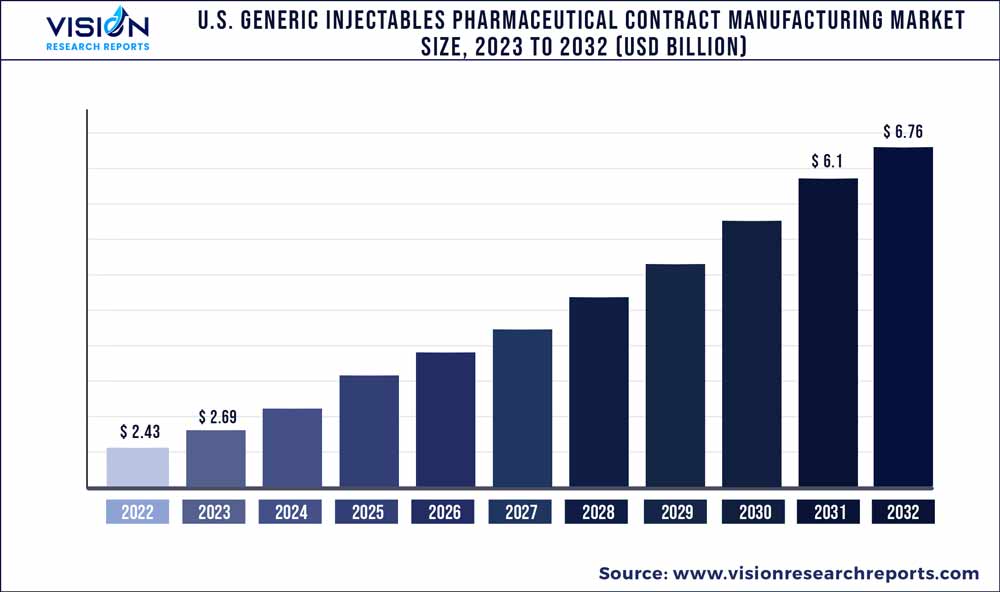
The U.S. generic injectables pharmaceutical contract manufacturing market was estimated at USD 2.43 billion in 2022 and it is expected to surpass around USD 6.76 billion by 2032, poised to grow at a CAGR of 10.77% from 2023 to 2032.
Key Pointers
Report Scope of the U.S. Generic Injectables Pharmaceutical Contract Manufacturing Market
| Report Coverage | Details |
| Market Size in 2022 | USD 2.43 billion |
| Revenue Forecast by 2032 | USD 6.76 billion |
| Growth rate from 2023 to 2032 | CAGR of 10.77% |
| Base Year | 2022 |
| Forecast Period | 2023 to 2032 |
| Market Analysis (Terms Used) | Value (US$ Million/Billion) or (Volume/Units) |
| Companies Covered | Hikma Pharmaceuticals plc; Pfizer Inc.; Fresenius Kabi; Sandoz AG; Jubilant Pharma Limited; Baxter; PCI Pharma Services; Gland Pharma Limited (USA); Dr. Reddy’s Laboratories Ltd.; Grand River Aseptic Manufacturing |
The rising frequency of injectable medications losing patent protection is one of the primary factors supporting the growth of the generic injectables pharmaceutical contract manufacturing space. For instance, in March 2022, Takeda Pharmaceutical Company Limited's blockbuster medication Velcade (bortezomib) saw its patent exclusivity in the U.S. expire, leading to the introduction of generic alternatives in the same year.
Furthermore, the manufacturing and supply of injectables are specialized and capital-intensive. Injectables require dedicated manufacturing lines, which can cost around USD 30-35 million. Owing to the aforementioned factors, the contract manufacturing of generic injectables is gaining traction, thus augmenting the growth of the U.S. market for generic injectables pharmaceutical contract manufacturing.
The COVID-19 pandemic significantly impacted the pharmaceutical industry, including the sector of generic injectables. It disrupted global supply chains, leading to a shortage of pharmaceutical ingredients and raw materials. This affected the manufacturing process of generic injectables, leading to potential supply shortages, excluding the injectables for COVID-19 treatment. Regulatory bodies in the U.S. prioritized the review of medical products and drugs pertaining to COVID-19 treatment, potentially delaying the approval of new generic injectables. Furthermore, economic uncertainties led to cost and pricing pressures in the pharmaceutical sector, thus affecting the pricing of generic injectables.
Geopolitical wars, crises, and conflicts profoundly and multifacetedly have impacted the generic injectables industry. However, the U.S. generic injectables pharmaceutical contract manufacturing sector managed to recover and achieve substantial sales growth in 2022 and 2023, largely driven by the expiration of patents for several blockbuster drugs during this period. In July 2023, the patent for Sanofi's highly successful medication Mozobil expired, leading to the introduction of generic versions of Plerixafor.
Molecule Type Insights
The large molecule segment held the dominant revenue share of 62% in 2022. The growing emphasis on tailored treatments and precision medicine is a major factor augmenting the growth of the large molecule generic injectable drugs segment in the U.S. Moreover, increasing product launches including biosimilars is a major factor supporting segment growth. For instance, in January 2023, Amgen announced the launch of AMJEVITA, a biosimilar to Humira, in the U.S. AMJEVITA was the first generic version of Humira approved by the U.S. FDA. Such factors are poised to support segment growth.
The small molecule segment is anticipated to register a stable CAGR of 10.53% during the forecast period. Small molecule-based injectable drugs continue to hold an essential position in the treatment of a wide range of illnesses, including cancer, blood disorders, infectious diseases, and cardiovascular ailments. The high growth of the segment is majorly due to the increasing rate of generics being launched across the U.S. and, concurrently, several contract manufacturers garnering profits by entering the manufacturing business of generic injectables. This is one of the main reasons supporting segment expansion across the forecast period.
Application Insights
The oncology segment held the largest revenue share of 29% in 2022 in the U.S. generic injectables pharmaceutical contract manufacturing market. This is due to increasing generic product launches pertaining to cancer treatment. A considerable number of contract manufacturers are focusing on the development of generic injectables for anti-cancer treatment therapy. Additionally, a substantial pipeline of generic products for anti-cancer therapies is in development, with numerous generic drugs set to be introduced to the market.
For instance, from 2007 to 2020, the U.S. Food and Drug Administration (FDA) approved 16 oncology biosimilars. It is anticipated that this number will continue to increase in the forecast period, contributing to the expansion of this segment. Furthermore, the growing prevalence of the condition further supports research and development activities pertaining to abbreviated new drug application (ANDA) of biosimilars for anti-cancer therapeutics.
On the other hand, the neurology segment is expected to register the fastest CAGR of 11.36% during the forecast period. This is mainly due to the rise in demand for biosimilars to treat several neurological conditions. Moreover, large scale contract manufacturers are focusing on the development of neurology-based biosimilars, thereby supporting the segment’s growth.
For instance, in April 2023, Teva Pharmaceuticals and MedinCell announced that the U.S. FDA had granted approval for UZEDY (risperidone) extended-release injectable suspension for treating schizophrenia in adults. Additionally, the incidence of neurological disorders such as Alzheimer's disease and Parkinson's disease has been steadily increasing over the past decade, which is further anticipated to support the growth of this segment.
U.S. Generic Injectables Pharmaceutical Contract Manufacturing Market Segmentations:
By Molecule Type
By Application
 Cross-segment Market Size and Analysis for
Mentioned Segments
Cross-segment Market Size and Analysis for
Mentioned Segments
 Additional Company Profiles (Upto 5 With No Cost)
Additional Company Profiles (Upto 5 With No Cost)
 Additional Countries (Apart From Mentioned Countries)
Additional Countries (Apart From Mentioned Countries)
 Country/Region-specific Report
Country/Region-specific Report
 Go To Market Strategy
Go To Market Strategy
 Region Specific Market Dynamics
Region Specific Market Dynamics Region Level Market Share
Region Level Market Share Import Export Analysis
Import Export Analysis Production Analysis
Production Analysis Others
Others