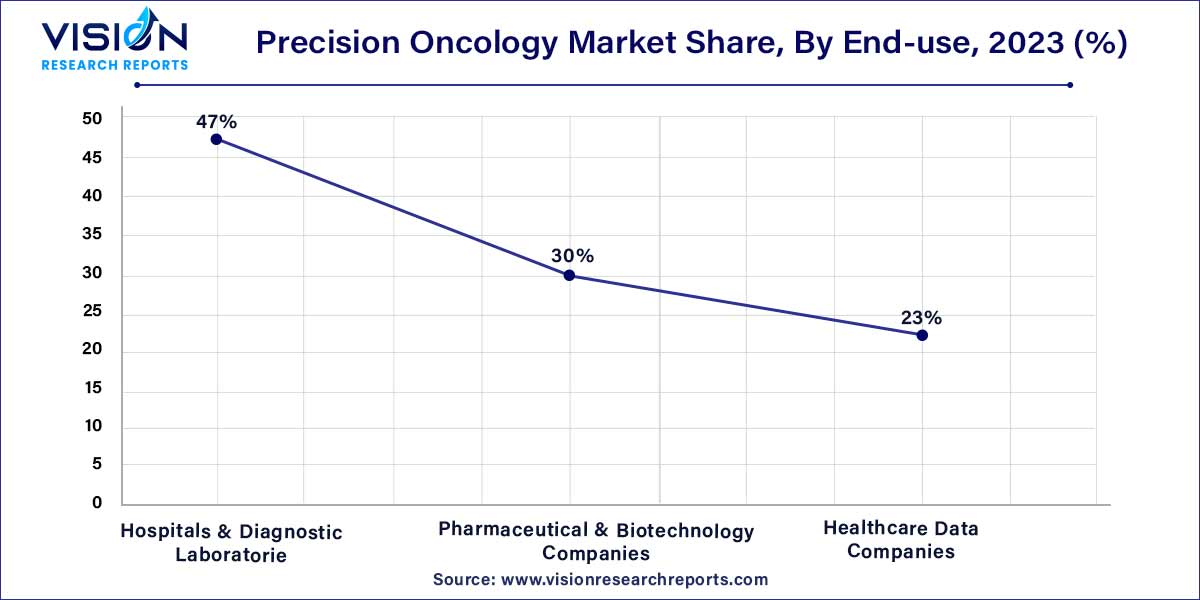
On the other hand, pharmaceutical & biotechnology companies are expected to experience a notable CAGR of 10.35% over the forecast period. These companies place a key focus on developing tailored end-to-end solutions to meet evolving customer requirements. The segment anticipates an uptick in large-scale projects supporting drug development programs as pharmaceutical and biotechnology companies strive to address the dynamic needs of the market.
In 2023, North America dominated the market with the largest market share of 43%. This region is marked by fierce competition, as key market players such as Thermo Fisher Scientific Inc., Invitae Corporation, Illumina, Inc., and Laboratory Corporation of America Holding operate prominently. The competitive landscape is further intensified by the prevalence of a significant cancer burden, numerous research activities, the development of innovative diagnostic software platforms, and a continual rise in healthcare expenditure. These factors collectively contribute to the creation of new growth opportunities within the precision oncology market in North America.
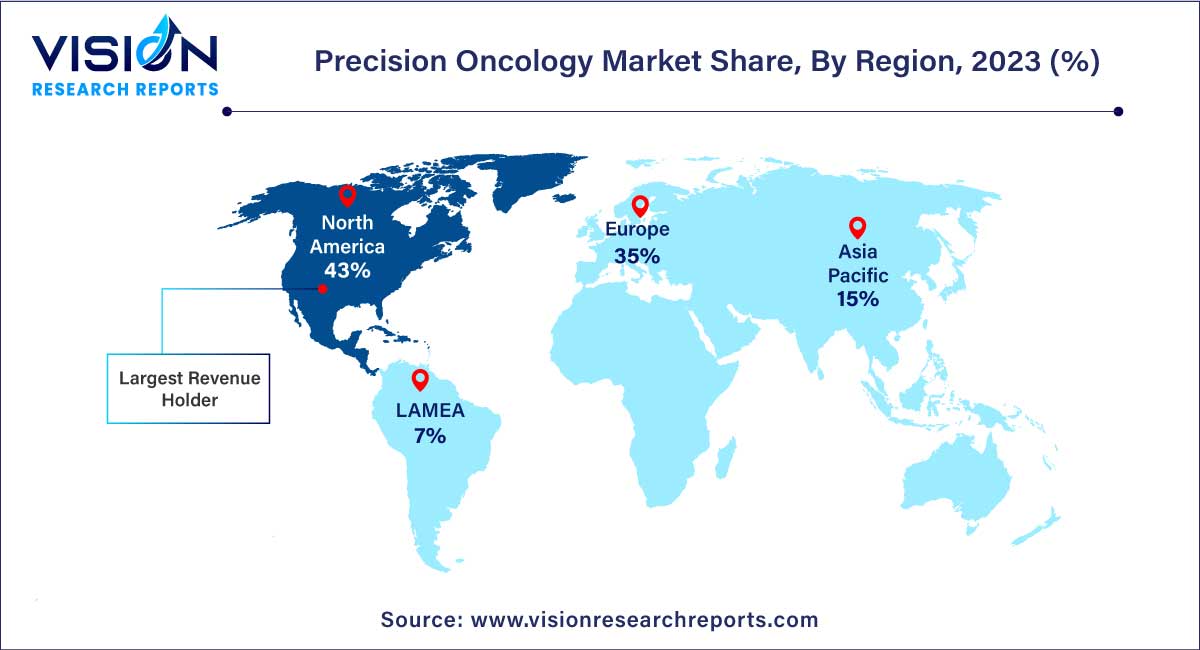
Concurrently, the Asia Pacific region is poised to witness the fastest CAGR of 10.93% over the forecast period. This accelerated growth can be attributed to the presence of a well-established biotechnology sector, ongoing developments in healthcare facilities, and a rising awareness of precision oncology. Notably, in August 2022, Bioserve Biotechnologies India Pvt. Ltd announced its receipt of accreditation from the National Accreditation Board for Testing and Calibration Laboratories for medical testing in genomics services. The accredited scope includes the BRCA 1 & 2 gene sequencing test, a service integral to precision oncology provided by the company. This accreditation reflects the region's commitment to advancing capabilities in the field of precision oncology.
By Product type
By Cancer type
By End use
By Region
Chapter 1. Introduction
1.1. Research Objective
1.2. Scope of the Study
1.3. Definition
Chapter 2. Research Methodology
2.1. Research Approach
2.2. Data Sources
2.3. Assumptions & Limitations
Chapter 3. Executive Summary
3.1. Market Snapshot
Chapter 4. Market Variables and Scope
4.1. Introduction
4.2. Market Classification and Scope
4.3. Industry Value Chain Analysis
4.3.1. Raw Material Procurement Analysis
4.3.2. Sales and Distribution Channel Analysis
4.3.3. Downstream Buyer Analysis
Chapter 5. COVID 19 Impact on Precision Oncology Market
5.1. COVID-19 Landscape: Precision Oncology Industry Impact
5.2. COVID 19 - Impact Assessment for the Industry
5.3. COVID 19 Impact: Global Major Government Policy
5.4. Market Trends and Opportunities in the COVID-19 Landscape
Chapter 6. Market Dynamics Analysis and Trends
6.1. Market Dynamics
6.1.1. Market Drivers
6.1.2. Market Restraints
6.1.3. Market Opportunities
6.2. Porter’s Five Forces Analysis
6.2.1. Bargaining power of suppliers
6.2.2. Bargaining power of buyers
6.2.3. Threat of substitute
6.2.4. Threat of new entrants
6.2.5. Degree of competition
Chapter 7. Competitive Landscape
7.1.1. Company Market Share/Positioning Analysis
7.1.2. Key Strategies Adopted by Players
7.1.3. Vendor Landscape
7.1.3.1. List of Suppliers
7.1.3.2. List of Buyers
Chapter 8. Global Precision Oncology Market, By Product type
8.1. Precision Oncology Market, by Product type, 2024-2033
8.1.1 Diagnostics
8.1.1.1. Market Revenue and Forecast (2021-2033)
8.1.2. Therapeutics
8.1.2.1. Market Revenue and Forecast (2021-2033)
Chapter 9. Global Precision Oncology Market, By Cancer type
9.1. Precision Oncology Market, by Cancer type, 2024-2033
9.1.1. Breast cancer
9.1.1.1. Market Revenue and Forecast (2021-2033)
9.1.2. Colorectal cancer
9.1.2.1. Market Revenue and Forecast (2021-2033)
9.1.3. Cervical cancer
9.1.3.1. Market Revenue and Forecast (2021-2033)
9.1.4. Prostate cancer
9.1.4.1. Market Revenue and Forecast (2021-2033)
9.1.5. Lung cancer
9.1.5.1. Market Revenue and Forecast (2021-2033)
9.1.5. Other cancers
9.1.5.1. Market Revenue and Forecast (2021-2033)
Chapter 10. Global Precision Oncology Market, By End use
10.1. Precision Oncology Market, by End use, 2024-2033
10.1.1. Hospitals & Diagnostic Laboratories
10.1.1.1. Market Revenue and Forecast (2021-2033)
10.1.2. Pharmaceutical & Biotechnology companies
10.1.2.1. Market Revenue and Forecast (2021-2033)
10.1.3. Healthcare data companies
10.1.3.1. Market Revenue and Forecast (2021-2033)
Chapter 11. Global Precision Oncology Market, Regional Estimates and Trend Forecast
11.1. North America
11.1.1. Market Revenue and Forecast, by Product type (2021-2033)
11.1.2. Market Revenue and Forecast, by Cancer type (2021-2033)
11.1.3. Market Revenue and Forecast, by End use (2021-2033)
11.1.4. U.S.
11.1.4.1. Market Revenue and Forecast, by Product type (2021-2033)
11.1.4.2. Market Revenue and Forecast, by Cancer type (2021-2033)
11.1.4.3. Market Revenue and Forecast, by End use (2021-2033)
11.1.5. Rest of North America
11.1.5.1. Market Revenue and Forecast, by Product type (2021-2033)
11.1.5.2. Market Revenue and Forecast, by Cancer type (2021-2033)
11.1.5.3. Market Revenue and Forecast, by End use (2021-2033)
11.2. Europe
11.2.1. Market Revenue and Forecast, by Product type (2021-2033)
11.2.2. Market Revenue and Forecast, by Cancer type (2021-2033)
11.2.3. Market Revenue and Forecast, by End use (2021-2033)
11.2.4. UK
11.2.4.1. Market Revenue and Forecast, by Product type (2021-2033)
11.2.4.2. Market Revenue and Forecast, by Cancer type (2021-2033)
11.2.4.3. Market Revenue and Forecast, by End use (2021-2033)
11.2.5. Germany
11.2.5.1. Market Revenue and Forecast, by Product type (2021-2033)
11.2.5.2. Market Revenue and Forecast, by Cancer type (2021-2033)
11.2.5.3. Market Revenue and Forecast, by End use (2021-2033)
11.2.6. France
11.2.6.1. Market Revenue and Forecast, by Product type (2021-2033)
11.2.6.2. Market Revenue and Forecast, by Cancer type (2021-2033)
11.2.6.3. Market Revenue and Forecast, by End use (2021-2033)
11.2.7. Rest of Europe
11.2.7.1. Market Revenue and Forecast, by Product type (2021-2033)
11.2.7.2. Market Revenue and Forecast, by Cancer type (2021-2033)
11.2.7.3. Market Revenue and Forecast, by End use (2021-2033)
11.3. APAC
11.3.1. Market Revenue and Forecast, by Product type (2021-2033)
11.3.2. Market Revenue and Forecast, by Cancer type (2021-2033)
11.3.3. Market Revenue and Forecast, by End use (2021-2033)
11.3.4. India
11.3.4.1. Market Revenue and Forecast, by Product type (2021-2033)
11.3.4.2. Market Revenue and Forecast, by Cancer type (2021-2033)
11.3.4.3. Market Revenue and Forecast, by End use (2021-2033)
11.3.5. China
11.3.5.1. Market Revenue and Forecast, by Product type (2021-2033)
11.3.5.2. Market Revenue and Forecast, by Cancer type (2021-2033)
11.3.5.3. Market Revenue and Forecast, by End use (2021-2033)
11.3.6. Japan
11.3.6.1. Market Revenue and Forecast, by Product type (2021-2033)
11.3.6.2. Market Revenue and Forecast, by Cancer type (2021-2033)
11.3.6.3. Market Revenue and Forecast, by End use (2021-2033)
11.3.7. Rest of APAC
11.3.7.1. Market Revenue and Forecast, by Product type (2021-2033)
11.3.7.2. Market Revenue and Forecast, by Cancer type (2021-2033)
11.3.7.3. Market Revenue and Forecast, by End use (2021-2033)
11.4. MEA
11.4.1. Market Revenue and Forecast, by Product type (2021-2033)
11.4.2. Market Revenue and Forecast, by Cancer type (2021-2033)
11.4.3. Market Revenue and Forecast, by End use (2021-2033)
11.4.4. GCC
11.4.4.1. Market Revenue and Forecast, by Product type (2021-2033)
11.4.4.2. Market Revenue and Forecast, by Cancer type (2021-2033)
11.4.4.3. Market Revenue and Forecast, by End use (2021-2033)
11.4.5. North Africa
11.4.5.1. Market Revenue and Forecast, by Product type (2021-2033)
11.4.5.2. Market Revenue and Forecast, by Cancer type (2021-2033)
11.4.5.3. Market Revenue and Forecast, by End use (2021-2033)
11.4.6. South Africa
11.4.6.1. Market Revenue and Forecast, by Product type (2021-2033)
11.4.6.2. Market Revenue and Forecast, by Cancer type (2021-2033)
11.4.6.3. Market Revenue and Forecast, by End use (2021-2033)
11.4.7. Rest of MEA
11.4.7.1. Market Revenue and Forecast, by Product type (2021-2033)
11.4.7.2. Market Revenue and Forecast, by Cancer type (2021-2033)
11.4.7.3. Market Revenue and Forecast, by End use (2021-2033)
11.5. Latin America
11.5.1. Market Revenue and Forecast, by Product type (2021-2033)
11.5.2. Market Revenue and Forecast, by Cancer type (2021-2033)
11.5.3. Market Revenue and Forecast, by End use (2021-2033)
11.5.4. Brazil
11.5.4.1. Market Revenue and Forecast, by Product type (2021-2033)
11.5.4.2. Market Revenue and Forecast, by Cancer type (2021-2033)
11.5.4.3. Market Revenue and Forecast, by End use (2021-2033)
11.5.5. Rest of LATAM
11.5.5.1. Market Revenue and Forecast, by Product type (2021-2033)
11.5.5.2. Market Revenue and Forecast, by Cancer type (2021-2033)
11.5.5.3. Market Revenue and Forecast, by End use (2021-2033)
Chapter 12. Company Profiles
12.1. F Hoffmann-La Roche AG
12.1.1. Company Overview
12.1.2. Product Offerings
12.1.3. Financial Performance
12.1.4. Recent Initiatives
12.2. Thermo Fisher Scientific Inc.
12.2.1. Company Overview
12.2.2. Product Offerings
12.2.3. Financial Performance
12.2.4. Recent Initiatives
12.3. Invitae Corporation
12.3.1. Company Overview
12.3.2. Product Offerings
12.3.3. Financial Performance
12.3.4. Recent Initiatives
12.4. Bristol-Myers Squibb
12.4.1. Company Overview
12.4.2. Product Offerings
12.4.3. Financial Performance
12.4.4. Recent Initiatives
12.5. QIAGEN
12.5.1. Company Overview
12.5.2. Product Offerings
12.5.3. Financial Performance
12.5.4. Recent Initiatives
12.6. Illumina, Inc.
12.6.1. Company Overview
12.6.2. Product Offerings
12.6.3. Financial Performance
12.6.4. Recent Initiatives
12.7. Quest Diagnostics
12.7.1. Company Overview
12.7.2. Product Offerings
12.7.3. Financial Performance
12.7.4. Recent Initiatives
12.8. Novartis AG
12.8.1. Company Overview
12.8.2. Product Offerings
12.8.3. Financial Performance
12.8.4. Recent Initiatives
12.9. AstraZeneca
12.9.1. Company Overview
12.9.2. Product Offerings
12.9.3. Financial Performance
12.9.4. Recent Initiatives
12.10. Laboratory Corporation of America Holdings
12.10.1. Company Overview
12.10.2. Product Offerings
12.10.3. Financial Performance
12.10.4. Recent Initiatives
Chapter 13. Research Methodology
13.1. Primary Research
13.2. Secondary Research
13.3. Assumptions
Chapter 14. Appendix
14.1. About Us
14.2. Glossary of Terms
 Cross-segment Market Size and Analysis for
Mentioned Segments
Cross-segment Market Size and Analysis for
Mentioned Segments
 Additional Company Profiles (Upto 5 With No Cost)
Additional Company Profiles (Upto 5 With No Cost)
 Additional Countries (Apart From Mentioned Countries)
Additional Countries (Apart From Mentioned Countries)
 Country/Region-specific Report
Country/Region-specific Report
 Go To Market Strategy
Go To Market Strategy
 Region Specific Market Dynamics
Region Specific Market Dynamics Region Level Market Share
Region Level Market Share Import Export Analysis
Import Export Analysis Production Analysis
Production Analysis Others
Others