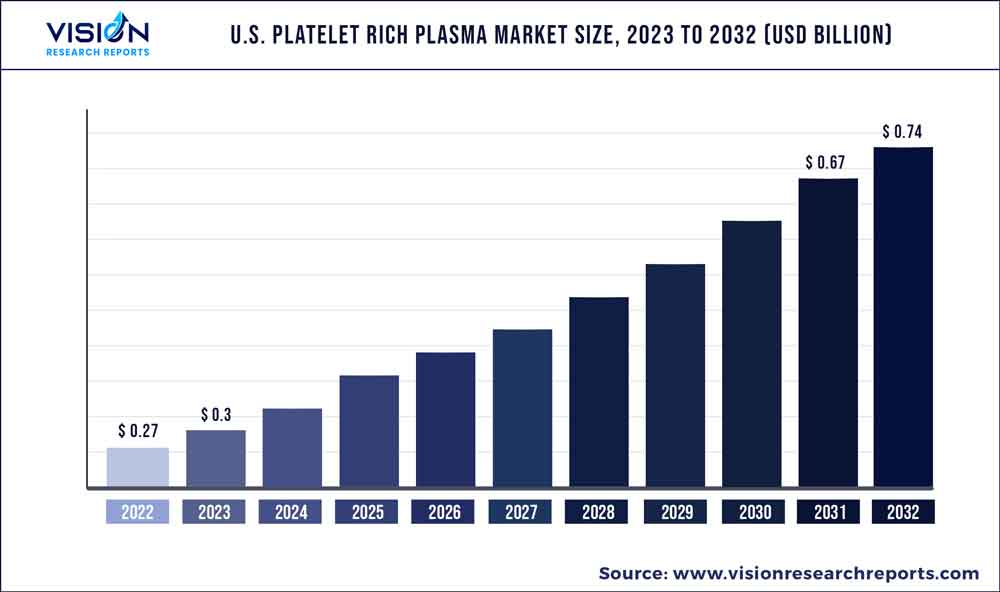
The U.S. platelet rich plasma market size was estimated at around USD 0.27 billion in 2022 and it is projected to hit around USD 0.74 billion by 2032, growing at a CAGR of 10.54% from 2023 to 2032.
Key Pointers
Report Scope of the U.S. Platelet Rich Plasma Market
| Report Coverage | Details |
| Market Size in 2022 | USD 0.27 billion |
| Revenue Forecast by 2032 | USD 0.74 billion |
| Growth rate from 2023 to 2032 | CAGR of 10.54% |
| Base Year | 2022 |
| Forecast Period | 2023 to 2032 |
| Market Analysis (Terms Used) | Value (US$ Million/Billion) or (Volume/Units) |
| Companies Covered | Johnson & Johnson Services, Inc; Arthrex, Inc.; EmCyte Corporation; Dr PRP USA LLC; Juventix Regenerative Medical, LLC; Terumo Corporation; Zimmer Biomet; Stryker; Apex Biologix; Celling Biosciences |
Platelet-rich Plasma (PRP) therapy has proven effective and safe in various medical applications, offering benefits such as accelerated healing, enhanced wound closure, reduced swelling and inflammation, and stabilization of bone or soft tissue. These advantages expand the use of PRP in treating chronic ailments, leading to boost market revenue. Platelets play a critical role in wound healing due to their hemostatic function and the presence of growth factors and cytokines. Research studies confirm the safety and affordability of platelet-rich plasma as a regenerative therapy for cutaneous wound healing, improving patient care.
While increased acceptability of PRP in dental and oral surgical procedures, such as managing bisphosphonate-related osteonecrosis of the jaw to enhance wound healing, have also yielded promising results. In the past few years, platelet rich plasma injections have gained significant traction among popular sports professionals, including Jermaine Defoe, Rafael Nadal, Alex Rodriguez, Tiger Woods, and many more. Furthermore, the World Anti-doping Association (WADA) removed the PRP from the prohibited substances list in 2011. Wide application of these products by high profile athletes in the U.S. for early osteoarthritis (OA) and chronic injuries significantly contribute to market growth.
PRP and stem cell-based biological interventions have been proven to accelerate recovery while maintaining the performance of athletes. Moreover, research studies have demonstrated that PRP can be used successfully in combination with other treatments to ensure rapid healing. The effects of PRP therapy combined with 70% glycolic acid effectively manage acne scars. Similarly, PRP along with hyaluronic acid significantly improves skin general appearance, firmness, and texture.
High costs associated with platelet rich plasma products make it difficult for clinicians to deploy this therapy on a large scale, which impedes market growth to some extent. Conversely, insurance firms cover few PRP therapy costs, including diagnostic tests, consultation fees, and other medical expenses. The CMS covers autologous PRP only for patients with chronic non-healing diabetic, venous wounds, or when enrolled in a clinical research study, thus reducing the amount of out-of-pocket charges.
Type Insights
Pure platelet rich plasma dominated the market with a revenue share of 51.83% in 2022. Certain benefits associated with this PRP type, including tissue generation & repair, rapid healing, and enhancement in overall function, have raised the demand for pure PRP across different therapeutic applications. In addition, effective elimination of adverse effects, such as an allergic or immune reaction, with this therapeutic approach has considerably benefitted the segment growth.
Pure platelet rich plasma is considered to be more suitable for application for bone regeneration than leukocyte platelet rich plasma. The combined use of this therapy with β-tricalcium phosphate is reflected to be an effective and safe alternative for the treatment of bone defects. Key players are also providing advanced products in this segment. Pure Spin PRP, a U.S.-based firm, is one such player offering an advanced PRP system for centrifugation with maximum platelet recovery.
Leukocyte-rich PRP (LR-PRP) is anticipated to grow at a CAGR of around 11.02% during the forecast period. LR-PRP promotes bone regeneration by improved viability, proliferation, migration of cells in vitro, osteogenesis, and angiogenesis in vitro & in vivo, however, these products produce harmful effects as compared to pure type. Conversely, these are powerful tools for soft tissue reconstruction with a reduction in operating time, postoperative pain, and risk of complications in wound healing.
Application Insights
In terms of revenue, the orthopedics held the maximum share of 26.73% in 2022 in the U.S. PRP market. Platelet rich plasma injections in orthopedic therapeutics have been recognized to be more convenient as compared to conventional therapeutics as the former stimulates healing and causes degenerative tissue to repair & regenerate itself. Long-lasting relief and optimized healing after surgery are certain advantages associated with this therapy that have supplemented the demand for PRP in soft tissue reconstruction and bone reconstruction.
Furthermore, an increase in the prevalence of OA in the U.S. boosts the demand for this regenerative therapy in orthopedics. As per the Centers for Disease Control and Prevention (CDC), there were 54 million cases of arthritis, and it is estimated that 78.0 million U.S. adults aged 18 years or older would have arthritis by 2040. Moreover, an increase in the number of research studies constantly illustrating the benefits of isolated PRP usage in combination with surgical treatments in the management of OA is set to propel the market growth.
An increase in cosmetic surgeries is set to boost the cosmetic surgery and dermatology segment, poised to grow at the highest CAGR through 2023-2032. As per the American Society of Plastic Surgeons, there were nearly 5.9 million reconstructive procedures in 2019 in the U.S. The rise in the adoption of PRP treatments by plastic surgeons & dermatologists to improve the volume, tone, and texture of the skin also drives segment growth. It reduces the appearance of wrinkles and offers positive aesthetic results when combined with fat grafting procedures.
End-use Insights
The hospital segment accounted for the largest revenue share of 50% in 2022 with a substantial number of hospitals in the U.S. that provide PRP-based therapies. In addition, the growth in the knee OA cases is a major health concern in the U.S. As there are limited or no definitive curative therapies for osteoarthritis, it creates a major opportunity for these therapies to be highly adopted by hospitals for the treatment of knee OA. This factor contributes to the segment’s dominance in the market.
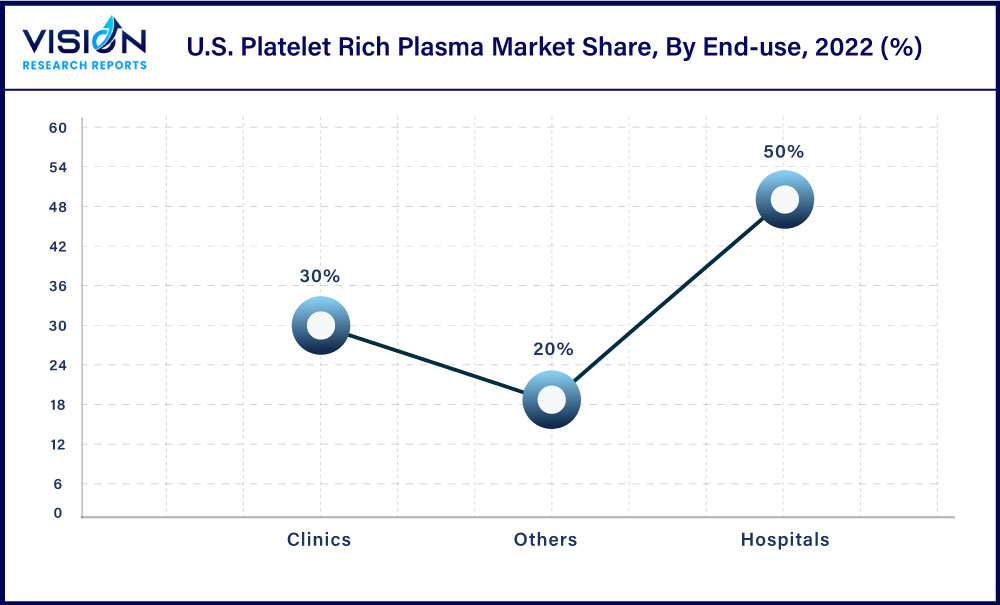
Others end-use segment includes academic institutes, research institutes, and Point-of-care (POC) settings and is estimated to witness the fastest growth rate of 11.33% over the forecast period. Recent product approval for the rapid and safe preparation of this therapy at PoC settings in the U.S. is one of the major factors responsible for high growth. For instance, in September 2021, Royal Biologics recently received FDA 510K approval for their Maxx™-PRP concentration system. This patented next-generation device enables the safe and rapid preparation of platelet-rich plasma (PRP) by concentrating autologous whole blood.
Similarly, the Fidia PRP Kit of the Fidia Farmaceutici s.p.a., an Italy-based company with operations in the U.S., received FDA approval in November 2019 for rapid and safe preparation of autologous platelet rich plasma at the patient’s PoC. Apex Biologix’s XCELL PRP Platelet Concentrating System 60ml was granted approval in April 2019. The availability of these systems and kits for PRP preparation to be used at POC settings is likely to drive the segment at a lucrative pace.
U.S. Platelet Rich Plasma Market Segmentations:
By Type
By Application
By End-use
Chapter 1. Introduction
1.1. Research Objective
1.2. Scope of the Study
1.3. Definition
Chapter 2. Research Methodology
2.1. Research Approach
2.2. Data Sources
2.3. Assumptions & Limitations
Chapter 3. Executive Summary
3.1. Market Snapshot
Chapter 4. Market Variables and Scope
4.1. Introduction
4.2. Market Classification and Scope
4.3. Industry Value Chain Analysis
4.3.1. Raw Material Procurement Analysis
4.3.2. Sales and Distribution Type Analysis
4.3.3. Downstream Buyer Analysis
Chapter 5. COVID 19 Impact on U.S. Platelet Rich Plasma Market
5.1. COVID-19 Landscape: U.S. Platelet Rich Plasma Industry Impact
5.2. COVID 19 - Impact Assessment for the Industry
5.3. COVID 19 Impact: Major Government Policy
5.4. Market Trends and Opportunities in the COVID-19 Landscape
Chapter 6. Market Dynamics Analysis and Trends
6.1. Market Dynamics
6.1.1. Market Drivers
6.1.2. Market Restraints
6.1.3. Market Opportunities
6.2. Porter’s Five Forces Analysis
6.2.1. Bargaining power of suppliers
6.2.2. Bargaining power of buyers
6.2.3. Threat of substitute
6.2.4. Threat of new entrants
6.2.5. Degree of competition
Chapter 7. Competitive Landscape
7.1.1. Company Market Share/Positioning Analysis
7.1.2. Key Strategies Adopted by Players
7.1.3. Vendor Landscape
7.1.3.1. List of Suppliers
7.1.3.2. List of Buyers
Chapter 8. U.S. Platelet Rich Plasma Market, By Type
8.1. U.S. Platelet Rich Plasma Market, by Type, 2023-2032
8.1.1 Pure Platelet Rich Plasma
8.1.1.1. Market Revenue and Forecast (2020-2032)
8.1.2. Leukocyte Rich Platelet Rich Plasma
8.1.2.1. Market Revenue and Forecast (2020-2032)
8.1.3. Others
8.1.3.1. Market Revenue and Forecast (2020-2032)
Chapter 9. U.S. Platelet Rich Plasma Market, By Application
9.1. U.S. Platelet Rich Plasma Market, by Application, 2023-2032
9.1.1. Orthopedics
9.1.1.1. Market Revenue and Forecast (2020-2032)
9.1.2. Sports Medicine
9.1.2.1. Market Revenue and Forecast (2020-2032)
9.1.3. Cosmetic Surgery
9.1.3.1. Market Revenue and Forecast (2020-2032)
9.1.4. Dermatology
9.1.4.1. Market Revenue and Forecast (2020-2032)
9.1.5. Ophthalmic Surgery
9.1.5.1. Market Revenue and Forecast (2020-2032)
9.1.6. Neurosurgery
9.1.6.1. Market Revenue and Forecast (2020-2032)
9.1.7. General Surgery
9.1.7.1. Market Revenue and Forecast (2020-2032)
9.1.8. Others
9.1.8.1. Market Revenue and Forecast (2020-2032)
Chapter 10. U.S. Platelet Rich Plasma Market, By Ophthalmic Surgery
10.1. U.S. Platelet Rich Plasma Market, by Ophthalmic Surgery, 2023-2032
10.1.1. Neurosurgery
10.1.1.1. Market Revenue and Forecast (2020-2032)
10.1.2. General Surgery
10.1.2.1. Market Revenue and Forecast (2020-2032)
10.1.3. Others
10.1.3.1. Market Revenue and Forecast (2020-2032)
Chapter 11. U.S. Platelet Rich Plasma Market, Regional Estimates and Trend Forecast
11.1. U.S.
11.1.1. Market Revenue and Forecast, by Type (2020-2032)
11.1.2. Market Revenue and Forecast, by Application (2020-2032)
11.1.3. Market Revenue and Forecast, by Ophthalmic Surgery (2020-2032)
Chapter 12. Company Profiles
12.1. Johnson & Johnson Services, Inc
12.1.1. Company Overview
12.1.2. Product Offerings
12.1.3. Financial Performance
12.1.4. Recent Initiatives
12.2. Arthrex, Inc.
12.2.1. Company Overview
12.2.2. Product Offerings
12.2.3. Financial Performance
12.2.4. Recent Initiatives
12.3. EmCyte Corporation
12.3.1. Company Overview
12.3.2. Product Offerings
12.3.3. Financial Performance
12.3.4. Recent Initiatives
12.4. Dr PRP USA LLC
12.4.1. Company Overview
12.4.2. Product Offerings
12.4.3. Financial Performance
12.4.4. Recent Initiatives
12.5. Juventix Regenerative Medical, LLC
12.5.1. Company Overview
12.5.2. Product Offerings
12.5.3. Financial Performance
12.5.4. Recent Initiatives
12.6. Terumo Corporation
12.6.1. Company Overview
12.6.2. Product Offerings
12.6.3. Financial Performance
12.6.4. Recent Initiatives
12.7. Zimmer Biomet
12.7.1. Company Overview
12.7.2. Product Offerings
12.7.3. Financial Performance
12.7.4. Recent Initiatives
12.8. Stryker
12.8.1. Company Overview
12.8.2. Product Offerings
12.8.3. Financial Performance
12.8.4. Recent Initiatives
12.9. Apex Biologix
12.9.1. Company Overview
12.9.2. Product Offerings
12.9.3. Financial Performance
12.9.4. Recent Initiatives
12.10. Celling Biosciences
12.10.1. Company Overview
12.10.2. Product Offerings
12.10.3. Financial Performance
12.10.4. Recent Initiatives
Chapter 13. Research Methodology
13.1. Primary Research
13.2. Secondary Research
13.3. Assumptions
Chapter 14. Appendix
14.1. About Us
14.2. Glossary of Terms
 Cross-segment Market Size and Analysis for
Mentioned Segments
Cross-segment Market Size and Analysis for
Mentioned Segments
 Additional Company Profiles (Upto 5 With No Cost)
Additional Company Profiles (Upto 5 With No Cost)
 Additional Countries (Apart From Mentioned Countries)
Additional Countries (Apart From Mentioned Countries)
 Country/Region-specific Report
Country/Region-specific Report
 Go To Market Strategy
Go To Market Strategy
 Region Specific Market Dynamics
Region Specific Market Dynamics Region Level Market Share
Region Level Market Share Import Export Analysis
Import Export Analysis Production Analysis
Production Analysis Others
Others