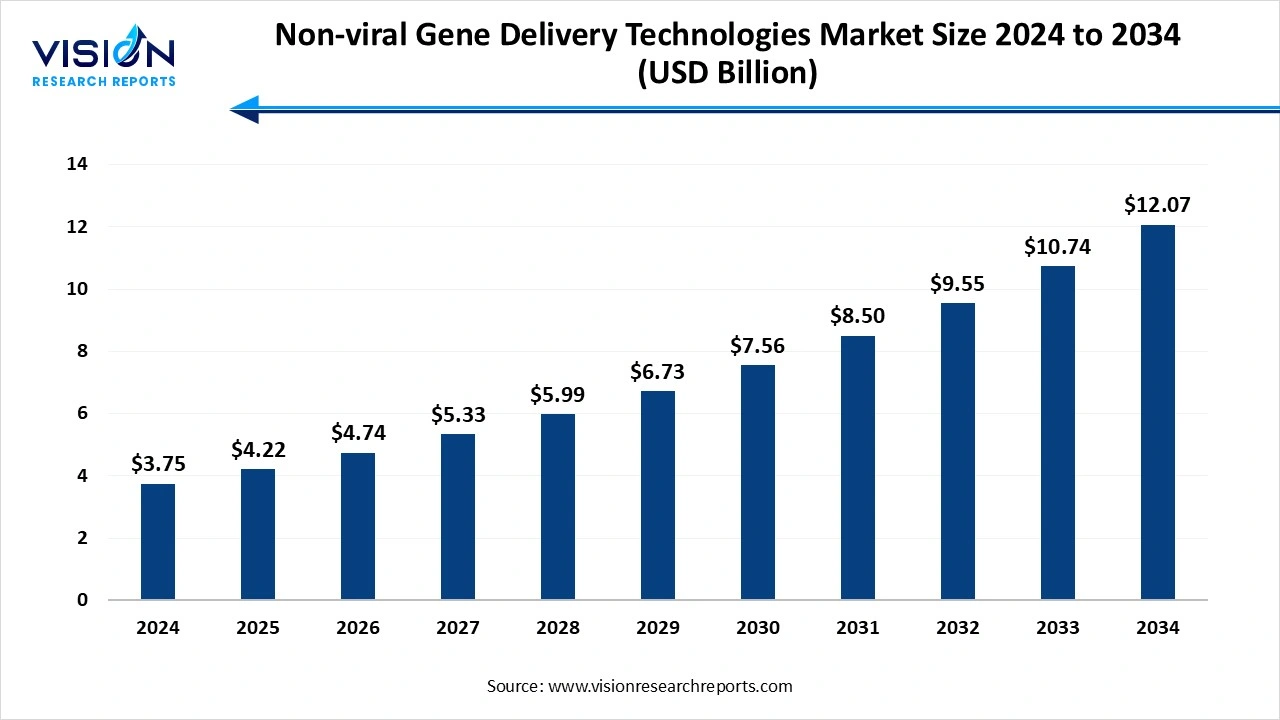
The global non-viral gene delivery technologies market was size valued at USD 3.75 billion in 2024, is projected to hit USD 4.22 billion in 2025, and is expected to hit USD 12.07 billion by 2034, growing at a CAGR of 12.4% during the forecast period. The market Growth is driven by the rising demand for personalized medicine, expanding use of CRISPR and mRNA therapeutics, and increasing cell and gene therapy applications. North America dominated the market in 2024, while Asia-Pacific is expected to grow the fastest, with gradual expansion across the Middle East and Africa.
The non-viral gene delivery technologies market is evolving rapidly, serving as a vital component for modern biotechnology and gene therapy strategies. Unlike viral vectors, non-viral methods provide a safer, more controllable, and more scalable way for gene delivery. It is particularly essential in applications that require repeated dosing or large-scale manufacturing. These types of technologies use synthetic or physical methods to transport genetic material into host cells, thus avoiding immunogenic risks and regulatory challenges that are often associated with viral vectors.
The non-viral gene delivery technologies market is witnessing rapid innovation, especially in areas such as lipid nanoparticles, polymers and CRISPR or Cas9-based delivery. Advancements in nanotechnology have led to the development of highly efficient, targeted delivery systems that have reduced toxicity. Continuous research and development efforts are underway, helping in improving the stability, payload capacity and specificity of these delivery methods. This in turn, enhances therapeutic outcomes in genetic disorders, oncology and immunotherapy.
The non-viral gene delivery technologies market is expected to see rapid growth in the upcoming years. This growth can be attributed to the rising demand for personalized medicine, the increasing adoption in cancer and rare disease research, the advancement of CRISPR and other advanced gene editing technologies, the rapid expansion of mRNA-based therapeutics and the robust growth of cell therapy and tissue engineering.
Key trends propelling the market forward include advancements in lipid nanoparticles and polymer-based vectors, integration of artificial intelligence and computational modeling, the emergence of next-generation lipid nanoparticles (LNPs) and continued progress in nanotechnology.
| Report Coverage | Details |
| Market Size in 2024 | USD 3.75 billion |
| Revenue Forecast by 2034 | USD 12.07 billion |
| Growth rate from 2025 to 2034 | CAGR of 12.4% |
| Base Year | 2024 |
| Forecast Period | 2025 to 2034 |
| Regions | North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
| Companies Covered | Thermo Fisher Scientific Inc., GenScript, Danaher, Merck KGaA, Bio-Rad Laboratories, Altogen Biosystems, Lonza, Sonidel, SIRION BIOTECH GmbH, and Innovative Cell Technologies, Inc. |
One of the major drivers driving the non-viral gene delivery technologies market is the rising incidence of genetic disorders and chronic diseases all over the world. As complex conditions like cystic fibrosis, muscular dystrophy and various cancers continue to rise, there is also a heightened need for innovative therapeutic approaches that are able to accurately target and correct genetic anomalies. Nonviral gene delivery systems are characterized by their reduced immunogenicity and lower risk of insertional mutagenesis than compared to viral vectors, and as a result, they being increasingly adopted in both, preclinical and clinical settings.
The increasing popularity of lipid nanoparticles in mRNA vaccines is another such key driver in the market. This technology has proven to be quite beneficial in the delivery of mRNA vaccines, as its potential extends to other therapeutic applications, including cancer immunotherapies. Additionally, the rise of CRISPR-based gene editing technologies is also up boosting the demand for non-viral delivery systems. As research in the field progresses more and more, the development of more efficient and targeted delivery methods is likely to shape market trends. Companies worldwide are also actively focusing on reducing the cost of gene delivery technologies, making them more accessible for a wider range of applications.
Despite promising growth prospects, the market does have its fair share of challenges. One of the key challenges faced by the market is ensuring the efficient and targeted delivery of therapeutic genes. Non-viral delivery methods are quite safe but often face challenges in achieving the desired efficiency in gene transfer. Additionally, there are high costs associated with the development and production of these technologies. This could limit their widespread adoption and make it difficult for small scale and medium scale companies to keep up with the market fluctuations.
One of the most prominent opportunities in the market is the development of next-generation nonviral vectors that combine high delivery efficiency with minimal toxicity and immunogenicity. Advancements in nanotechnology, materials science and synthetic biology are boosting the design of multifunctional carriers capable of targeted delivery, controlled release and real-time tracking of genetic payloads. These innovations show immense potential in opening new avenues for the treatment of complex diseases and the development of personalized therapies.
The market is also witnessing a surge in research and development activities. These activities are supported by substantial investments from both, public as well as private sectors. Governments and research organizations all across the world are actively allocating significant funds in order to advance gene therapy research. There is also an increasing focus on improving the safety and efficacy of gene delivery methods. Pharmaceutical and biotechnology companies are seen actively partnering up with academic institutions to develop next generation non-viral delivery platforms, with aims to overcome the limitations that are associated with traditional viral vectors.
North America led the market share of 42% in 2024. The region has a high concentration of biotechnology companies, top-tier academic research institutions and also benefits from favorable regulatory pathways that support innovation in gene therapy and drug delivery technologies. Moreover, the region is equipped with a strong IP ecosystem and venture capital availability, making it a hotbed for non-viral delivery innovations. The region is also home to key companies such as Moderna and Intellia, helping North America maintain its position as a global leader and competitor.
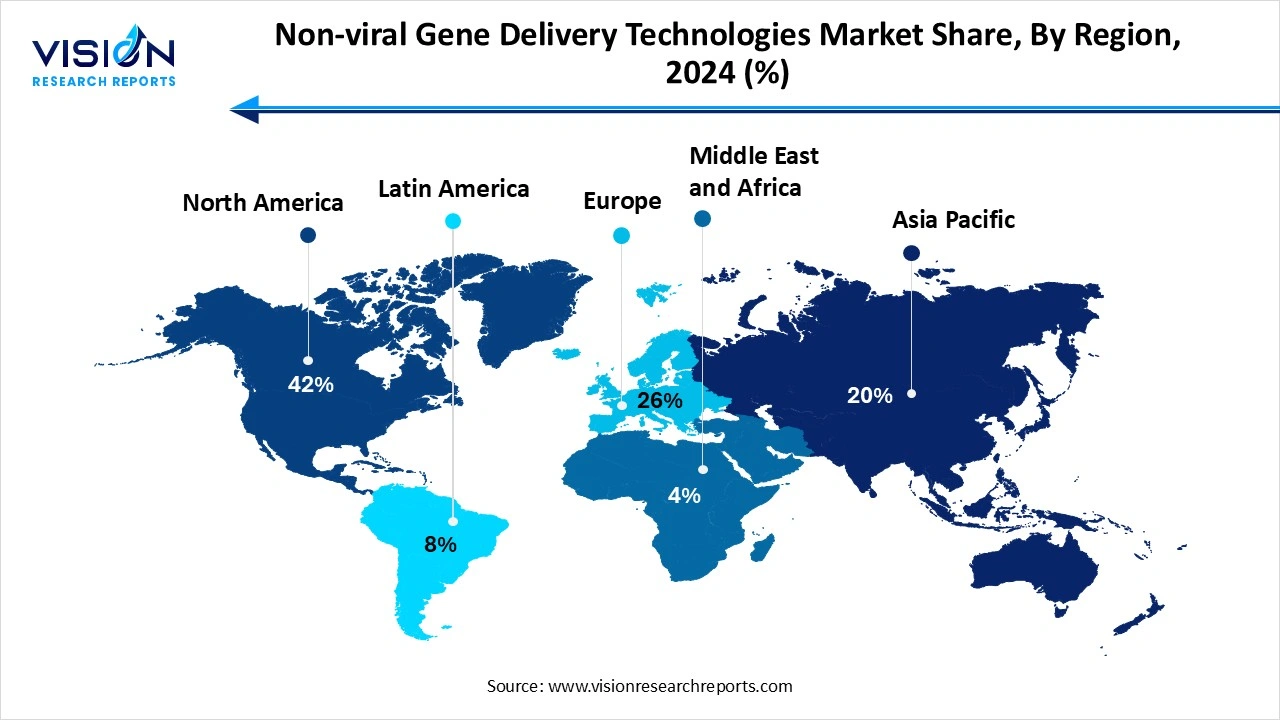
Asia Pacific region is seen to experience the fastest growth throughout the forecast period. This growth is driven by increasing investments in biotechnology infrastructure and favorable government policies. Countries like China, South Korea and Japan are seen rapidly advancing their capabilities in gene therapy and mRNA therapeutics. Moreover, the growing demand for affordable and accessible therapies, a large growing population and rising healthcare awareness all contribute to the region’s growth and development.
Which mode dominated the market in 2024?
The chemical mode segment dominated the market in 2024. With lipid nanoparticles (LNPs) being the most dominant sub-segment. This segment has gained immense popularity during the global Covid-19 pandemic and its connection with mRNA vaccines. The advantage of this segment lies in its ability to encapsulate, protect and facilitate the cellular uptake of nucleic acids without triggering immune responses. This has positioned them as a cornerstone in research and commercial applications.
The physical mode segment is seen to have the fastest growth rate, with polymer-based vectors being the most popular sub segment. They are gaining traction due to their tunable properties and biodegradability. The segment offers various alternatives, particularly for DNA-based therapies and CRISPR applications. The segment is set to expand even more in the near future as new startups enter the field with solutions that are aimed at overcoming traditional limitations of delivery efficiency and toxicity.
Which application led the market as of this year?
The gene therapy led the market as of this year. This dominance can be attributed to the growing number of genetic and acquired diseases that can be treated through targeted gene modification. Nonviral gene delivery systems are increasingly being used in both ex vivo and in vivo gene therapy protocols as they offer a safer alternative to viral vectors, especially in applications that require repeated dosing or large-scale gene transfer. The ability to deliver not only DNA but also RNA and gene-editing components such as CRISPR-Cas9 has driven the growth and development of this particular segment.
The cell therapy is seen to be the fastest growing segment. These therapies often require ex vivo manipulation of immune cells, making electroporation and polymer-based delivery a vital process for successful gene insertion. Vaccine development, especially personalized cancer vaccines is said to be a high-growth area, highlighting the broad role of non-viral vectors beyond traditional genetic disease treatment.
Which end user held the largest market share in 2024?
The biotechnology and biopharmaceutical companies held the largest market share in 2024. This is due to their extensive investment in developing and commercializing gene and cell therapies. Key market players all over the world are actively utilizing non-viral vectors for delivering RNA therapeutics and CRISPR tools. These organizations are also seen investing heavily in research and development activities in order to develop novel therapeutics and delivery platforms.
The academic and research institutes are expected to have the fastest growth rate throughout the forecast period. These institutions are at the forefront of innovation, driving the discovery of new delivery vectors, optimizing existing methods and exploring novel applications in gene editing and functional genomics. The increasing availability of advanced research tools and funding support further enables academic centers to contribute in the development and validation of nonviral gene delivery technologies.
By Mode
By Application
By End User
By Region
Non-viral Gene Delivery Technologies Market
Chapter 1. Introduction
1.1. Research Objective
1.2. Scope of the Study
1.3. Definition
Chapter 2. Research Methodology
2.1. Research Approach
2.2. Data Sources
2.3. Assumptions & Limitations
Chapter 3. Executive Summary
3.1. Market Snapshot
Chapter 4. Market Variables and Scope
4.1. Introduction
4.2. Market Classification and Scope
4.3. Industry Value Chain Analysis
4.3.1. Raw Material Procurement Analysis
4.3.2. Sales and Distribution Mode Analysis
4.3.3. Downstream Buyer Analysis
Chapter 5. COVID 19 Impact on Non-viral Gene Delivery Technologies Market
5.1. COVID-19 Landscape: Non-viral Gene Delivery Technologies Industry Impact
5.2. COVID 19 - Impact Assessment for the Industry
5.3. COVID 19 Impact: Global Major Government Policy
5.4. Market Trends and Opportunities in the COVID-19 Landscape
Chapter 6. Market Dynamics Analysis and Trends
6.1. Market Dynamics
6.1.1. Market Drivers
6.1.2. Market Restraints
6.1.3. Market Opportunities
6.2. Porter’s Five Forces Analysis
6.2.1. Bargaining power of suppliers
6.2.2. Bargaining power of buyers
6.2.3. Threat of substitute
6.2.4. Threat of new entrants
6.2.5. Degree of competition
Chapter 7. Competitive Landscape
7.1.1. Company Market Share/Positioning Analysis
7.1.2. Key Strategies Adopted by Players
7.1.3. Vendor Landscape
7.1.3.1. List of Suppliers
7.1.3.2. List of Buyers
Chapter 8. Global Non-viral Gene Delivery Technologies Market, By Mode
8.1. Non-viral Gene Delivery Technologies Market, by Mode
8.1.1 Chemical
8.1.1.1. Market Revenue and Forecast
8.1.2. Physical
8.1.2.1. Market Revenue and Forecast
Chapter 9. Global Non-viral Gene Delivery Technologies Market, By Application
9.1. Non-viral Gene Delivery Technologies Market, by Application
9.1.1. Gene Therapy
9.1.1.1. Market Revenue and Forecast
9.1.2. Cell Therapy
9.1.2.1. Market Revenue and Forecast
9.1.3. Vaccines
9.1.3.1. Market Revenue and Forecast
9.1.4. Others
9.1.4.1. Market Revenue and Forecast
Chapter 10. Global Non-viral Gene Delivery Technologies Market, By End User
10.1. Non-viral Gene Delivery Technologies Market, by End User
10.1.1. Biotechnology and Biopharmaceutical Companies
10.1.1.1. Market Revenue and Forecast
10.1.2. Research and Academic Institutes
10.1.2.1. Market Revenue and Forecast
10.1.3. Others
10.1.3.1. Market Revenue and Forecast
Chapter 11. Global Non-viral Gene Delivery Technologies Market, Regional Estimates and Trend Forecast
11.1. North America
11.1.1. Market Revenue and Forecast, by Mode
11.1.2. Market Revenue and Forecast, by Application
11.1.3. Market Revenue and Forecast, by End User
11.1.4. U.S.
11.1.4.1. Market Revenue and Forecast, by Mode
11.1.4.2. Market Revenue and Forecast, by Application
11.1.4.3. Market Revenue and Forecast, by End User
11.1.5. Rest of North America
11.1.5.1. Market Revenue and Forecast, by Mode
11.1.5.2. Market Revenue and Forecast, by Application
11.1.5.3. Market Revenue and Forecast, by End User
11.2. Europe
11.2.1. Market Revenue and Forecast, by Mode
11.2.2. Market Revenue and Forecast, by Application
11.2.3. Market Revenue and Forecast, by End User
11.2.4. UK
11.2.4.1. Market Revenue and Forecast, by Mode
11.2.4.2. Market Revenue and Forecast, by Application
11.2.4.3. Market Revenue and Forecast, by End User
11.2.5. Germany
11.2.5.1. Market Revenue and Forecast, by Mode
11.2.5.2. Market Revenue and Forecast, by Application
11.2.5.3. Market Revenue and Forecast, by End User
11.2.6. France
11.2.6.1. Market Revenue and Forecast, by Mode
11.2.6.2. Market Revenue and Forecast, by Application
11.2.6.3. Market Revenue and Forecast, by End User
11.2.7. Rest of Europe
11.2.7.1. Market Revenue and Forecast, by Mode
11.2.7.2. Market Revenue and Forecast, by Application
11.2.7.3. Market Revenue and Forecast, by End User
11.3. APAC
11.3.1. Market Revenue and Forecast, by Mode
11.3.2. Market Revenue and Forecast, by Application
11.3.3. Market Revenue and Forecast, by End User
11.3.4. India
11.3.4.1. Market Revenue and Forecast, by Mode
11.3.4.2. Market Revenue and Forecast, by Application
11.3.4.3. Market Revenue and Forecast, by End User
11.3.5. China
11.3.5.1. Market Revenue and Forecast, by Mode
11.3.5.2. Market Revenue and Forecast, by Application
11.3.5.3. Market Revenue and Forecast, by End User
11.3.6. Japan
11.3.6.1. Market Revenue and Forecast, by Mode
11.3.6.2. Market Revenue and Forecast, by Application
11.3.6.3. Market Revenue and Forecast, by End User
11.3.7. Rest of APAC
11.3.7.1. Market Revenue and Forecast, by Mode
11.3.7.2. Market Revenue and Forecast, by Application
11.3.7.3. Market Revenue and Forecast, by End User
11.4. MEA
11.4.1. Market Revenue and Forecast, by Mode
11.4.2. Market Revenue and Forecast, by Application
11.4.3. Market Revenue and Forecast, by End User
11.4.4. GCC
11.4.4.1. Market Revenue and Forecast, by Mode
11.4.4.2. Market Revenue and Forecast, by Application
11.4.4.3. Market Revenue and Forecast, by End User
11.4.5. North Africa
11.4.5.1. Market Revenue and Forecast, by Mode
11.4.5.2. Market Revenue and Forecast, by Application
11.4.5.3. Market Revenue and Forecast, by End User
11.4.6. South Africa
11.4.6.1. Market Revenue and Forecast, by Mode
11.4.6.2. Market Revenue and Forecast, by Application
11.4.6.3. Market Revenue and Forecast, by End User
11.4.7. Rest of MEA
11.4.7.1. Market Revenue and Forecast, by Mode
11.4.7.2. Market Revenue and Forecast, by Application
11.4.7.3. Market Revenue and Forecast, by End User
11.5. Latin America
11.5.1. Market Revenue and Forecast, by Mode
11.5.2. Market Revenue and Forecast, by Application
11.5.3. Market Revenue and Forecast, by End User
11.5.4. Brazil
11.5.4.1. Market Revenue and Forecast, by Mode
11.5.4.2. Market Revenue and Forecast, by Application
11.5.4.3. Market Revenue and Forecast, by End User
11.5.5. Rest of LATAM
11.5.5.1. Market Revenue and Forecast, by Mode
11.5.5.2. Market Revenue and Forecast, by Application
11.5.5.3. Market Revenue and Forecast, by End User
Chapter 12. Company Profiles
12.1. Thermo Fisher Scientific Inc.
12.1.1. Company Overview
12.1.2. Product Offerings
12.1.3. Financial Performance
12.1.4. Recent Initiatives
12.2. GenScript.
12.2.1. Company Overview
12.2.2. Product Offerings
12.2.3. Financial Performance
12.2.4. Recent Initiatives
12.3. Danaher.
12.3.1. Company Overview
12.3.2. Product Offerings
12.3.3. Financial Performance
12.3.4. Recent Initiatives
12.4. Merck KGaA.
12.4.1. Company Overview
12.4.2. Product Offerings
12.4.3. Financial Performance
12.4.4. Recent Initiatives
12.5. Bio-Rad Laboratories.
12.5.1. Company Overview
12.5.2. Product Offerings
12.5.3. Financial Performance
12.5.4. Recent Initiatives
12.6. Altogen Biosystems
12.6.1. Company Overview
12.6.2. Product Offerings
12.6.3. Financial Performance
12.6.4. Recent Initiatives
12.7. Lonza
12.7.1. Company Overview
12.7.2. Product Offerings
12.7.3. Financial Performance
12.7.4. Recent Initiatives
12.8. Sonidel
12.8.1. Company Overview
12.8.2. Product Offerings
12.8.3. Financial Performance
12.8.4. Recent Initiatives
12.9. SIRION BIOTECH GmbH.
12.9.1. Company Overview
12.9.2. Product Offerings
12.9.3. Financial Performance
12.9.4. Recent Initiatives
12.10. Innovative Cell Technologies, Inc.
12.10.1. Company Overview
12.10.2. Product Offerings
12.10.3. Financial Performance
12.10.4. Recent Initiatives
Chapter 13. Research Methodology
13.1. Primary Research
13.2. Secondary Research
13.3. Assumptions
Chapter 14. Appendix
14.1. About Us
14.2. Glossary of Terms
 Cross-segment Market Size and Analysis for
Mentioned Segments
Cross-segment Market Size and Analysis for
Mentioned Segments
 Additional Company Profiles (Upto 5 With No Cost)
Additional Company Profiles (Upto 5 With No Cost)
 Additional Countries (Apart From Mentioned Countries)
Additional Countries (Apart From Mentioned Countries)
 Country/Region-specific Report
Country/Region-specific Report
 Go To Market Strategy
Go To Market Strategy
 Region Specific Market Dynamics
Region Specific Market Dynamics Region Level Market Share
Region Level Market Share Import Export Analysis
Import Export Analysis Production Analysis
Production Analysis Others
Others