The U.S. non invasive prenatal testing market size was estimated at USD 1.77 billion in 2023 and it is expected to surpass around USD 3.91 billion by 2033, poised to grow at a CAGR of 8.22% from 2024 to 2033.
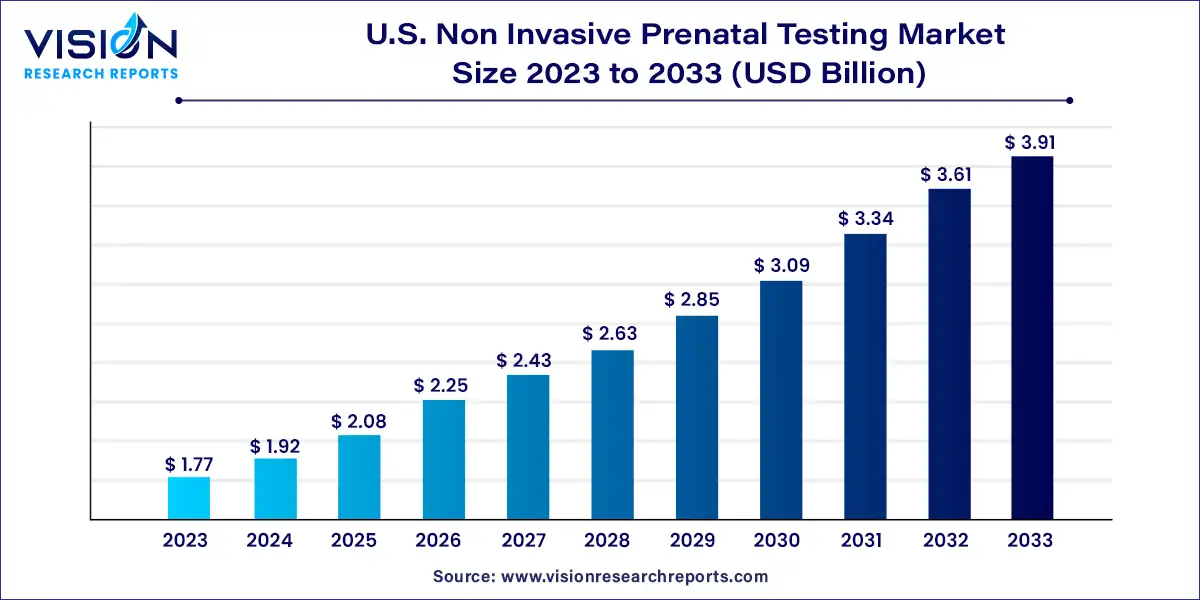
The U.S. non-invasive prenatal testing (NIPT) market has witnessed significant growth in recent years, driven by advancements in prenatal screening technologies, rising maternal age, and increasing awareness about the benefits of non-invasive testing methods.
The growth of the U.S. non-invasive prenatal testing (NIPT) market is propelled by the trend of increasing maternal age contributes significantly, as older mothers are at a higher risk of chromosomal abnormalities in their fetuses, necessitating early and accurate prenatal screening. Additionally, continuous technological advancements in NIPT methods enhance the accuracy, sensitivity, and specificity of testing, making them more reliable for detecting chromosomal abnormalities. Furthermore, heightened awareness among healthcare providers and patients about the benefits of NIPT, including reduced risk of miscarriage and invasive procedures, drives demand. Lastly, favorable reimbursement policies from government and private payers make NIPT procedures more accessible to a broader population, further fueling market growth.
| Report Coverage | Details |
| Market Size in 2023 | USD 1.77 billion |
| Revenue Forecast by 2033 | USD 3.91 billion |
| Growth rate from 2024 to 2033 | CAGR of 8.22% |
| Base Year | 2023 |
| Forecast Period | 2024 to 2033 |
| Market Analysis (Terms Used) | Value (US$ Million/Billion) or (Volume/Units) |
The 13-24 weeks segment dominated the U.S. NIPT market in 2023 with a revenue share of 52%. As the maximum number of NIPT procedures are performed in the second trimester. Several companies have focused on the analysis of cfDNA in a sample of maternal blood collected in the first trimester to develop a more accurate and reliable NIPT. The market's expansion is anticipated to be fueled by the complementary use of ultrasonography with alpha-fetoprotein testing and NIPT beyond 12 weeks of pregnancy. Additionally, tests used in quad screening, including Alpha-Fetoprotein (AFP), Unconjugated Estriol (EU), Human Chorionic Gonadotropin (hCG), and Inhibin A, are helping to boost this segment's income.
The 0-12 weeks segmentis expected to showcase lucrative growth during the forecast period due to a wide range of products being offered in the market. A considerable amount of income is generated in this segment as a result of the first-trimester aneuploidy testing and maternal-fetal DNA screening performed during this time. Additionally, the combined information from biochemistry and sonography can be used to detect genetic changes with an accuracy level of 91% to 96%, making first-trimester risk screening preferable to other trimesters.
The high & average pregnancy risk segment dominated the U.S. non invasive prenatal testing market in 2023 with a revenue share of 78% owing to the wide adoption of NIPT in this risk segment. Moreover, the high-risk nature of pregnancies in women aged over 35 years is anticipated to drive the segment. The available products in the market provide sensitivity within the range of 99.0% to 99.9% for detecting Down syndrome. Additionally, extended health coverage and plans for NIPT drive the market growth.
For instance, in October 2020, Natera, Inc. A prominent health plan announced an extension of its coverage to all singleton pregnancies, adding 17 million new covered lives. According to the most recent round of medical policy revisions in 2020, the number of covered lives for average risk NIPT has more than doubled. According to Natera, 139 million commercial lives-or nearly 77% of covered lives-are currently covered in the United States for average risk NIPT.
The low-risk segment is expected to register the fastest CAGR during the forecast period. Support from the government, such as budget assignment for average-risk pregnancies, is expected to be a favorable factor for growth. For instance, as per the reports of the National Centre for Biotechnology Information published in the year 2019, testing in pregnancies with average risk made it feasible to find an increased percentage of afflicted cases, leading to an increase in funding for this group in Ontario of USD 35 million.
The cell-free DNA in maternal plasma tests segment dominated the U.S. NIPT market in 2023 with a revenue share of 71%. Wide acceptance, high cost of tests, and rapid adoption of tests that detect circulating biomarkers, such as cell-free DNA fragments, have contributed to the largest market share held by this segment. Moreover, rapid technological advancements and the launch of new products in the segment are likely to have a positive impact on market growth.
For instance, in September 2019, Laboratory Corporation of America Holdings published findings from the largest cell-free chromosomal DNA screening investigation in a multifetal pregnancy. The research, which was published in PLOS ONE, found that non invasive cell-free DNA testing utilizing the MaterniT21 PLUS test produced precise results that were comparable to those for singleton pregnancies.
Biochemical tests are projected to grow at a lucrative rate. They are conducted at 8 to 24 weeks of pregnancy. However, the adoption rate varies due to the accuracy’s dependency on the mother’s health, especially in conditions such as obesity, which can hinder test results. However, the adoption rate for these tests is still high. For instance, 75% to 90% of fetuses with defects in the neural tube are detected by alpha-fetoprotein testing. The wide adoption rate is anticipated to act as a driver for the segment.
The others segment which include rolling cycle amplification, karyotyping, Sanger sequencing, ultrasound scanning, and other blood tests held the maximum revenue share of 55% in 2023. A companion test to the cell-free DNA-based NIPT is ultrasound detection. Massively parallel shotgun sequencing, digital PCR, and microarray-based techniques are the other methods included in NIPT. Also, the advancements in ultrasound detection techniques and 3D-4D imaging have improved real-time monitoring, safety, and efficiency of the test, which is further expected to propel the segment growth.
The NGS segment is the fastest-growing segment. Currently, most of the commercially approved products are based on whole exome and whole genome sequencing to detect chromosomal anomalies. Furthermore, expanding the technology for detecting monogenic diseases is likely to boost revenue in this segment. It is frequently used to detect chromosomal aneuploidies, microdeletions, and trisomy disorders. As opposed to other methods, NGS-based noninvasive prenatal diagnostics can be carried out as early as 10 weeks of pregnancy. Additionally, NGS-based assays are more accurate than other noninvasive prenatal diagnostics, which have an accuracy rate of above 99%.
The consumables and reagents segment dominated the U.S. NIPT market with a share of over 73% in 2023. Illumina Inc.; F. Hoffmann-La Roche Ltd.; Natera, Inc.; and other regional players offer a wide range of consumables & reagents for NIPT. These players offer various kits, consumables, reagents & instruments to perform prenatal tests. For instance, in August 2020, the Society for Maternal-Fetal Medicine and the American College of Obstetricians and Gynecologists released joint guidelines recommending that prenatal screening to detect fetal chromosomal abnormalities be offered to all pregnant women irrespective of their baseline risk & maternal age.
The consumables and reagents segment is also projected to grow at the fastest CAGR during the forecast period. The market for consumables and reagents in NIPT is impacted by a number of variables, including supplier competition, regulatory restrictions, and technical improvements. Companies operating in this market concentrate on creating consumables and reagents that are of the highest caliber, most dependable, and easiest to use to satisfy the needs of NIPT laboratories and healthcare providers.
The trisomy segment dominated the U.S. non-invasive prenatal testing market with a share of over 55% in 2023. Growing incidence of trisomy, increasing awareness among people, and favorable initiatives undertaken by government bodies are key factors propelling the segment’s growth. High diagnosis rates, widespread acceptance, and public awareness, as well as the launch of novel and cutting-edge tests, are further factors driving market expansion in the United States.
For instance, in May 2021, in addition to the current IONA test, which provides screening for trisomies 21, 18, and 13, as well as fetal sex determination, Yourgene established IONA Care, a non invasive prenatal test service. This test can determine whether a pregnant woman is carrying a fetus with autosomal aneuploidies and sex chromosome aneuploidies. Therefore, during the projection period, such product introductions are anticipated to boost market growth.
However, the microdeletion syndrome segment is anticipated to grow at the fastest rate over the forecast period. To identify specific microdeletions linked to particular syndromes, NIPT for microdeletion syndromes examines the mother's blood for cell-free DNA (cfDNA). The NIPT can be used to screen for a number of microdeletion syndromes, including the DiGeorge syndrome, Cri-du-chat syndrome, Angelman syndrome, Prader-Willi syndrome, and Wolf-Hirschhorn syndrome.
The scope of prenatal genetic screening has been broadened with the inclusion of microdeletion syndromes in NIPT, enabling expecting parents to acquire more thorough information about the genetic health of their child. Even though NIPT can detect certain diseases, confirmation of the diagnosis frequently necessitates further diagnostic procedures, such as fluorescence in situ hybridization (FISH) or chromosomal microarray analysis.
The diagnostic laboratories segment dominated the U.S. NIPT market with a share of over 63% in 2023, attributable to numerous diagnostic facilities offering NIPT all over the world. For the diagnosis of Trisomies 21, 18, and 13, Monosomy X, and other sex chromosomal abnormalities, laboratories like MedGenome Labs Ltd. offer MedGenome Claria NIPT testing. Similar to this, MedGenome's Claria NIPT Plus can identify Angelman syndrome, Edwards' syndrome, Down syndrome, 1p36 deletion syndrome, Klinefelter syndrome, Triple X, triploidy, monosomy X (Turner syndrome), Jacob's syndrome, 22q11.2 deletion syndrome, Prader-Willi syndrome, Patau syndrome, and Cri-du-chat syndrome.
Key developers, such as Illumina, are outsourcing their sample processing to Illumina CLIA labs when in-house facilities are not sufficient. Moreover, laboratories conducting NIPT tests across the U.S. are operated in compliance with quality-assurance regulations to safeguard test quality and reproducibility.
The diagnostic laboratories segment is anticipated to grow at the fastest CAGR over the forecast period. The NIPT market has experienced rapid growth, and diagnostic laboratories are essential to providing and carrying out NIPT services. These labs offer the tools, personnel, and technology required to conduct the screening test and provide accurate findings to medical professionals and pregnant parents.
By Gestation Period
By Risk
By Method
By Technology
By Product
By Application
By End-use
Chapter 1. Introduction
1.1. Research Objective
1.2. Scope of the Study
1.3. Definition
Chapter 2. Research Methodology
2.1. Research Approach
2.2. Data Sources
2.3. Assumptions & Limitations
Chapter 3. Executive Summary
3.1. Market Snapshot
Chapter 4. Market Variables and Scope
4.1. Introduction
4.2. Market Classification and Scope
4.3. Industry Value Chain Analysis
4.3.1. Raw Material Procurement Analysis
4.3.2. Sales and Distribution Channel Analysis
4.3.3. Downstream Buyer Analysis
Chapter 5. Market Dynamics Analysis and Trends
5.1. Market Dynamics
5.1.1. Market Drivers
5.1.2. Market Restraints
5.1.3. Market Opportunities
5.2. Porter’s Five Forces Analysis
5.2.1. Bargaining power of suppliers
5.2.2. Bargaining power of buyers
5.2.3. Threat of substitute
5.2.4. Threat of new entrants
5.2.5. Degree of competition
Chapter 6. Competitive Landscape
6.1.1. Company Market Share/Positioning Analysis
6.1.2. Key Strategies Adopted by Players
6.1.3. Vendor Landscape
6.1.3.1. List of Suppliers
6.1.3.2. List of Buyers
Chapter 7. U.S. Non Invasive Prenatal Testing Market, By Gestation Period
7.1. U.S. Non Invasive Prenatal Testing Market, by Gestation Period, 2024-2033
7.1.1. 0-12 weeks
7.1.1.1. Market Revenue and Forecast (2021-2033)
7.1.2. 13-24 weeks
7.1.2.1. Market Revenue and Forecast (2021-2033)
7.1.3. 25-35 weeks
7.1.3.1. Market Revenue and Forecast (2021-2033)
Chapter 8. U.S. Non Invasive Prenatal Testing Market, By Risk
8.1. U.S. Non Invasive Prenatal Testing Market, by Risk, 2024-2033
8.1.1. High & Average Risk
8.1.1.1. Market Revenue and Forecast (2021-2033)
8.1.2. Low Risk
8.1.2.1. Market Revenue and Forecast (2021-2033)
Chapter 9. U.S. Non Invasive Prenatal Testing Market, By Method
9.1. U.S. Non Invasive Prenatal Testing Market, by Method, 2024-2033
9.1.1. Biochemical Screening Tests
9.1.1.1. Market Revenue and Forecast (2021-2033)
9.1.2. Cell-free DNA in Maternal Plasma Tests
9.1.2.1. Market Revenue and Forecast (2021-2033)
Chapter 10. U.S. Non Invasive Prenatal Testing Market, By Technology
10.1. U.S. Non Invasive Prenatal Testing Market, by Technology, 2024-2033
10.1.1. NGS
10.1.1.1. Market Revenue and Forecast (2021-2033)
10.1.2. Array Technology
10.1.2.1. Market Revenue and Forecast (2021-2033)
10.1.3. PCR
10.1.3.1. Market Revenue and Forecast (2021-2033)
10.1.4. Others
10.1.4.1. Market Revenue and Forecast (2021-2033)
Chapter 11. U.S. Non Invasive Prenatal Testing Market, By Product
11.1. U.S. Non Invasive Prenatal Testing Market, by Product, 2024-2033
11.1.1. Consumables & Reagents
11.1.1.1. Market Revenue and Forecast (2021-2033)
11.1.2. Instruments
11.1.2.1. Market Revenue and Forecast (2021-2033)
Chapter 12. U.S. Non Invasive Prenatal Testing Market, By Application
12.1. U.S. Non Invasive Prenatal Testing Market, by Application, 2024-2033
12.1.1. Trisomy
12.1.1.1. Market Revenue and Forecast (2021-2033)
12.1.2. Microdeletion Syndrome
12.1.2.1. Market Revenue and Forecast (2021-2033)
12.1.3. Other Applications
12.1.3.1. Market Revenue and Forecast (2021-2033)
Chapter 13. U.S. Non Invasive Prenatal Testing Market, By End-use
13.1. U.S. Non Invasive Prenatal Testing Market, by End-use, 2024-2033
13.1.1. Hospitals & Clinics
13.1.1.1. Market Revenue and Forecast (2021-2033)
13.1.2. Diagnostic Laboratories
13.1.2.1. Market Revenue and Forecast (2021-2033)
Chapter 14. U.S. Non Invasive Prenatal Testing Market, Regional Estimates and Trend Forecast
14.1. U.S.
14.1.1. Market Revenue and Forecast, by Gestation Period (2021-2033)
14.1.2. Market Revenue and Forecast, by Risk (2021-2033)
14.1.3. Market Revenue and Forecast, by Method (2021-2033)
14.1.4. Market Revenue and Forecast, by Technology (2021-2033)
14.1.5. Market Revenue and Forecast, by Product (2021-2033)
14.1.6. Market Revenue and Forecast, by End-use (2021-2033)
14.1.7. Market Revenue and Forecast, by Application (2021-2033)
Chapter 15. Company Profiles
15.1. Laboratory Corporation of America Holdings
15.1.1. Company Overview
15.1.2. Product Offerings
15.1.3. Financial Performance
15.1.4. Recent Initiatives
15.2. Illumina, Inc.
15.2.1. Company Overview
15.2.2. Product Offerings
15.2.3. Financial Performance
15.2.4. Recent Initiatives
15.3. Natera, Inc.
15.3.1. Company Overview
15.3.2. Product Offerings
15.3.3. Financial Performance
15.3.4. Recent Initiatives
15.4. QIAGEN
15.4.1. Company Overview
15.4.2. Product Offerings
15.4.3. Financial Performance
15.4.4. Recent Initiatives
15.5. Ariosa Diagnostics (Roche)
15.5.1. Company Overview
15.5.2. Product Offerings
15.5.3. Financial Performance
15.5.4. Recent Initiatives
15.6. Myriad Women’s Health, Inc.
15.6.1. Company Overview
15.6.2. Product Offerings
15.6.3. Financial Performance
15.6.4. Recent Initiatives
15.7. Biora Therapeutics, Inc.
15.7.1. Company Overview
15.7.2. Product Offerings
15.7.3. Financial Performance
15.7.4. Recent Initiatives
15.8. Quest Diagnostics Incorporated
15.8.1. Company Overview
15.8.2. Product Offerings
15.8.3. Financial Performance
15.8.4. Recent Initiatives
15.9. Eurofins Scientific
15.9.1. Company Overview
15.9.2. Product Offerings
15.9.3. Financial Performance
15.9.4. Recent Initiatives
15.10. BioReference Health, LLC (Subsidiary of OPKO Health, Inc.)
15.10.1. Company Overview
15.10.2. Product Offerings
15.10.3. Financial Performance
15.10.4. Recent Initiatives
Chapter 16. Research Methodology
16.1. Primary Research
16.2. Secondary Research
16.3. Assumptions
Chapter 17. Appendix
17.1. About Us
17.2. Glossary of Terms
 Cross-segment Market Size and Analysis for
Mentioned Segments
Cross-segment Market Size and Analysis for
Mentioned Segments
 Additional Company Profiles (Upto 5 With No Cost)
Additional Company Profiles (Upto 5 With No Cost)
 Additional Countries (Apart From Mentioned Countries)
Additional Countries (Apart From Mentioned Countries)
 Country/Region-specific Report
Country/Region-specific Report
 Go To Market Strategy
Go To Market Strategy
 Region Specific Market Dynamics
Region Specific Market Dynamics Region Level Market Share
Region Level Market Share Import Export Analysis
Import Export Analysis Production Analysis
Production Analysis Others
Others