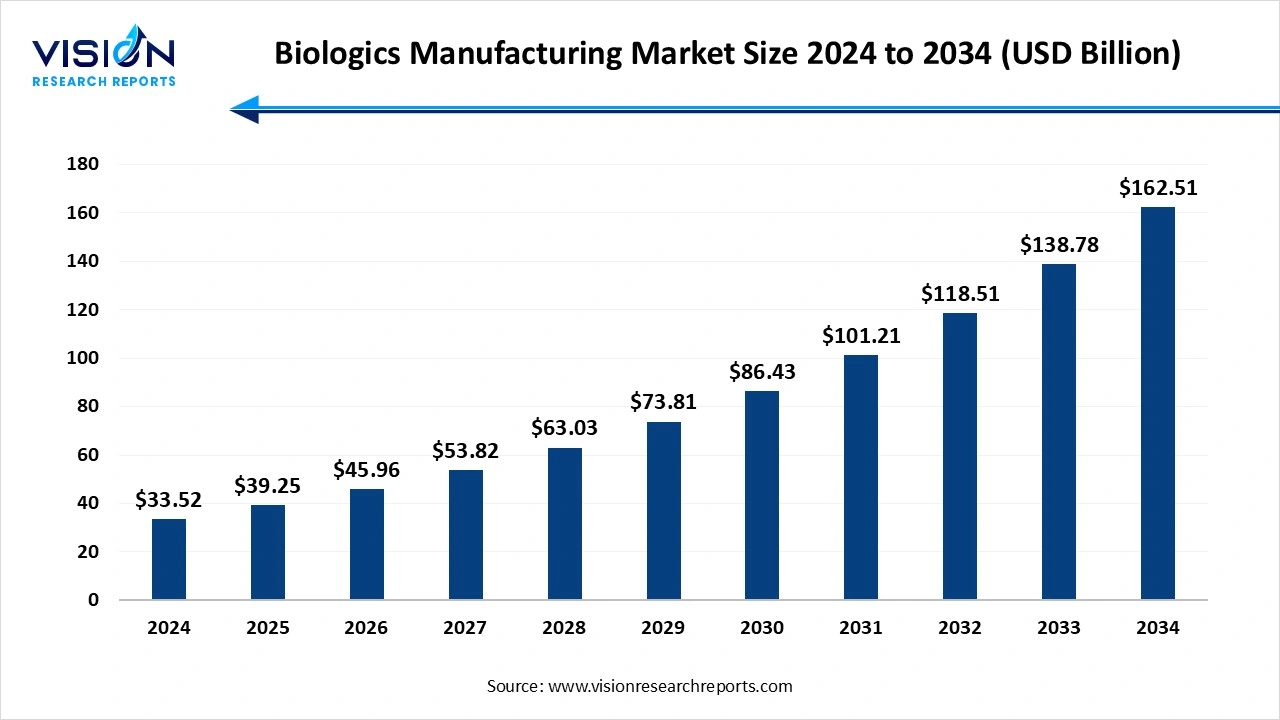
The global biologics manufacturing market size stood at USD 33.52 billion in 2024 and is estimated to reach USD 39.25 billion in 2025. It is projected to hit USD 162.51 billion by 2034, registering a robust CAGR of 17.1% from 2025 to 2034. This strong market expansion is driven by the rising prevalence of chronic diseases, rapid advancements in monoclonal antibody engineering, increasing adoption of cell and gene therapies, and growing demand for targeted and personalized biologics.
The biologics market has maintained strong momentum in biopharmaceutical innovation and targeted therapies. The market continues to reshape the treatment landscape across fields like oncology, immunology and rare diseases. Rising prevalence of chronic conditions, favorable reimbursement policies and accelerated regulatory pathways have driven widespread market adoption. As biologic pipelines continue to mature and diversify, pharmaceutical and biotech companies are focusing more on their investments in research, development and manufacturing capabilities, further expanding the landscape.
Biologic manufacturing is the process of producing medicine with the help of living organisms. Biotech and pharmaceutical companies make use of this approach in order to produce biologics like mAbs (monoclonal antibodies) or mRNA vaccines both at small and large scale, bringing several advantages over traditional small-molecule drug products.
Central practices in biomanufacturing consist of various stages of production, from upstream processes involving cell culturing and harvesting to downstream processes that focus on purification and formulation.
| Report Coverage | Details |
| Market Size in 2024 | USD 33.52 Billion |
| Revenue Forecast by 2034 | USD 162.51 Billion |
| Growth rate from 2025 to 2034 | CAGR of 17.1% |
| Base Year | 2024 |
| Forecast Period | 2025 to 2034 |
| Regions | North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
| Companies Covered |
Samsung Biologics, Amgen Inc., Novo Nordisk A/S, AbbVie Inc., Sanofi, Johnson & Johnson Services Inc., Celltrion Healthcare Co. Ltd., Bristol-Myers Squibb Company, Eli Lilly and Company, F. Hoffmann-La Roche Ltd., Boehringer Ingelheim International GmbH, and Lonza. |
AI is revolutionizing the biologics manufacturing market in various domains like research, development and process development. AI enables a more precise and predictive approach to designing biologics. Digital twins are virtual models of bioreactors can simulate and optimize process parameters before any physical trials begin, which saves both time and resources. Machine learning is seen increasingly being applied to genomic, proteomic and metabolomic data as it deepens our understanding of how cells behave when producing a biologic, thus creating new ways to enhance productivity and quality. AI can also predict optimal media formulations and feeding strategies, replacing traditional trial-and-error work with data-driven accuracy.
Predictive maintenance systems are also a key component as they help to analyze sensor data to predict when equipment will require servicing, this prevents costly downtime and avoids production interruptions. AI also carries out continues monitoring can track critical quality attributes and flag anomalies earlier than manual methods, helping to ensure consistency across every batch. AI can also optimize production scheduling by using throughput data to maximize capacity and efficiency. Through all these factors, we can see how AI and Machine learning is changing the biologics manufacturing landscape, fostering innovation.
Rise of Chronic Diseases
Chronic diseases such as cancer, diabetes, and autoimmune disorders, require long-term management and are on the rise on a global scale. These diseases are often related to complex biological pathways, and traditional therapies cannot effectively manage them. Biologics are targeted to act upon these specific molecules or pathways, providing a more precise and effective approach towards treatment. Unlike traditional therapies, biologics are designed to work alongside with the body's normal processes to provide treatments complementary to the underlying causes of chronic diseases and are not just symptomatic. This targeted approach helps in enhancing therapeutic outcomes while also reducing the likelihood of adverse effects.
Additionally, these chronic disease burdens have put a greater emphasis on treatment options through innovation. Increasing awareness among physicians and patients regarding the benefits of biologics has increased market demand.
High Costs
Despite multiple prospects, the market does have its fair share of challenges that could potentially hinder its growth. The high cost of biologic therapies poses to be a significant restraint. This is because biologics often require intricate, living systems-based manufacturing, which necessitates advanced bioreactors, specialized personnel and stringent regulatory compliance. This can be a challenge in markets who are lacking universal healthcare or robust insurance systems. This disparity slows down market entry and development.
Innovation and Strong Regulatory Landscape
The rising need for innovative and focused treatments is a key opportunity that is propelling the biologics manufacturing market. Targeted biologics, including monoclonal antibodies, antibody-drug conjugates and cell and gene therapies specifically target the molecular causes of any illnesses. These treatments are ideal for diseases such as cancer, autoimmune disorders and rare genetic diseases because of their precision. This increases treatment efficacy and reduces side effects. Pharmaceutical and biotechnology companies al, over the world are seen making significant investments in research pipelines to launch new advanced targeted biologics.
Regulatory bodies worldwide are establishing favorable pathways for their approval to support the adoption of biologics in clinical practice, opening up new avenues of opportunities. Targeted therapy success has also led to increased industry competition, compelling producers to increase production capacity, embrace cutting-edge bioprocessing technologies and form strategic alliances. As businesses continue to work to satisfy the growing demand for novel treatments, the market is witnessing accelerated growth.
North America dominated the market as of this year in 2024. This is because the market’s ecosystem strong. The region also has a strong presence of cutting-edge startups like KBI Biopharma, Oxford Biomedicine and Aldevron. The region is also home to a well-developed healthcare infrastructure, has significant investment in research and development and also a favorable regulatory environment. This regulatory support, coupled with the availability of advanced medical facilities, solidifies the region’s leadership in the biologics market.
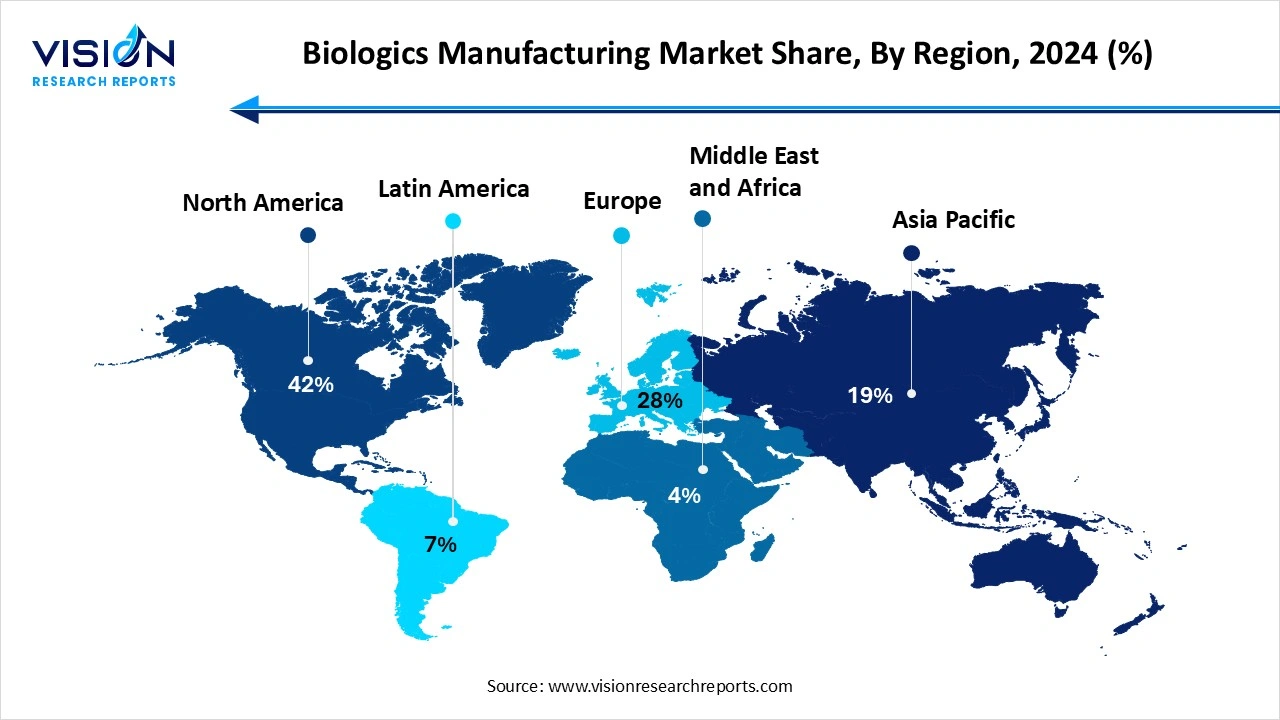
Asia Pacific is expected to grow at the fastest rate during the forecast period. This growth is due to its large population and continuously expanding healthcare infrastructure. The region is also witnessing increasing collaboration between government agencies and biopharma companies are increasing research capabilities, which helps to accelerate drug development and commercialization. The market’s rapid expansion is also a result of supportive policies and a growing focus on innovation.
Which modality segment dominated the market in 2024?
The monoclonal antibodies segment dominated the market in 2024. Thid dominance is because they are widely used to treat infectious diseases, autoimmune disorders and cancer. Strong clinical success rates, high specificity, and fewer side effects than compared to traditional therapies have fueled their widespread adoption. The increasing trend toward biosimilar mAbs and technological developments in bioprocessing further reinforces the segment's position.
The cell and gene therapies segment is expected to grow at the fastest rate during the forecast period. This growth is driven by the increasing number of FDA and EMA approvals, expanding clinical pipelines and strong investment inflows from biopharma companies and venture capital firms worldwide. This segment's growth is also attributed to the growing emphasis on personalized medicine and the expanding partnerships between CDMOs and biopharmaceutical companies.
Which disease category led the market as of this year?
The Oncology segment led the market as of this year. This dominance is due to the high incidence of cancer worldwide and the ever increasingly need for targeted therapies. Advanced MABs have now become standard therapeutic tools in cancer treatment. Biologics are also critical in immuno-oncology, where they modulate immune response against tumor cells. With ongoing clinical trials in areas like CAR-T cell therapy and bispecific antibodies, oncology remains the most dominant and innovation-heavy segment.
The Infectious diseases segment is seen to be the fastest-growing segment throughout the forecast period. This segment’s growth was further propelled by the Covid-19 pandemic and has led to continued focus on vaccines and antivirals. The success of biologic vaccines like mRNA-based COVID-19 vaccines has prompted a surge in development activity for other viral threats like RSV and influenza.
Which source segment dominated the market as of this year?
Mammalian cells dominated the market as of this year in 2024. The advantage of this segment is their ability to produce complex, human-like proteins with appropriate glycosylation. These cells are also suitable for large-scale commercial production and have a proven track record of regulatory approval. As companies continue to innovate in microbial production to improve yield and reduce costs, the demand for biologics will rise, solidifying this segment’s role.
Microbial systems are expected to be the fastest-growing segment, particularly when it comes to vaccines and recombinant proteins. These systems offer high-yield and cost-effective production along with simplified culture requirements. These platforms are also increasingly used for producing biosimilars and non-glycosylated proteins. Innovations in strain engineering and fermentation optimization are making microbial expression systems have enabled for more versatile and scalable options.
Which manufacturing segment held the largest market in 2024?
In-house manufacturing held the largest market share in 2024. This is because this segment enables better control and surveillance over the production process. Controlling the production process leads to better quality assurance, IP protection and seamless regulatory compliance. Various firms all over the world continue to invest in expanding their in-house capacities through advanced, cutting edge biomanufacturing campuses.
Outsourced manufacturing is estimated to be the fastest growing segment during the forecast years. particularly among emerging biotech companies and during clinical trial phases. This growth is driven by the increasing need for companies to reduce costs, enhance operational efficiency and lessen time-to-market. Outsourcing also provides a level of scalability, allowing companies to respond flexibly to the growing global demand. As biologics developers increasingly navigate the complexities, outsourcing remains a cost-effective and efficient solution for maintaining a competitive edge in the market.
By Modality
By Disease
By Source
By Manufacturing
By Region
Biologics Manufacturing Market
Chapter 1. Introduction
1.1. Research Objective
1.2. Scope of the Study
1.3. Definition
Chapter 2. Research Methodology
2.1. Research Approach
2.2. Data Sources
2.3. Assumptions & Limitations
Chapter 3. Executive Summary
3.1. Market Snapshot
Chapter 4. Market Variables and Scope
4.1. Introduction
4.2. Market Classification and Scope
4.3. Industry Value Chain Analysis
4.3.1. Raw Material Procurement Analysis
4.3.2. Sales and Distribution Channel Analysis
4.3.3. Downstream Buyer Analysis
Chapter 5. COVID 19 Impact on Biologics Manufacturing Market
5.1. COVID-19 Landscape: Biologics Manufacturing Industry Impact
5.2. COVID 19 - Impact Assessment for the Industry
5.3. COVID 19 Impact: Global Major Government Policy
5.4. Market Trends and Opportunities in the COVID-19 Landscape
Chapter 6. Market Dynamics Analysis and Trends
6.1. Market Dynamics
6.1.1. Market Drivers
6.1.2. Market Restraints
6.1.3. Market Opportunities
6.2. Porter’s Five Forces Analysis
6.2.1. Bargaining power of suppliers
6.2.2. Bargaining power of buyers
6.2.3. Threat of substitute
6.2.4. Threat of new entrants
6.2.5. Degree of competition
Chapter 7. Competitive Landscape
7.1.1. Company Market Share/Positioning Analysis
7.1.2. Key Strategies Adopted by Players
7.1.3. Vendor Landscape
7.1.3.1. List of Suppliers
7.1.3.2. List of Buyers
Chapter 8. Global Biologics Manufacturing Market, By Modality
8.1. Biologics Manufacturing Market, by Modality
8.1.1. Monoclonal Antibodies (mAbs)
8.1.1.1. Market Revenue and Forecast
8.1.2. Biosimilar & Recombinant Proteins
8.1.2.1. Market Revenue and Forecast
8.1.3. Vaccines (recombinant/mRNA/Viral)
8.1.3.1. Market Revenue and Forecast
8.1.4. Cell & Gene Therapies
8.1.4.1. Market Revenue and Forecast
8.1.5. RNA-based Therapeutics
8.1.5.1. Market Revenue and Forecast
8.1.6. Others
8.1.6.1. Market Revenue and Forecast
Chapter 9. Global Biologics Manufacturing Market, By Disease
9.1. Biologics Manufacturing Market, by Disease
9.1.1. Oncology
9.1.1.1. Market Revenue and Forecast
9.1.2. Infectious Diseases
9.1.2.1. Market Revenue and Forecast
9.1.3. Immunological Disorders
9.1.3.1. Market Revenue and Forecast
9.1.4. Cardiovascular Disorders
9.1.4.1. Market Revenue and Forecast
9.1.5. Hematological Disorders
9.1.5.1. Market Revenue and Forecast
9.1.6. Others
9.1.6.1. Market Revenue and Forecast
Chapter 10. Global Biologics Manufacturing Market, By Source
10.1. Biologics Manufacturing Market, by Source
10.1.1. Microbial
10.1.1.1. Market Revenue and Forecast
10.1.2. Mammalian
10.1.2.1. Market Revenue and Forecast
10.1.3. Others
10.1.3.1. Market Revenue and Forecast
Chapter 11. Global Biologics Manufacturing Market, By Manufacturing
11.1. Biologics Manufacturing Market, by Manufacturing
11.1.1. Outsourced
11.1.1.1. Market Revenue and Forecast
11.1.2. In-house
11.1.2.1. Market Revenue and Forecast
Chapter 12. Global Biologics Manufacturing Market, Regional Estimates and Trend Forecast
12.1. North America
12.1.1. Market Revenue and Forecast, by Modality
12.1.2. Market Revenue and Forecast, by Disease
12.1.3. Market Revenue and Forecast, by Source
12.1.4. Market Revenue and Forecast, by Manufacturing
12.1.5. U.S.
12.1.5.1. Market Revenue and Forecast, by Modality
12.1.5.2. Market Revenue and Forecast, by Disease
12.1.5.3. Market Revenue and Forecast, by Source
12.1.5.4. Market Revenue and Forecast, by Manufacturing
12.1.6. Rest of North America
12.1.6.1. Market Revenue and Forecast, by Modality
12.1.6.2. Market Revenue and Forecast, by Disease
12.1.6.3. Market Revenue and Forecast, by Source
12.1.6.4. Market Revenue and Forecast, by Manufacturing
12.2. Europe
12.2.1. Market Revenue and Forecast, by Modality
12.2.2. Market Revenue and Forecast, by Disease
12.2.3. Market Revenue and Forecast, by Source
12.2.4. Market Revenue and Forecast, by Manufacturing
12.2.5. UK
12.2.5.1. Market Revenue and Forecast, by Modality
12.2.5.2. Market Revenue and Forecast, by Disease
12.2.5.3. Market Revenue and Forecast, by Source
12.2.5.4. Market Revenue and Forecast, by Manufacturing
12.2.6. Germany
12.2.6.1. Market Revenue and Forecast, by Modality
12.2.6.2. Market Revenue and Forecast, by Disease
12.2.6.3. Market Revenue and Forecast, by Source
12.2.6.4. Market Revenue and Forecast, by Manufacturing
12.2.7. France
12.2.7.1. Market Revenue and Forecast, by Modality
12.2.7.2. Market Revenue and Forecast, by Disease
12.2.7.3. Market Revenue and Forecast, by Source
12.2.7.4. Market Revenue and Forecast, by Manufacturing
12.2.8. Rest of Europe
12.2.8.1. Market Revenue and Forecast, by Modality
12.2.8.2. Market Revenue and Forecast, by Disease
12.2.8.3. Market Revenue and Forecast, by Source
12.2.8.4. Market Revenue and Forecast, by Manufacturing
12.3. APAC
12.3.1. Market Revenue and Forecast, by Modality
12.3.2. Market Revenue and Forecast, by Disease
12.3.3. Market Revenue and Forecast, by Source
12.3.4. Market Revenue and Forecast, by Manufacturing
12.3.5. India
12.3.5.1. Market Revenue and Forecast, by Modality
12.3.5.2. Market Revenue and Forecast, by Disease
12.3.5.3. Market Revenue and Forecast, by Source
12.3.5.4. Market Revenue and Forecast, by Manufacturing
12.3.6. China
12.3.6.1. Market Revenue and Forecast, by Modality
12.3.6.2. Market Revenue and Forecast, by Disease
12.3.6.3. Market Revenue and Forecast, by Source
12.3.6.4. Market Revenue and Forecast, by Manufacturing
12.3.7. Japan
12.3.7.1. Market Revenue and Forecast, by Modality
12.3.7.2. Market Revenue and Forecast, by Disease
12.3.7.3. Market Revenue and Forecast, by Source
12.3.7.4. Market Revenue and Forecast, by Manufacturing
12.3.8. Rest of APAC
12.3.8.1. Market Revenue and Forecast, by Modality
12.3.8.2. Market Revenue and Forecast, by Disease
12.3.8.3. Market Revenue and Forecast, by Source
12.3.8.4. Market Revenue and Forecast, by Manufacturing
12.4. MEA
12.4.1. Market Revenue and Forecast, by Modality
12.4.2. Market Revenue and Forecast, by Disease
12.4.3. Market Revenue and Forecast, by Source
12.4.4. Market Revenue and Forecast, by Manufacturing
12.4.5. GCC
12.4.5.1. Market Revenue and Forecast, by Modality
12.4.5.2. Market Revenue and Forecast, by Disease
12.4.5.3. Market Revenue and Forecast, by Source
12.4.5.4. Market Revenue and Forecast, by Manufacturing
12.4.6. North Africa
12.4.6.1. Market Revenue and Forecast, by Modality
12.4.6.2. Market Revenue and Forecast, by Disease
12.4.6.3. Market Revenue and Forecast, by Source
12.4.6.4. Market Revenue and Forecast, by Manufacturing
12.4.7. South Africa
12.4.7.1. Market Revenue and Forecast, by Modality
12.4.7.2. Market Revenue and Forecast, by Disease
12.4.7.3. Market Revenue and Forecast, by Source
12.4.7.4. Market Revenue and Forecast, by Manufacturing
12.4.8. Rest of MEA
12.4.8.1. Market Revenue and Forecast, by Modality
12.4.8.2. Market Revenue and Forecast, by Disease
12.4.8.3. Market Revenue and Forecast, by Source
12.4.8.4. Market Revenue and Forecast, by Manufacturing
12.5. Latin America
12.5.1. Market Revenue and Forecast, by Modality
12.5.2. Market Revenue and Forecast, by Disease
12.5.3. Market Revenue and Forecast, by Source
12.5.4. Market Revenue and Forecast, by Manufacturing
12.5.5. Brazil
12.5.5.1. Market Revenue and Forecast, by Modality
12.5.5.2. Market Revenue and Forecast, by Disease
12.5.5.3. Market Revenue and Forecast, by Source
12.5.5.4. Market Revenue and Forecast, by Manufacturing
12.5.6. Rest of LATAM
12.5.6.1. Market Revenue and Forecast, by Modality
12.5.6.2. Market Revenue and Forecast, by Disease
12.5.6.3. Market Revenue and Forecast, by Source
12.5.6.4. Market Revenue and Forecast, by Manufacturing
Chapter 13. Company Profiles
13.1. Samsung Biologics
13.1.1. Company Overview
13.1.2. Product Offerings
13.1.3. Financial Performance
13.1.4. Recent Initiatives
13.2. Amgen Inc.
13.2.1. Company Overview
13.2.2. Product Offerings
13.2.3. Financial Performance
13.2.4. Recent Initiatives
13.3. Novo Nordisk A/S
13.3.1. Company Overview
13.3.2. Product Offerings
13.3.3. Financial Performance
13.3.4. Recent Initiatives
13.4. AbbVie Inc.
13.4.1. Company Overview
13.4.2. Product Offerings
13.4.3. Financial Performance
13.4.4. Recent Initiatives
13.5. Sanofi
13.5.1. Company Overview
13.5.2. Product Offerings
13.5.3. Financial Performance
13.5.4. Recent Initiatives
13.6. Johnson & Johnson Services, Inc.
13.6.1. Company Overview
13.6.2. Product Offerings
13.6.3. Financial Performance
13.6.4. Recent Initiatives
13.7. Celltrion Healthcare Co., Ltd.
13.7.1. Company Overview
13.7.2. Product Offerings
13.7.3. Financial Performance
13.7.4. Recent Initiatives
13.8. Bristol-Myers Squibb Company
13.8.1. Company Overview
13.8.2. Product Offerings
13.8.3. Financial Performance
13.8.4. Recent Initiatives
13.9. Eli Lilly and Company
13.9.1. Company Overview
13.9.2. Product Offerings
13.9.3. Financial Performance
13.9.4. Recent Initiatives
13.10. F. Hoffmann La-Roche Ltd.
13.10.1. Company Overview
13.10.2. Product Offerings
13.10.3. Financial Performance
13.10.4. Recent Initiatives
Chapter 14. Research Methodology
14.1. Primary Research
14.2. Secondary Research
14.3. Assumptions
Chapter 15. Appendix
15.1. About Us
15.2. Glossary of Terms
 Cross-segment Market Size and Analysis for
Mentioned Segments
Cross-segment Market Size and Analysis for
Mentioned Segments
 Additional Company Profiles (Upto 5 With No Cost)
Additional Company Profiles (Upto 5 With No Cost)
 Additional Countries (Apart From Mentioned Countries)
Additional Countries (Apart From Mentioned Countries)
 Country/Region-specific Report
Country/Region-specific Report
 Go To Market Strategy
Go To Market Strategy
 Region Specific Market Dynamics
Region Specific Market Dynamics Region Level Market Share
Region Level Market Share Import Export Analysis
Import Export Analysis Production Analysis
Production Analysis Others
Others